Question stringlengths 2 3k | label int64 0 1 |
|---|---|
At serfdom's greatest extent, what percentage of Western Europeans were serfs? | 0 |
2. $\int \frac{x^{2}+1}{x\left(x^{2}-1\right)} d x$ | 1 |
30. n-Propyl bromide on treatment with ethanolic potassium hydroxide produces
(a) Propane
(b) Propene
(c) Propyne
(d) Propanol | 1 |
What was the name of Pugin's book? | 0 |
When was Nazi Germany in a one-party system? | 0 |
35. The compound whose $0.1 \mathrm{M}$ solution is basic is :
(a) ammonium acetate
(b) ammonium chloride
(c) ammonium sulphate
(d) sodium acetate | 1 |
What author would Jerome rather read than the Bible? | 0 |
13. Which of the following is not the general solution of $2^{\cos 2 x}+1=3.2^{-\sin ^{2} x}$ ?
a. $n \pi, n \in Z$
$\mathbf{b}\left(n+\frac{1}{2}\right) \pi, n \in Z$
c. $\left(n-\frac{1}{2}\right) \pi, n \in Z$
d none of these | 1 |
What is computer programming? | 0 |
What are the strangest phobias some people have and what causes them? | 0 |
36. ${ }_{90}^{234} \mathrm{Th}$ disintegrates to give ${ }_{82}^{206} \mathrm{~Pb}$ as the final product. How many alpha and beta particles are emitted during this process? | 1 |
21. For a $d$-electron, the orbital angular momentum is
(a) $\sqrt{6}(h / 2 \pi)$
(b) $\sqrt{2}(h / 2 \pi)$
(c) $(h / 2 \pi)$
(d) $2(h / 2 \pi)$ | 1 |
What is the second empires? | 0 |
68 If first-order reflection occurs in a crystal at Bragg angle 3.4${ }^{\circ}$, at what Bragg angle does second-order reflection occur from the same family of reflecting planes? | 1 |
How do I get my questions answered? | 0 |
Example 7.18 When HCl reacts with finely powdered iron, it forms ferrous chloride and not ferric chloride. Why? | 1 |
What kind of methods use Tarantino in his scripts? | 0 |
Question 37:
Prove $\int_0^{\frac{\pi}{2}} \sin ^3 x d x=\frac{2}{3}$ | 1 |
Who have a better orgasm: men or women? | 0 |
How could you hack someone’s mobile phone? | 0 |
What are qualifications needed for teaching in school? | 0 |
9. If the rate constant $k$ of a reaction is $1.6 \times 10^{-3}(\mathrm{~mol} / \mathrm{L})\left(\mathrm{min}^{-1}\right)$, the order of the reaction is
(a) 0
(b) 1
(c) 2
(d) cannot be known | 1 |
What are the safety precautions on handling shotguns proposed by the NRA in Alaska? | 0 |
What did SASO find wrong with the KInsey scale and Klein Sexual Orientation Grid? | 0 |
8. As per Hardy-Schulze formulation, the flocculation values of the following for ferric hydroxide sol are in the order:
(a) $\mathrm{K}_{3}\left[\mathrm{Fe}(\mathrm{CN})_{6}\right]<\mathrm{K}_{2} \mathrm{CrO}_{4}<\mathrm{KBr}=\mathrm{KNO}_{3}=\mathrm{AlCl}_{3}$
(b) $\mathrm{K}_{3}\left[\mathrm{Fe}(\mathrm{CN})_{6}... | 1 |
How much did SAT exams cost as of 2002 in Nigeria? | 0 |
30. A block tied between two identical springs is in equilibrium. If upper spring is cut, then the acceleration of the block just after cut is $5 \mathrm{~ms}^{-2}$. Now if instead of upper string lower spring is cut, then the acceleration of the block just after the cut will be (Take $g=10 \mathrm{~m} / \mathrm{s}^{2}... | 1 |
5. Two equal resistances when connected in series to a battery consume electric power of $60 \mathrm{~W}$. If these resistances are now connected in parallel combination to the same battery, the electric power consumed will be (2019 Main, 11 Jan I)
(a) $60 \mathrm{~W}$
(b) $30 \mathrm{~W}$
(c) $240 \mathrm{~W}$
(d) $12... | 1 |
34. In the NaCl crystal, which of the following facts is not true?
(a) $\mathrm{Na}^{+}$ions form f.c.c. lattice
(b) $\mathrm{Cl}^{-}$ions form f.c.c. lattice
(c) $\mathrm{Na}^{+} \mathrm{Cl}^{-}$units form f.c.c. structure
(d) CN of each $\mathrm{Na}^{+}$and $\mathrm{Cl}^{-}$is 6 | 1 |
What does Howard Stern think about Donald Trump's 2016 campaign? | 0 |
Where can I download an ftp client with code editor? | 0 |
Should I kiss my girlfriend? | 0 |
15. A piston filled with $0.04 \mathrm{~mol}$ of an ideal gas expands reversibly from $50.0 \mathrm{~mL}$ to $375 \mathrm{~mL}$ at a constant temperature of $37.0^{\circ} \mathrm{C}$. As it does so, it absorbs $208 \mathrm{~J}$ of heat. The values of $\mathrm{q}$ and $\mathrm{w}$ for the process will be:
$(\mathrm{R}=... | 1 |
2. Preparation of Bakelite proceeds via reactions:
[Main Jan. 08, 2020 (II)]
(a) Electrophilic addition and dehydration
(b) Condensation and elimination
(c) Electrophilic substitution and dehydration
(d) Nucleophilic addition and dehydration | 1 |
Lev Grossman: Who would win in a fight between Gandalf and Dumbledore? | 0 |
1. Correct order for the heat of combustion of the following is :
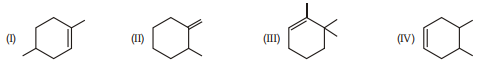
(A) I $>$ II $>$ IV $>$ III
(B) IV $>$ I $>$ II $>$ III
(C) III $>$ I $>$ IV $>$ II
(D) III $>$ II $>$ IV $>$ I | 1 |
Based on the recent threats and issues regarding the US, Russia and China, is World War 3 near? | 0 |
13. A gas can be taken from $A$ to $B$ via two different processes $A C B$ and $A D B$
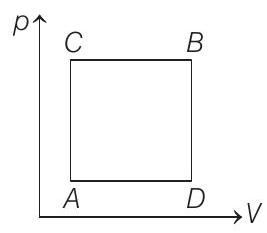
When path $A C B$ is used $60 \mathrm{~J}$ of heat flows into the system and $30 \mathrm{~J}$ of wo... | 1 |
Is NAFTA a good or bad thing for the US economy? | 0 |
What is wrong with me? Sometimes I wake up with extreme positivity and ambition and some other days I'm the most pessimistic depressed person. | 0 |
9. If $A=\left[a_{i j}\right]_{4 \times 4}$, such that $a_{i j}=\left\{\begin{array}{ll}2, & \text { when } i=j \\ 0, & \text { when } i \neq j\end{array}\right.$, then $\left\{\frac{\operatorname{det}(\operatorname{adj}(\operatorname{adj} A))}{7}\right\}$ is (where $\{\cdot\}$ represents fractional part function)
a. $... | 1 |
How do I stop thinking about thinking? | 0 |
Example 1.21 Find the domain of the functions.
$$
f(x)=\frac{1}{1+2 \sin x}
$$ | 1 |
Instagram made me change my password, but I'm not logged into my Facebook and I don't know my email for the account. What should I do? | 0 |
5. Let $\alpha$ and $\beta$ be the roots of the quadratic equation $x^{2} \sin \theta-x(\sin \theta \cos \theta+1)+\cos \theta=0\left(0<\theta<45^{\circ}\right)$ and $\alpha<\beta$. Then, $\sum_{n=0}^{\infty} \alpha^{n}+\frac{(-1)^{n}}{\beta^{n}}$ is equal to
(a) $\frac{1}{1-\cos \theta}-\frac{1}{1+\sin \theta}$
(b) $\... | 1 |
21. Find the equivalent capacitance across $A$ and $B$ for the arrangement shown in figure. All the capacitors are of capacitance $C$
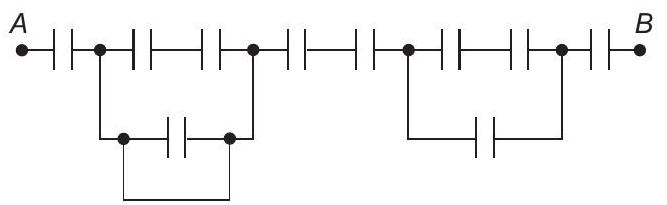
(a) $\frac{3 C}{14}$
(b) $\frac{C}{8}$
(c) $\frac{3 C}{1... | 1 |
4. An electric bulb is hanging over a table at a height of $1 \mathrm{~m}$ above it. The illuminance on the table directly below the bulb is 40 lux. The illuminance at a point on the table $1 \mathrm{~m}$ away from the first point will be about
(a) 10 lux
(b) 14 lux
(c) 20 lux
(d) 28 lux. | 1 |
What year is considered the transition between the classical period and the Middle Ages? | 0 |
2. A soap bubble, blown by a mechanical pump at the mouth of a tube, increases in volume, with time, at a constant rate. The graph that correctly depicts the time dependence of pressure inside the bubble is given by
(2019 Main, 12 Jan II)
(a)
 $O A$
(b) $O B$
(c) $O C$
(d)... | 1 |
12. If $r, s, t$ are prime numbers and $p, q$ are the positive integers such that LCM of $p, q$ is $r^{2} s^{4} t^{2}$, then the number of ordered pairs $(p, q)$ is
(2006, 3M)
(a) 252
(b) 254
(c) 225
(d) 224 | 1 |
8. $\quad$ For one mole of a van der Waal's gas when $b=0$ and $T=300 \mathrm{~K}$, the $P V$ vs, $1 / V$ plot is shown below. The value of the van der Waal's
constant $a\left(\right.$ atm. liter $\left.\mathrm{mol}^{2}\right)$ is :

$$
This equation is applicable to a
(a) system that satisfies the van der Waals equation of state
[Adv. 2020]... | 1 |
6. Statement 1: If a point $\left(x_{1}, y_{1}\right)$ lies in the shaded region $\frac{\dot{x}^{2}}{a^{2}}-\frac{y^{2}}{b^{2}}=1$, shown in the figure, then $\frac{x_{1}^{2}}{a^{2}}-\frac{y_{1}^{2}}{b^{2}}<0$.
 $15 \mathrm{~cm}$ diverging
(b) $15 \mathrm{~cm}$ converging
(c) $10 \mathrm{~cm}$ diverging
(d) $10 \mathrm{~cm}$ converg... | 1 |
4. The intensity of $X$-rays from a coolidge tube is plotted against wavelength $\lambda$ as shown in the figure. The minimum wavelength found is $\lambda_{c}$ and the wavelength of the $K_{\alpha}$ line is $\lambda_{k}$. As the accelerating voltage is increased
 Decarboxylation | (a) Addition reaction |
| (ii) Ozonolysis ... | 1 |
When did the United Nations Convention against Corruption join? | 0 |
What groups is named after the trait of growth by moulting or ecdysis? | 0 |
Can cannabis oil cure cancer? | 0 |
Which is the English song with best lyrics according to you? | 0 |
When did the Somer Isles Company take over managing Bermuda? | 0 |
30. Native silver metal forms a water soluble complex with a dilute aqueous solution of $\mathrm{NaCN}$ in the presence of
(a) nitrogen
(b) oxygen
(c) carbon dioxide
(d) argon | 1 |
52. Two moles of a perfect gas undergo the following processes:
(a) a reversible isobaric expansion from (1.0 atm, 20.0L) to (1.0 atm, 40.0 $\mathrm{L})$
(b) a reversible isochoric change of state from $(1.0 \mathrm{~atm}, 40.0 \mathrm{~L})$ to $(0.5$ $\operatorname{atm}, 40.0 \mathrm{~L})$
(c) a reversible isother... | 1 |
What do religions share across their belief structures? | 0 |
24. If $f(x)$ and $g(x)$ are two positive and increasing functions, then which of the following is not always true?
a. $[f(x)]^{g(x)}$ is always increasing
h. if $[f(x)]^{(x)}$ is decreasing, then $f(x)<1$
c. if $[f(x)]^{g(x)}$ is increasing, then $f(x)>1$
d if $f(x) \triangleright 1$, then $[f(x)]^{g(x)}$ is increa... | 1 |
8. In a resonance tube experiment to determine the speed of sound in air, a pipe of diameter $5 \mathrm{~cm}$ is used. The air column in pipe resonates with a tuning fork of frequency $480 \mathrm{~Hz}$ when the minimum length of the air column is $16 \mathrm{~cm}$. Find the speed of sound in air at room temperature. | 1 |
How do I recover a forgotten Gmail password? | 0 |
What is the difference between welfare capitalism and social democracy? | 0 |
18. Consider a capacitor - charging circuit. Let $Q_{1}$ be the charge given to the capacitor in time interval of $20 \mathrm{~ms}$ and $Q_{2}$ be the charge given in the next time interval of $20 \mathrm{~ms}$. Let $10 \mu \mathrm{C}$ charge be deposited in a time interval $t_{1}$ and the next $10 \mu \mathrm{C}$ char... | 1 |
43. The value of $x$ which satisfies equation $2 \tan ^{-1} 2 x=\sin ^{-1} \frac{4 x}{1+4 x^{2}}$ is valid in the interval
a. $\left[\frac{1}{2}, \infty\right)$
$\mathbf{h}\left(-\infty,-\frac{1}{2}\right]$
c. $[-1,1]$
d $\left[-\frac{1}{2}, \frac{1}{2}\right]$ | 1 |
Can someone help me learn how to use Paytm? | 0 |
Should I study for the GRE? | 0 |
2. A plane mirror is falling vertically as shown in the figure. If $S$ is a point source of light, the rate of increase of the length $A B$ is
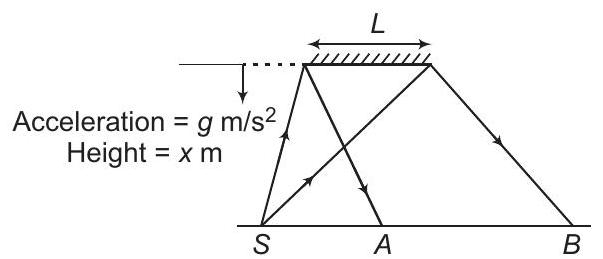
(a) directly proportional to $x$
(b) constant ... | 1 |
10. A wall has two layers $A$ and $B$ each made of different materials. Both the layers have the same thickness. The thermal conductivity of material $A$ is twice of $B$. Under thermal equilibrium the temperature difference across the layer $B$ is $36^{\circ} \mathrm{C}$. The temperature difference across layer $A$ is
... | 1 |
How do I know whether I am in love with a person or not? | 0 |
What type of paper is The Siasat Daily? | 0 |
2. (a) Find the frequencies of revolution of electrons in $n=1$ and $n=2$ Bohr orbits.
(b) What is the frequency of the photon emitted when an electron in an $n=2$ orbit drops to an $n=1$ hydrogen orbit?
(c) An electron typically spends about $10^{-8} \mathrm{~S}$ in an excited state before it drops to a lower state ... | 1 |
Who is she? | 0 |
Who has argued that emotions are discrete? | 0 |
New Delhi is located on the floodplains of what river? | 0 |
What is the best site to download anime torrents? | 0 |
56. The nitrogen oxide(s) that contain(s) $\mathrm{N}-\mathrm{N}$ bond(s) is(are) (a) $\mathrm{N}_{2} \mathrm{O}$
(b) $\mathrm{N}_{2} \mathrm{O}_{3}$
(c) $\mathrm{N}_{2} \mathrm{O}_{4}$
(d) $\mathrm{N}_{2} \mathrm{O}_{5}$ | 1 |
20. Two blocks of masses $2 \mathrm{~kg}$ and $4 \mathrm{~kg}$ are connected by a light string and kept on horizontal surface. A force of $16 \mathrm{~N}$ is acted on $4 \mathrm{~kg}$ block horizontally as shown in figure. Besides, it is given that coefficient of friction between $4 \mathrm{~kg}$ and ground is 0.3 and ... | 1 |
What two fields began to grow apart? | 0 |
What does it take to become a professional F1 driver? | 0 |
23. In Wilkinson's catalyst, the hybridisation of central metal ion and its shape are respectively:
(a) $s p^{3} d$, trigonal bipyramidal
(b) $d^{2} s p^{3}$, octahedral
(c) $d s p^{2}$, square planar
(d) $s p^{3}$, tetrahedral | 1 |
8. A string of length $1 \mathrm{~m}$ and mass $5 \mathrm{~g}$ is fixed at both ends. The tension in the string is $8.0 \mathrm{~N}$. The string is set into vibration using an external vibrator of frequency $100 \mathrm{~Hz}$. The separation between successive nodes on the string is close to
(a) $16.6 \mathrm{~cm}$
(... | 1 |
What are the most popular newspapers in the UK? | 0 |
5. Just on crossing the yield region, the material will have
(a) increased and breaking stress
(b) reduced and breaking stress
(c) constant stress
(d) None of these | 1 |
Why do software companies like Google, Facebook, LinkedIn, Microsoft etc prefer to set up there offices in Bangalore/Hyderabad over Mumbai? | 0 |
52. If $X=\left\{4^{n}-3 n-1: n \in N\right\}$ and
$Y=\{9(n-1): n \in N\}$, where $N$ is the set of natural numbers, then $X \cup Y$ is equal to
(a) $N$
(b) $Y-X$
(c) $X$
(d) $Y$ | 1 |
What do other people use to determine a persons sexual idenity? | 0 |
5. In a YDSE experiment, $d=1 \mathrm{~mm}, \lambda=6000 \AA$ and $D=1 \mathrm{~m}$. The minimum distance between two points on screen having $75 \%$ intensity of the maximum intensity will be
(a) $0.50 \mathrm{~mm}$
(b) $0.40 \mathrm{~mm}$
(c) $0.30 \mathrm{~mm}$
(d) $0.20 \mathrm{~mm}$ | 1 |
How can I get my lost Samsung j7 back? | 0 |
What improvements can SCMP news outlet make for its foreign subscribers and readers? | 0 |
What was a major concern for Western civilization during the 21 century? | 0 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.