Question stringlengths 2 3k | label int64 0 1 |
|---|---|
What does toe jam cheese taste like? | 0 |
17. Consider the function $f(x)=\left\{\begin{array}{ll}x-[x]-\frac{1}{2}, & \text { If } x \notin I \\ 0, & \text { If } x \in I\end{array}\right.$ where [.] denotes the fractional integral function and $I$ is the set of integers. Then find $g(x)=\max .\left\{x^{2}, f(x),|x|\right\} ;-2 \leq x \leq 2$. | 1 |
What are most important things in life? | 0 |
23. The phases of the light wave at $c, d, e$ and $f$ are $\varphi_{c}, \varphi_{d}, \varphi_{e}$ and $\varphi_{f}$ respectively. It is given that $\varphi_{c} \neq \varphi_{f} \quad
(a) $\varphi_{c}$ cannot be equal to $\varphi_{d}$
(b) $\varphi_{d}$ can be equal to $\varphi_{e}$
(c) $\left(\varphi_{d}-\varphi_{f}\... | 1 |
What produces tracks similar to the fossilized tracks discovered by paleontologists? | 0 |
Aside from Shakespeare, what is another book that is a major focus of textual criticism? | 0 |
22. Consider a $p-n$ junction diode having the characteristic $i=i_{0}\left(e^{e V / k T}-1\right)$ where $i_{0}=20 \mu \mathrm{A}$. The diode is operated at $T=300 \mathrm{~K}$. (a) Find the current through the diode when a voltage of $300 \mathrm{mV}$ is applied across it in forward bias. (b) At what voltage does the... | 1 |
Which is best book for preparing CLAT 2017? | 0 |
What are the best learning sites for Python? | 0 |
15. The chloride of a metal ' M ' contains $47.23 \%$ of the metal. 1 g of this metal displaced from a compound 0.88 g of another metal ' N '. Find the equivalent weight of ' M ' and ' N ' respectively.
(31.77 ; 27.96) | 1 |
Do you think the current Indian education system is correct? If not, then give your suggestions. | 0 |
Why did Arnab Goswami left ET now? | 0 |
What where people asked to do in these research studies? | 0 |
Example 13.16 Gravitational potential varies along $x$-axis as shown in figure.
(a) Plot E versus $x$ graph corresponding to given $V$-x graph.
(b) A mass of $2 \mathrm{~kg}$ is kept at $x=3 \mathrm{~m}$. Find gravitational force on it. | 1 |
14. Statement 1: If $\left(a^{2}-4\right) x^{2}+\left(a^{2}-3 a+2\right) x+\left(a^{2}-7 a+10\right)$ $=0$ is an identity, then the value of $a$ is 2 .
Statement 2: If $a-b=0$, then $a x^{2}+b x+c=0$ is an identity. | 1 |
55. For which of the following reactions, $\Delta H$ is equal to $\Delta U$ ?
(a) $2 \mathrm{HI}(\mathrm{g}) \rightleftharpoons \mathrm{H}_{2}(\mathrm{~g})+\mathrm{I}_{2}(\mathrm{~g})$
(b) $2 \mathrm{NO}_{2}(\mathrm{~g}) \rightleftharpoons \mathrm{N}_{2} \mathrm{O}_{4}(\mathrm{~g})$
(c) $\mathrm{N}_{2}(\mathrm{~g})+3 \... | 1 |
22. The angular velocity of the engine (and hence of the wheel) of a scooter is proportional to the petrol input per second. The scooter is moving on a frictionless road with uniform velocity. If the petrol input is increased by $10 \%$, the linear velocity of the scooter is increased by
(a) $50 \%$
(b) $10 \%$
(c) $20... | 1 |
Which is the highest paid job in civil engineering field? | 0 |
Slovene is a major language in what country? | 0 |
20. If the energy difference between the ground state of an atom and its excited state is $4.4 \times 10^{-19} \mathrm{~J}$, what is the wavelength of the photon required to produce this transition?
$$
\left(4.5 \times 10^{-7} \mathrm{~m}\right)
$$
[Hint: $\left.\lambda=\frac{h c}{\Delta E}\right]$ | 1 |
33. A wave disturbance in a medium is described by $y(x, t)=0.02 \cos \left(50 \pi t+\frac{\pi}{2}\right) \cos (10 \pi x)$, where $x$ and $y$ are in metre and $t$ is in second.
(a) A node occurs at $x=0.15 \mathrm{~m}$
(b) An antinode occurs at $x=0.3 \mathrm{~m}$
(c) The speed of wave is $5 \mathrm{~ms}^{-1}$
(d... | 1 |
Which desert is in the northwestern part of Rajasthan? | 0 |
What are the advantages of a VAT/GST over a Sales Tax? | 0 |
How should my iPhone settings be to use Tor? | 0 |
Does a normal car windshield block UV rays? | 0 |
Example 9.25 Let $A, B, C$ be three events. If the probability of occurring exactly one event out of $A$ and $B$ is $1-a$, out of $B$ and $C$ is $1-2 a$, out of $C$ and $A$ is $1-a$ and that of occurring three events simultaneously is $a^{2}$, then prove that probability that at least one out of $A, B, C$ will occur gr... | 1 |
5. The solubility product of $\mathrm{Cr}(\mathrm{OH})_{3}$ at $298 \mathrm{~K}$ is $6.0 \times 10^{-31}$. The concentration of hydroxide ions in a saturated solution of $\mathrm{Cr}(\mathrm{OH})_{3}$ will be:
(a) $\left(2.22 \times 10^{-31}\right)^{1 / 4}$
(b) $\left(18 \times 10^{-31}\right)^{1 / 4}$
(c) $\left(18... | 1 |
34. The precipitate of
$$
\mathrm{CaF}_{2}\left(K_{s p}=1.7 \times 10^{-10}\right)
$$
is obtained when equal volumes of the following are mixed
(a) $10^{-4} \mathrm{M} \mathrm{Ca}^{2+}+10^{-4} \mathrm{M} \mathrm{F}^{-}$
(b) $10^{-2} \mathrm{M} \mathrm{Ca}^{2+}+10^{-3} \mathrm{M} \mathrm{F}$
(c) $10^{-5} \mathrm{M}... | 1 |
What should you do if you want to lose a lot of weight? | 0 |
Ex. 18. A 100-watt, 110-volt incandescent lamp is connected in series with an electrolytic cell containing cadmium sulphate solution. What weight of cadmium will be deposited by the current flowing for 10 hours?
(IIT 1987) | 1 |
I want to learn to code. Which programming language should I start with? | 0 |
13. If $S_{n}=\cot ^{-1}(3)+\cot ^{-1}(7)+\cot ^{-1}(13)+\cot ^{-1}(21)+\cdots n$ terms, then
a. $S_{10}=\tan ^{-1} \frac{5}{6}$.
c. $S_{6}=\sin ^{-1} \frac{4}{5}$
d. $S_{20}=\cot ^{-1} 1.1$
h. $S_{\infty}=\frac{\pi}{4}$ | 1 |
What is best place for sex in world? | 0 |
Who would win in a war between Australia and Indonesia? | 0 |
Which religion is widely practiced in Southeast Asia? | 0 |
What are the main types of reading materials used in education? | 0 |
1. Find resistance of the annular disc between centre and periphery of the disc. The inner and outer radius of the disc is $a$ and $b$ respectively. The thickness of the disc is $t$ and conductivity of its material is $\sigma$.
(a) $R=\frac{1}{2 \pi \sigma t} \ln \left(\frac{b}{a}\right)$
(b) $R=\frac{1}{2 \pi \sigma... | 1 |
1. A magnetic field can be produced by
(a) a moving charge
(b) a changing electric field
(c) none of them
(d) both of them. | 1 |
12. $\lim _{x \rightarrow-\infty} f(x)$ equals
a. $\ln a_{n}$
c. $a_{1}$
d. $a_{n}$
h. $e^{a_{1}}$ | 1 |
11. A light ray travelling in glass medium is incident on glass-air interface at an angle of incidence $\theta$. The reflected $(R)$ and transmitted $(T)$ intensities, both as function of $\theta$, are plotted. The correct sketch is
(a)
$ is equal to
(2019 Main, 12 April II)
(a) 0
(b) $\alpha \beta$
(c) $\alpha \gamma$
(d) $\beta \gamma$ | 1 |
What would be a good script for a movie that could scientifically motivate an entire generation to explore ecology and stewardship for the Earth in new ways? | 0 |
9. Prove that $(\vec{a} \cdot \hat{i})(\vec{a} \times \hat{i})+(\vec{a} \cdot \hat{j})(\vec{a} \times \vec{j})+(\vec{a} \cdot \hat{k})(\vec{a} \times \hat{k})=\overrightarrow{0}$. | 1 |
5. How many structural isomers does $\mathrm{C}_{4} \mathrm{H}_{8}$ have ?
(A) 3
(B) 4
(C) 5
(D) 6 | 1 |
6.48. Calculate the separation between the particles of a system in the ground state, the corresponding binding energy, and the wavelength of the first line of the Lyman series, if such a system is
(a) a mesonic hydrogen atom whose nucleus is a proton (in a mesonic atom an electron is replaced by a meson whose charge ... | 1 |
What were the reactions to the idea that function should come before other concerns? | 0 |
Where are the regulations from executive branch agencies found? | 0 |
How do you manage to deal with stress? | 0 |
What are the cons of sweatshops? | 0 |
18. A black body is at a temperature of $2880 \mathrm{~K}$. The energy of radiation emitted by this body with wavelength between $499 \mathrm{~nm}$ and $500 \mathrm{~nm}$ is $U_{1}$, between $999 \mathrm{~nm}$ and $1000 \mathrm{~nm}$ is $U_{2}$ and between $1499 \mathrm{~nm}$ and $1500 \mathrm{~nm}$ is $U_{3}$. The Wie... | 1 |
Will smoking cigarettes make me lose weight? Why or why not? | 0 |
12. Normality of $1 \% \mathrm{H}_{2} \mathrm{SO}_{4}$ solution is nearly
(a) 2.5
(b) $0 \cdot 1$
(c) 0.2
(d) 1 | 1 |
6. The sum of all two digit positive numbers which when divided by 7 yield 2 or 5 as remainder is
(2019 Main, 10 Jan I)
(a) 1256
(b) 1465
(c) 1356
(d) 1365 | 1 |
15. If dichloromethane $(\mathrm{DCM})$ and water $\left(\mathrm{H}_{2} \mathrm{O}\right)$ are used for differential extraction, which one of the following state-ments is correct?
(a) DCM and $\mathrm{H}_{2} \mathrm{O}$ would stay as lower and upper layer respectively in the S.F.
(b) DCM and $\mathrm{H}_{2} \mathrm... | 1 |
What is the equation for speed of light? | 0 |
When did the universe begin? | 0 |
How do I download music to my iPad? | 0 |
Example 2.41. Evaluate $\lim _{x \rightarrow a} \frac{\log x-\log a}{x-a}$. | 1 |
6. A source is approaching towards an observer with constant speed along the line joining them. After crossing the observer, source recedes from observer with same speed. Let $f$ is apparent frequency heard by observer. Then,
(a) $f$ will keep on increasing during approaching
(b) $f$ will keep on decreasing during re... | 1 |
7. What would be the best name for the following compound?
(A) 3-methylhydroxybenzene
(B) 3-methylcyclohexa-13,5-trien-1-ol
(C) 3-methylphenol
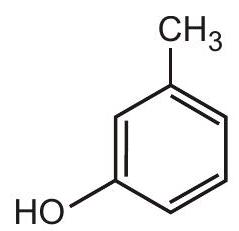
(D) 2-hydroxytoluene | 1 |
7. An unknown chlorohydrocarbon has $3.55 \%$ of chlorine. If each molecule of the hydrocarbon has one chlorine atom only, chlorine atoms present in $1 \mathrm{~g}$ of chlorohydrocarbon are:
(Atomic wt. of $\mathrm{Cl}=35.5 \mathrm{u}$; Avogadro constant $=6.023 \times 10^{23} \mathrm{~mol}^{-1}$
(a) $6.023 \times 1... | 1 |
Is Heath Ledger's death Christopher Nolan's Fault? | 0 |
10. A tuning fork of frequency $256 \mathrm{~Hz}$ is moving towards a wall with a velocity of $5 \mathrm{~m} / \mathrm{s}$. If the speed of sound is $330 \mathrm{~m} / \mathrm{s}$, then the number of beats heard per second by a stationary observer lying between tuning fork and the wall is
(a) 2
(b) 4
(c) zero
(d) 8 | 1 |
23. A $0.004 \mathrm{M}$ solution of $\mathrm{Na}_{2} \mathrm{SO}_{4}$ is isotonic with $0.010 \mathrm{M}$ solution of glucose at same temperature. The percentage dissociation of $\mathrm{Na}_{2} \mathrm{SO}_{4}$ is
(a) $25 \%$
(b) $50 \%$
(c) $75 \%$
(d) $85 \%$ | 1 |
What is India's role in SAARC? | 0 |
1. ' $\mathrm{X}$ ' melts at low temperature and is a bad conductor of electricity in both liquid and solid state. $\mathrm{X}$ is:
(a) Zinc sulphide
(b) Mercury
(c) Silicon carbide
(d) Carbon tetrachloride | 1 |
10. If $f:[0, \infty) \rightarrow[0, \infty)$ and $f(x)=\frac{x}{1+x}$, then $f$ is
(2003, 2M)
(a) one-one and onto
(b) one-one but not onto
(c) onto but not one-one
(d) neither one-one nor onto | 1 |
How much control can one have over one's emotions? | 0 |
What workout attire would guys wear in the summer like it's the year 1990? | 0 |
Rest in peace means what? | 0 |
How are masculinity and femininity more appropriately measured? | 0 |
When was the status of Dominions as equal autonomous communities given legal force? | 0 |
12. A thin convex lens made from crown glass $(\mu=3 / 2)$ has focal length $f$. When it is measured in two different liquids having refractive indices $4 / 3$ and $5 / 3$. It has the focal lengths $f_{1}$ and $f_{2}$, respectively. The correct relation between the focal length is
(a) $f_{1}=f_{2}<f$
(b) $f_{1}>f$ ... | 1 |
What area in Hyderabad contains the Hyderabad Airport? | 0 |
7. Which of the following statements is correct ?
[Main Jan. 07, 2020 (II)]
(a) Gluconic acid can form cyclic (acetal/hemiacetal) structure
(b) Gluconic acid is a dicarboxylic acid
(c) Gluconic acid is a partial oxidation product of glucose
(d) Gluconic acid is obtained by oxidation of glucose with $\mathrm{HNO}_{... | 1 |
5. The potential of hydrogen electrode is -118 mV . The concentration of $\mathrm{H}^{+}$in the solution is
(a) 0.01 M
(b) 2 M
(c) $10^{-4} \mathrm{M}$
(d) 1 M | 1 |
What is the best workout? | 0 |
11. Number of ways of selecting three integers from $\{1,2,3, \ldots, n\}$ if their sum is divisible by 3 is
a. $3\left({ }^{(n / 3} C_{3}\right)+(n / 3)^{3}$ if $n=3 k, k \in N$
b. $2\left({ }^{(n-1) / 3} C_{3}\right)+\left({ }^{(n+2) / 3} C_{3}\right)+((n-1) / 3)^{2}(n+2)$, if $n=3 k+1$, $k \in N$
c. $2\left({ }^{(n-... | 1 |
29. Which of the following is an intensive property?
(a) Volume
(b) Internal energy
(c) Entropy
(d) Mass/volume | 1 |
Why egg becomes hard when we boil it? | 0 |
What would the world be like if humans were aquatic? | 0 |
Replacing 500 and 1000 notes- how will this move reduce black money? | 0 |
(iv) $\left(\mathrm{CH}_{3}\right)_{3} \mathrm{CNH}_{2}$ | 1 |
What is the best treatment for hair loss? | 0 |
How much would it cost to replace my broken screen for a Motorola Moto G 1st gen? | 0 |
In what century did humanistic phycology rise in prominence? | 0 |
2.19 A solution containing 30 g of non-volatile solute exactly in 90 g of water has a vapour pressure of 2.8 kPa at 298 K . Further, 18 g of water is then added to the solution and the new vapour pressure becomes 2.9 kPa at 298 K . Calculate: (i) molar mass of the solute (ii) vapour pressure of water at 298 K . | 1 |
6. A rigid container of negligible heat capacity contains one mole of an ideal gas. The temperature of the gas increases by $1^{\circ} \mathrm{C}$ if $3.0 \mathrm{cal}$ of heat is added to it. The gas may be
(a) helium
(b) argon
(c) oxygen
(d) carbon dioxide. | 1 |
3. The coordinates of $A$ and $B$ are
(a) $(3,0)$ and $(0,2)$
(b) $-\frac{8}{5}, \frac{2 \sqrt{161}}{15}$ and $-\frac{9}{5}, \frac{8}{5}$
(c) $-\frac{8}{5}, \frac{2 \sqrt{161}}{15}$ and $(0,2)$
(d) $(3,0)$ and $-\frac{9}{5}, \frac{8}{5}$ | 1 |
23. Compute total circuit resistance and battery current as shown in figure.
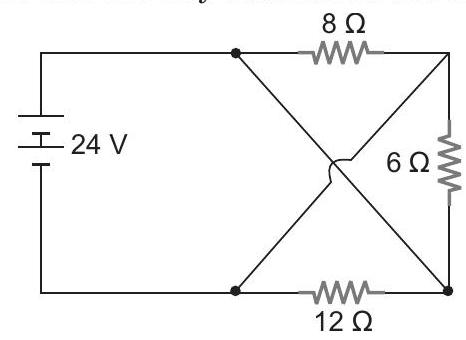 | 1 |
9. The function $f(x)=\sum_{K=1}^{5}(x-K)^{2}$ assumes the minimum value of $x$ given by
a. 5
b $\frac{5}{2}$
c. 3
d. 2 | 1 |
Why is make-up sex so good? | 0 |
18. Read the following Statement-1 (Assertion) and Statement -2 (Reason) and answer as per the options given below :
Statement-1 : Bromobenzene upon reaction with $\mathrm{Br}_{2} / \mathrm{Fe}$ gives 1,4dibromobenzene as the major product.
Statement-2 : In bromobenzene, the inductive effect of the bromo group is mor... | 1 |
Why can't India make indigenous fighter jets and weapons? | 0 |
5.11 A student forgot to add the reaction mixture to the round bottomed flask at $27^{\circ} \mathrm{C}$ but instead he/she placed the flask on the flame. After a lapse of time, he realized his mistake, and using a pyrometer he found the temperature of the flask was 477 ${ }^{\circ} \mathrm{C}$. What fraction of air wo... | 1 |
Is the idea formed by Greg limited to a specific time period? | 0 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.