Question stringlengths 2 3k | label int64 0 1 |
|---|---|
4. The standard Gibbs energy for the given cell reaction in $\mathrm{kJ} \mathrm{mol}^{-1}$ at $298 \mathrm{~K}$ is:
$\mathrm{Zn}(\mathrm{s})+\mathrm{Cu}^{2+}(\mathrm{aq}) \rightarrow \mathrm{Zn}^{2+}(\mathrm{aq})+\mathrm{Cu}(\mathrm{s})$,
[Main April 9, 2019 (I)]
$\mathrm{E}^{\circ}=2 \mathrm{~V}$ at $298 \mathrm{~... | 1 |
How can I do a global change in MS Excel? | 0 |
Which is your favourite TV series and why is that so? | 0 |
17. Let $\vec{a}$ and $\vec{b}$ be two unit vectors and $\theta$ is the angle between them. Then $\vec{a}+\vec{b}$ is a unit vector if
(A) $\theta=\frac{\pi}{4}$
(B) $\theta=\frac{\pi}{3}$
(C) $\theta=\frac{\pi}{2}$
(D) $\theta=\frac{2 \pi}{3}$ | 1 |
What are some tips on making it through the job interview process at Charter Communications? | 0 |
What is a firewall? | 0 |
Why are there so many pointless questions on Quora? | 0 |
17. A prism can have a maximum refracting angle of $\left(\theta_{C}=\right.$ critical angle for the material of prism
(a) $60^{\circ}$
(b) $\theta_{C}$
(c) $2 \theta_{C}$
(d) slightly less than $180^{\circ}$ | 1 |
Which is the worlds best electric shaver? | 0 |
11. Hydrogen peroxide in its reaction with $\mathrm{KIO}_{4}$ and $\mathrm{NH}_{2} \mathrm{OH}$ respectively, is acting as a
(a) Reducing agent, oxidising agent
(b) Reducing agent, reducing agent
(c) Oxidising agent, oxidising agent
(d) Oxidising agent, reducing agent | 1 |
How do I start freelancing as web developer? | 0 |
Which mobile I should buy under 15k? | 0 |
8. Three identical rods are hinged at point $A$ as shown. The angle made by rod $A B$ with vertical is
(a) $\tan ^{-1}\left(\frac{1}{\sqrt{3}}\right)$
(b) $\tan ^{-1}\left(\frac{3}{4}\right)$
(c) $\tan ^{-1}(1)$
(d) $\tan ^{-1}\left(\frac{4}{3}\right)$
 y$
b. $n(n+1) y$
c. $n y$
d. $n^{2} y$ | 1 |
What did the Scientific Revolution cause? | 0 |
52. The rate of reaction doubles when its temperature changes from 300 K to 310 K . Activation energy of such a reaction will be $(R=8.314 \mathrm{~J} / \mathrm{mol} \mathrm{K}$ and $\log 2=0.301)$.
(a) $53.6 \mathrm{~kJ} \mathrm{~mol}^{-1}$
(b) $48.6 \mathrm{~kJ} \mathrm{~mol}^{-1}$
(c) $58.5 \mathrm{~kJ} \mathrm{~mol... | 1 |
24. Given $z=(1+i \sqrt{3})^{100}$, then $[\operatorname{RE}(z) / \operatorname{IM}(z)]$ equals
a. $2^{100}$
b. $2^{50}$
c. $\frac{1}{\sqrt{3}}$
d. $\sqrt{3}$ | 1 |
What is sexual orientation defined as? | 0 |
What dynamic needs were the reason for building to be done? | 0 |
What is it like to be a male escort? | 0 |
How can I be, what I can describe? | 0 |
Why does Colorado have so much molybdenum? | 0 |
Ex. 51. 20.27 g of benzene containing 0.2965 g of benzoic acid (mol. $w \mathrm{t} .=122$ ) freezes at $0.317^{\circ}$ below the freezing point of pure benzene. If benzoic acid exists as dimer in benzene, find its degree of association. $K_{f}$ for benzene is $5 \cdot 12^{\circ} \mathrm{C} \cdot \mathrm{m}^{-1}$. | 1 |
How can I learn how to be disciplined in school? | 0 |
What about the placement of IEM Kolkata? | 0 |
(a) the potential difference across the resistor, | 1 |
Can World War 3 ever take place? | 0 |
How can I calculate the surface area of a trapezoid? | 0 |
How do I get contact details of CEOs? | 0 |
4. Which one of the following equations does not correctly represent the first law of thermodynamics for the given processes involving an ideal gas? (Assume non-expansion work is zero)
(a) Cyclic process : $q=-w$
(b) Adiabatic process : $\Delta U=-w$
(c) Isochoric process: $\Delta U=q$
(d) Isothermal process: $q=-w... | 1 |
1. Which of these will produce the highest yield in Friedel Crafts reaction?
(a) 
(b) 
(... | 1 |
7. In the determination of Young's modulus $[Y=\frac{4 M L g}{\pi l d^{2}}]$ by using Searle's method, a wire of length $L=2 \mathrm{~m}$ and diameter $d=0.5 \mathrm{~mm}$ is used. For a load $M=2.5 \mathrm{~kg}$, an extension $l=0.25 \mathrm{~mm}$ in the length of the wire is observed. Quantities $d$ and $l$ are measu... | 1 |
How much time it will take for the beginners to learn the basic of guitars? | 0 |
What are some facts about MVSR Engineering college which others don't know? | 0 |
Ex. 17. Calculate the pH of a $10^{-5} \mathrm{M} \mathrm{HCl}$ solution if 1 mL of it is diluted to $1000 \mathrm{~mL} . K_{\mathrm{w}}=1 \times 10^{-14}$. | 1 |
What's it like working for Amazon? | 0 |
39. When light of wavelength $\lambda$ is incident on an equilateral prism, kept on its minimum deviation position, it is found that the angle of deviation equals the angle of the prism itself. The refractive index of the material of the prism for the wavelength $\lambda$ is
(a) $\sqrt{3}$
(b) $\sqrt{3 / 2}$
(c) 2
(d) ... | 1 |
Why do the isomers have different melting points? | 0 |
What kind of music is dholak ke geet? | 0 |
What are some jobs that will be in high demand in the next 10 years or so in the SAARC region? | 0 |
When is Xiaomi going to release Lollipop version for Redmi 1S? | 0 |
27. The period of function $2^{\{x\}}+\sin \pi x+3^{\{x / 2\}}+\cos 2 \pi x$ (where $\{x\}$ denotes the fractional part of $x$ ) is
a. 2
b. 1
c. 3
d. None of these | 1 |
What's the best way to deal with a boss who pushes you too hard? | 0 |
If the universe is expanding faster than light then how do we see everything? | 0 |
Do sports teams pay for families to travel with the family member on the team? | 0 |
How can a girl know if a boy is in a love with her? | 0 |
What does it feel like to be stalked by women? | 0 |
3. Minimum number of times a fair coin must be tossed so that the probability of getting atleast one head is more than $99 \%$ is
(2019 Main 10 April II)
(a) 8
(b) 6
(c) 7
(d) 5 | 1 |
What term can mean a non-fiction piece of a collection of short pieces? | 0 |
I'm going to enter high school on 1st August. I'm on my holiday right now. What should I learn about Chemistry for High School? | 0 |
Which motor areas of the brain control breathing and swallowing? | 0 |
What was a concern of the Kinsey scale? | 0 |
How long can someone inexperienced survive on a desert island? | 0 |
1. Which of the following is most acidic in nature.
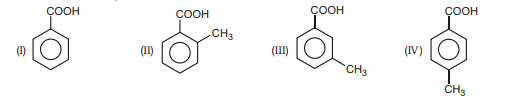
(A) I
(B) II
(C) III
(D) IV | 1 |
6. The value of $\frac{x+y}{1-x y}$ is
a. $\frac{2 a b}{a^{2}-c^{2}}$
b.frac{2 a c}{a^{2}-c^{2}}$
c. $\frac{c^{2}-b^{2}}{a^{2}+b^{2}}$
d none of these | 1 |
20. $5.6 \mathrm{~L}$ of helium gas at STP is adiabatically compressed to $0.7 \mathrm{~L}$. Taking the initial temperature to be $T_{1}$, the work done in the process is
(a) $\frac{9}{8} R T_{1}$
(b) $\frac{3}{2} R T_{1}$
(c) $\frac{15}{8} R T_{1}$
(d) $\frac{9}{2} R T_{1}$ | 1 |
16. In plotting stress versus strain curves for two materials $P$ and $Q$, a student by mistake puts strain on the $Y$-axis and stress on the $X$-axis as shown in the figure. Then the correct statements is/are
(2015 Adv.)
 Both bulbs will glow alternatively
(b) Both bulbs will glow with same brightness provided
$$
f=\frac{1}{2 \pi} \sqrt{(1 / ... | 1 |
How can I install Apache Cassandra on Windows Operating System? | 0 |
3. A particle of mass $20 \mathrm{~g}$ is released with an initial velocity 5 $\mathrm{m} / \mathrm{s}$ along the curve from the point $A$, as shown in the figure. The point $A$ is at height $h$ from point $B$. The particle slides along the frictionless surface. When the particle reaches point $B$, its angular momentum... | 1 |
What's the difference between "had been" and "were"? | 0 |
How many ships did Greece's navy have in the late 1970s? | 0 |
15. Figure shows an inverted cone filled completely with a liquid of mass $m$. The axis of the cone is vertical. The force exerted by the lateral surface of the cone on the liquid is :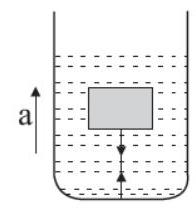
(a) 3... | 1 |
I am a mechanical engineering student and have decided to go for the IAS exams. What is the best subject to learn excluding maths and physics? | 0 |
What is your favorite sport and why do you like it so much? | 0 |
111. The point of intersection of the tangents of the parabola $y^{2}=4 x$, drawn at end points of the chord $x+y=2$ lies on
a. $x-2 y=0$
b. $x+2 y=0$
c. $y-x=0$
d. $x+y=0$ | 1 |
18. The crystalline form of borax has
(a) tetranuclear $\left[\mathrm{B}_{4} \mathrm{O}_{5}(\mathrm{OH})_{4}\right]^{2-}$ unit
(b) all boron atoms in the same plane
(c) equal number of $s p^{2}$ and $s p^{3}$ hybridized boron atoms
(d) one terminal hydroxide per boron atom | 1 |
12. Let the pairs $\vec{a}, \vec{b}$ and $\vec{c}, \vec{d}$ each determine a plane. Then the pianes are parallel if
a. $(\vec{a} \times \vec{c}) \times(\vec{b} \times \vec{d})=\overrightarrow{0}$
b. $(\vec{a} \times \vec{c}) \cdot(\vec{b} \times \vec{d})=\overrightarrow{0}$
c. $(\vec{a} \times \vec{b}) \times(\vec{c} \... | 1 |
11. The root mean square velocity of an ideal gas
at constant pressure varies with density (d) as (a) $d^2$
(b) $d$
(c) $\sqrt{d}$
(d) $1 / \sqrt{d}$ | 1 |
30. The kinetic energy of any gas molecule at $0^{\circ} \mathrm{C}$ is
(a) $5.66 \times 10^{-21} \mathrm{~J}$
(b) 3408 J
(c) 2 cal
(d) 0 | 1 |
15.6 A bat emits ultrasonic sound of frequency 1000 kHz in air. If the sound meets a water surface, what is the wavelength of (a) the reflected sound, (b) the transmitted sound? Speed of sound in air is $340 \mathrm{~m} \mathrm{~s}^{-1}$ and in water $1486 \mathrm{~m} \mathrm{~s}^{-1}$. | 1 |
1. Find the point on the curve $y^{2}=a x$ the tangent at which makes an angle of $45^{\circ}$ with $x$-axis. | 1 |
8. $E_{\text {cell }}^{0}$ for $\mathrm{Zn}(\mathrm{s})+\mathrm{Pb}^{2+}(1 \mathrm{M}) \rightarrow \mathrm{Zn}^{2+}(1 \mathrm{M})+\mathrm{Pb}(\mathrm{s})$ is +0.66 volt. $E_{\text {cell }}$ for the reaction
$$
\begin{equation*}
\mathrm{Zn}(\mathrm{~s})+\mathrm{Pb}^{2+}(0 \cdot 1 \mathrm{M}) \rightarrow \mathrm{Zn}^{2+... | 1 |
- Example 1.8 Find the area of the quadrilateral $A B C D$ whose vertices are respectively $A(1,1), B(7,-3), C(12,2)$, and $D(7,21)$. | 1 |
How do I connect my Samsung J7 with a Sony Bravia R51C? | 0 |
13. If $f(x)= \begin{cases}-x-\frac{\pi}{2}, & x \leq-\frac{\pi}{2} \\ -\cos x, & -\frac{\pi}{2}<x \leq 0 \text {, then } \\ x-1, & 0<x \leq 1 \\ \ln x, & x>1\end{cases}$
a. $f(x)$ is continuous at $x=-\pi / 2$
b. $f(x)$ is not differentiable at $x=0$
c. $f(x)$ is differentiable at $x=1$
d. $f(x)$ is differentiable at ... | 1 |
How do you solve the equation u÷9-4=-4? | 0 |
What city is the official capital of the National Capital Territory of Delhi? | 0 |
11. Two metal spheres of capacitances $C_{1}$ and $C_{2}$ carry some charges. They are put in contact and then separated. The final charges $Q_{1}$ and $Q_{2}$ on them will satisfy
(a) $\frac{Q_{1}}{Q_{2}}<\frac{C_{1}}{C_{2}}$
(b) $\frac{Q_{1}}{Q_{2}}=\frac{C_{1}}{C_{2}}$
(c) $\frac{Q_{1}}{Q_{2}}>\frac{C_{1}}{C_{2}}$
(... | 1 |
Was Maxwell the first to consider electricity in terms of oscillations? | 0 |
What is the most evil thing on deep web? | 0 |
A simple model is also referred to as what? | 0 |
11. A uniform but time varying magnetic field exists in a cylindrical region as shown in the figure. The direction of magnetic field is into the plane of the paper and its magnitude is decreasing at a constant rate of $2 \times 10^{-3} \mathrm{~T} / \mathrm{s}$. A particle of charge $1 \mu \mathrm{C}$ is moved slowly a... | 1 |
40. The points $(-a,-b),(0,0),(a, b)$ and $\left(a^{2}, a^{3}\right)$ are
(a) collinear
(b) vertices of a rectangle
(c) vertices of a parallelogram
(d) None of the above | 1 |
25. A block $A$ of mass $2 \mathrm{~kg}$ rests on another block $B$ of mass $8 \mathrm{~kg}$ which rests on a horizontal floor. The coefficient of friction between $A$ and $B$ is 0.2 while that between $B$ and floor is 0.5 . When a horizontal force $F$ of $25 \mathrm{~N}$ is applied on the block $B$, the force of frict... | 1 |
How many days does it take before a woman becomes pregnant after sex? | 0 |
What do people speak in Bulgaria? | 0 |
29. One end of a taut string of length $3 \mathrm{~m}$ along the $X$-axis is fixed at $x=0$. The speed of the waves in the string is $100 \mathrm{~ms}^{-1}$. The other end of the string is vibrating in the $y$-direction so that stationary waves are set up in the string. The possible waveform(s) of these stationary wave... | 1 |
Is there a way to see deleted Instagram photos? | 0 |
12. Four capacitors are connected in series with a battery of emf $10 \mathrm{~V}$ as shown in the figure. The point $P$ is earthed. The potential of point $A$ is equal in magnitude to potential of point $B$ but opposite in sign if
$ from the given position in such a way that it compresses a spring and released. The ti... | 1 |
How accurate is carbon dating? | 0 |
35. Resonance structures of a molecule should have :
(a) identical arrangement of atoms
(b) nearly the same energy content
(c) the same number of paired electrons
(3) 10 Subjective Problems | 1 |
7. Which of the given compounds will have minimum enol content?
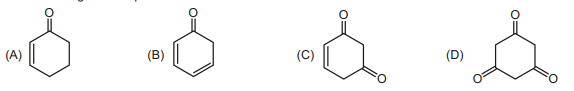 | 1 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.