Question stringlengths 2 3k | label int64 0 1 |
|---|---|
How many important emotional elements are present in the components processing model? | 0 |
What is the best backend for an ionic video messaging app? | 0 |
28. If $A$ is a nilpotent matrix of index 2 , then for any positive integer $n, A(I+A)^{n}$ is equal to
a. $A^{-1}$
b. $A$
c. $A^{n}$
d. $I_{n}$ | 1 |
Why is 'geometric' progression called so? | 0 |
35. When equal volumes of the following solutions are mixed, precipitation of $\mathrm{AgCl}\left(K_{\mathrm{sp}}=1.8 \times 10^{-10}\right)$ will occur only with
(a) $10^{-4} \mathrm{M}\left(\mathrm{Ag}^{+}\right)$and $10^{-4} \mathrm{M}\left(\mathrm{Cl}^{-}\right)$
(b) $10^{-5} \mathrm{M}\left(\mathrm{Ag}^{+}\right)$... | 1 |
What is the difference between Android Lollipop 5.1.1 and Marshmallow 6.0? | 0 |
27. For a cell, the terminal potential difference is $2.2 \mathrm{~V}$, when circuit is open and reduces to $1.8 \mathrm{~V}$. When cell is connected to a resistance $R=5 \Omega$, the internal resistance of cell $(r)$ is
(a) $\frac{10}{9} \Omega$
(b) $\frac{9}{10} \Omega$
(c) $\frac{11}{9} \Omega$
(d) $\frac{5}{9} ... | 1 |
- Example 3.2 In the diagram shown in figure, find the magnitude and nature of zero error.
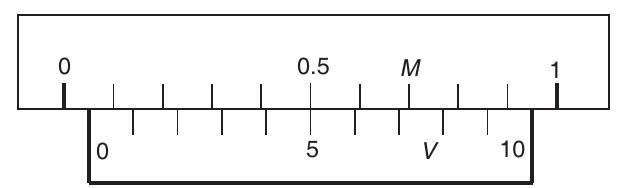
Fig. 3.4 | 1 |
What are the benefits of running every day? | 0 |
What are the best available smartphones gadgets? | 0 |
How efficient is a MacBook Pro for gaming? | 0 |
What was hot in Silicon Valley VC firms in May 2016? | 0 |
What is the difference between string s= new string(); and string s="xyz"? | 0 |
Do men really think about sex most of the time? Why? | 0 |
Golconda is a type of what kind of painting? | 0 |
We have to choose 11 players for cricket team from eight batsmen, six bowlers, four all rounders and two wicket keepers in the following conditions
1. The number of selections when almost one all rounder and one wicket keeper will play
a. ${ }^{4} C_{1} \times{ }^{14} C_{10}+{ }^{2} C_{1} \times{ }^{14} C_{10}+{ }^{4}... | 1 |
27. The slope of tanget to a curve $y=f(x)$ at $[x, f(x)]$ is $2 x+1$. If the curve passes through the point $(1,2)$, then the area bounded by the curve, the X-axis and the line $x=1$ is
(a) $\frac{3}{2}$
(b) $\frac{4}{3}$
(c) $\frac{5}{6}$
(d) $\frac{1}{12}$ | 1 |
38. A cylindrical vessel open at the top is $20 \mathrm{~cm}$ high and $10 \mathrm{~cm}$ in diameter. A circular hole of cross sectional area $1 \mathrm{~cm}^{2}$ is cut at the centre of the bottom of the vessel. Water flows from a tube above it into the vessel at the rate of $10^{2} \mathrm{~cm}^{3} / \mathrm{s}$. The... | 1 |
8. A boy of height $1.5 \mathrm{~m}$ with his eye level at $1.4 \mathrm{~m}$ stands before a plane mirror of length $0.75 \mathrm{~m}$ fixed on the wall. The height of the lower edge of the mirror above the floor is $0.8 \mathrm{~m}$. Then,
(a) the boy will see his full image
(b) the boy cannot see his hair
(c) the boy... | 1 |
Why is Donald Trump so popular? | 0 |
Which college is better - IIIT Allahabad or IIIT Hyderabad? | 0 |
Who stated "the heart has reasons of which reason does not know"? | 0 |
8. Suppose that the points $(h, k),(1,2)$ and $(-3,4)$ lie on the line $L_{1}$. If a line $L_{2}$ passing through the points $(h, k)$ and $(4,3)$ is perpendicular to $L_{1}$, then $k / h$ equals
(a) $-\frac{1}{7}$
(b) $\frac{1}{3}$
(c) 3
(d) 0 | 1 |
18. A $2 \mathrm{~kg}$ object is slid along a rough floor (coefficient of sliding friction $=0.3$ ) with initial velocity $(10 \mathrm{~m} / \mathrm{s}) \hat{\mathrm{i}}$. Describe the motion of the object relative to car assuming that the coefficient of static friction is greater than 0.5 . | 1 |
What is the longest time that you have ever gone without food, water, and going shopping? | 0 |
32. If the solubility of $\mathrm{Al}(\mathrm{OH})_3$ is $S$ moles/litre, the solubility product is
(a) $S^3$
(b) $27 S^4$
(c) $S^2$
(d) $4 S^3$ | 1 |
(a) the terminal velocity achieved by the rod, and | 1 |
6.201. Calculate the interval (in $\mathrm{eV}$ units) between neighbouring levels of free electrons in a metal at $T=0$ near the Fermi level, if the concentration of free electrons is $n=2.0 \cdot 10^{22} \mathrm{~cm}^{-3}$ and the volume of the metal is $V=1.0 \mathrm{~cm}^{3}$. | 1 |
What length of time do the 200,000 works of art in the National Museum encompass? | 0 |
6. The two monomers for the synthesis of nylon 6,6 are :
[Main Jan. 12, 2019 (II)]
(a) $\mathrm{HOOC}\left(\mathrm{CH}_{2}\right)_{4} \mathrm{COOH}, \mathrm{H}_{2} \mathrm{~N}\left(\mathrm{CH}_{2}\right)_{6} \mathrm{NH}_{2}$
(b) $\mathrm{HOOC}\left(\mathrm{CH}_{2}\right)_{6} \mathrm{COOH}, \mathrm{H}_{2} \mathrm{~N}... | 1 |
23. The probability that the cube selected has two sides painted is
a. $1 / 9$
b. $4 / 9$
c. $8 / 27$
d. none of these | 1 |
9. If $\mathrm{A}=[\begin{array}{cc}\alpha & \beta \\ \gamma & -\alpha\end{array}]$ is such that $\mathrm{A}^{2}=\mathrm{I}$, then
(A) $1+\alpha^{2}+\beta \gamma=0$
(B) $1-\alpha^{2}+\beta \gamma=0$
(C) $1-\alpha^{2}-\beta \gamma=0$
(D) $1+\alpha^{2}-\beta \gamma=0$ | 1 |
47. A compound that gives a positive iodoform test is
(a) 1-pentanol
(b) 2-pentanone
(c) 3-pentanone
(d) pentanal | 1 |
19. The molarity of $98 \% \mathrm{H}_{2} \mathrm{SO}_{4}(d=1.8 \mathrm{~g} / \mathrm{mL})$ by wt. is
(a) 6 M
(b) 18 M
(c) 10 M
(d) 4 M | 1 |
16. Decomposition of $\mathrm{H}_{2} \mathrm{O}_{2}$ follows a first order reaction. In fifty minutes the concentration of $\mathrm{H}_{2} \mathrm{O}_{2}$ decreases from 0.5 to $0.125 \mathrm{M}$ in one such decomposition. When the concentration of $\mathrm{H}_{2} \mathrm{O}_{2}$ reaches $0.05 \mathrm{M}$, the rate of ... | 1 |
33. A system is said to be ................ if it can neither exchange matter nor energy with the surroundings. | 1 |
36. State with balanced equations what happens when :
Ethylene glycol is obtained by the reaction of ethylene with potassium permanganate. | 1 |
What gives regulations imposed by federal agencies the right to enforce their regulations? | 0 |
Brown is associated with what parties? | 0 |
Example 5.114 If $n(A)=5$ and $n(B)=3$, then find the number of onto functions from $A$ to $B$. | 1 |
1.\begin{array}{|c|c|}
\hline \text { Col } & \text { nII } \\
\hline \begin{array}{l}
\text { a. Differentiable function } f(x) \\
\text { satisfies the relation } f(1-x) \\
=f(1+x) \text { for all } x \in R \\
\text { b. Differentiable function } f(x) \\
\text { satisfies the relation } f(2-x) \\
+f(x)=0 \text { for ... | 1 |
4. Which of the following statements is/are correct?
(a) A photon is a positively charged nuclear particle.
(b) A photon is a particle of light energy.
(c) A photon is a quantum of light.
(d) A photon is a bundle of energy of definite magnitudes but not necessarily light energy. | 1 |
What were Religious officials perceived as in the Pre-Modern era? | 0 |
What was the time span of the Gupta Empire? | 0 |
22. The end correction (e) is $\left(l_{1}=\right.$ length of air column at first resonance and $l_{2}$ is length of air column at second resonance)
(a) $e=\frac{l_{2}-3 l_{1}}{2}$
(b) $e=\frac{l_{1}-3 l_{2}}{2}$
(c) $e=\frac{l_{2}-2 l_{1}}{2}$
(d) $e=\frac{l_{1}-2 l_{2}}{2}$ | 1 |
9. Two closed bulbs of equal volume $(V)$ containing an ideal gas initially at pressure $p_{i}$ and temperature $T_{l}$ are connected through a narrow tube of negligible volume. The temperature of one of the bulbs is then raised to $T_{2}$. The final pressure $p_{f}$ is :

(A) I $>$ III $>$ II
(B) II $>$ III $>$ I
(C) I $>$ II $>$ III
(D) III $>$ I $>$ II | 1 |
Why is an anion larger than a cation? | 0 |
46. At the point $P\left(a, a^{n}\right)$ on the graph of $y=x^{n}(n \in N)$ in the first quadrant, a normal is drawn. The normal intersects the $y$-axis at the point $(0, b)$. If $\lim _{a \rightarrow 0} b=\frac{1}{2}$, then $n$ equals
$$
\begin{array}{llll}
\text { a. } 1 & \text { b. } 3 & \text { c. } 2 & \text { ... | 1 |
19. One mole of a monatomic ideal gas is taken along two cyclic processes $E \rightarrow F \rightarrow G \rightarrow E$ and $E \rightarrow F \rightarrow H \rightarrow E$ as shown in the $p-V$ diagram.
$, then which of the following is/are correct?
(a) At $t=0$, particle is moving parallel to $y$-axis
(b) At $t=0$, direction of velocity and acceleration are perpendicular
(c) At $t=\sqrt{\frac{2}{3}}$, particle is movi... | 1 |
5. A radioactive nucleus $A$ with a half-life $T$, decays into a nucleus $B$. At $t=0$, there is no nucleus $B$. After sometime $t$, the ratio of the number of $B$ to that of $A$ is 0.3 . Then, $t$ is given by
(a) $t=T \frac{\log 1.3}{\log 2}$
(b) $t=T \log 1.3$
(c) $t=\frac{T}{\log 1.3}$
(d) $t=\frac{T \log 2}{2... | 1 |
94. $P(x)$ is a polynomial with integral coefficients such that for four distinct integers $a, b, c, d ; P(a)=P(b)=P(c)=P(d)=3$. If $P(e)=5$ ( $e$ is an integer), then
a. $e=1$
b. $e=3$
c. $e=4$
d. no real value of $e$ | 1 |
4. If $\Delta=\left|\begin{array}{ccc}\sin \theta \cos \phi & \sin \theta \sin \phi & \cos \theta \\ \cos \theta \cos \phi & \cos \theta \sin \phi & -\sin \theta \\ -\sin \theta \sin \phi & \sin \theta \cos \phi & 0\end{array}\right|$ then
a. $\Delta$ is independent of $\theta$
b. $\Delta$ is independent of $\phi$
c.... | 1 |
9. A police car with a siren of frequency $8 \mathrm{kHz}$ is moving with uniform velocity $36 \mathrm{~km} / \mathrm{h}$ towards a tall building which reflects the sound waves. The speed of sound in air is $320 \mathrm{~m} / \mathrm{s}$. The frequency of the siren heard by the car driver is
(a) $8.50 \mathrm{kHz}$
(b)... | 1 |
6. A homogeneous solid cylinder of length $L$ and cross-sectional area $A / 5$ is immersed such that it floats with its axis vertical at the liquid-liquid interface with length $L / 4$ in the denser liquid as shown in the figure. The lower density liquid is open to atmosphere having pressure $p_{0}$.
Then, density $D$... | 1 |
What have scientists started spending the most time on? | 0 |
13. In a simple harmonic motion
(a) the potential energy is always equal to the kinetic
energy
(b) the potential energy is never equal to the kinetic
energy ... | 1 |
57. Calcium burns in nitrogen to produce a white powder which dissolves in sufficient water to produce a gas (A) and an alkaline solution. The solution on exposure to air produces a thin solid layer of (B) on the surface. Identify the compounds (A) and (B). | 1 |
What are states not bound by? | 0 |
When Britain had to choose between its Japanese alliance or the US, which did it choose? | 0 |
How difficult is it to become a military drone pilot? | 0 |
1. Identify, which of the following energies can be positive (or zero) only?
(a) Kinetic energy
(b) Potential energy
(c) Mechanical energy
(d) Both kinetic and mechanical energies | 1 |
5. In figure the angle of inclination of the inclined plane is $30^{\circ}$. The horizontal velocity $V_{0}$ so that the particle hits the inclined plane perpendicularly is
(a) $V_{0}=\sqrt{\frac{2 g H}{5}}$
(b) $V_{0}=\sqrt{\frac{2 g H}{7}}$
(c) $V_{0}=\sqrt{\frac{g H}{5}}$
(d) $V_{0}=\sqrt{\frac{g H}{7}}$
 & Force of friction & (p) & field force \\
(b) & Normal reaction & (q) & contact force \\
(c) Force between two neutrons & (r) & ele... | 1 |
Is the United States about to go to war with Iran? | 0 |
Ex. 38. An organic compound $\mathrm{C}_{x} \mathrm{H}_{2 y} \mathrm{O}_{y}$ was burnt with twice the amount of oxygen needed for complete combustion to $\mathrm{CO}_{2}$ and $\mathrm{H}_{2} \mathrm{O}$. The hot gases when cooled to $0^{\circ} \mathrm{C}$ and 1 atm pressure measured 2.24 litres. The water collected dur... | 1 |
15. For a positive integer $n$, if the quadratic equation, $x(x+1)+(x+1)(x+2)+\ldots \quad+(x+\overline{n-1})(x+n)=10 n$ has two consecutive integral solutions, then $n$ is equal to
(2017 Main)
(a) 12
(b) 9
(c) 10
(d) 11 | 1 |
48. The value of $m$ for which one of the roots of $x^{2}-3 x+2 m=0$ is double of one of the roots of $x^{2}-x+m=0$ is
a. -2
b. 1
c. 2
d. none of these | 1 |
Has anyone read the essay How to raise a happy dog? Can anybody tell me why is it so important for human being to know that their dog is happy when the reality is that they don't even care about their dog's condition? What do you think is the relationship between human beings and their pets is? | 0 |
61. If $I=\int \sqrt{\frac{5-x}{2+x}} d x$, then $I$ equals
a. $\sqrt{x+2} \sqrt{5-x}+3 \sin ^{-1} \sqrt{\frac{x+2}{3}}+C$
b. $\sqrt{x+2} \sqrt{5-x}+7 \sin ^{-1} \sqrt{\frac{x+2}{7}}+C$
c. $\sqrt{x+2} \sqrt{5-x}+5 \sin ^{-1} \sqrt{\frac{x+2}{5}}+C$
d. None of these | 1 |
What is the best way to clear the HP2-T16 exam? | 0 |
Example 36.2 A message signal of frequency $10 \mathrm{kHz}$ and peak voltage of $10 \mathrm{~V}$ is used to modulate a carrier wave of frequency $1 \mathrm{MHz}$ and peak voltage of $20 \mathrm{~V}$.
Determine
(a) modulation index, (b) the side bands produced. | 1 |
Did Rey know it was the Millennium Falcon when she told Finn it hasn't been flown in years as they were escaping the First Order on Jakku? | 0 |
Why is Fetty Wap so popular? | 0 |
28. Which ore contains both iron and copper?
(a) Cuprite
(b) Chalcocite
(c) Chalcopyrite
(d) Malachite | 1 |
What does the Clinton Foundation do? | 0 |
What are some interesting facts about Google? | 0 |
What were the Allied powers referred to before 1917? | 0 |
What did The European renaissance mark? | 0 |
15. Prove that $\frac{3 !}{2(n+3)}=\sum_{r=0}^{n}(-1)^{r} \frac{{ }^{n} C_{r}}{{ }^{r+3} C_{r}}$. | 1 |
4. If $x>1, y>1, z>1$ are in GP, then $\frac{1}{1+\ln x}, \frac{1}{1+\ln y}$, $\frac{1}{1+\ln z}$ are in
(1998, 2M)
(a) AP
(b) HP
(c) GP
(d) None of these | 1 |
11. A uniform but increasing with time magnetic field exists in a cylindrical region. The direction of force on an electron at $P$ is
(a) towards right
(b) towards left
(c) into the plane of paper
(d) out of the plane of paper
 is
(a) $m g l$
(b) $(M+m) g l$
(c) $\frac{M}{2}+m \quad g l$
(d) $\frac{(M+m)}{2} g l$ | 1 |
7. A uniform cylinder of length $L$ and mass $M$ having cross-sectional area $A$ is suspended, with its length vertical from a fixed point by a massless spring such that it is half submerged in a liquid of density $\sigma$ at equilibrium position The extensition $x_{0}$ of the spring when it is in equilibrium is
(2013... | 1 |
4. The oxidation states of $\mathrm{Cr}$ in $\left[\mathrm{Cr}\left(\mathrm{H}_{2} \mathrm{O}\right)_{6}\right] \mathrm{Cl}_{3},\left[\mathrm{Cr}\left(\mathrm{C}_{6} \mathrm{H}_{6}\right)_{2}\right]$, and $\mathrm{K}_{2}\left[\mathrm{Cr}(\mathrm{CN})_{2}(\mathrm{O})_{2}\left(\mathrm{O}_{2}\right)\left(\mathrm{NH}_{3}\r... | 1 |
Is revenge good for health? | 0 |
Should I invest jio reliance? | 0 |
What did The Antebellum age mark? | 0 |
4.15 A magnetic field of $100 \mathrm{G}\left(1 \mathrm{G}=10^{-4} \mathrm{~T}\right)$ is required which is uniform in a region of linear dimension about 10 cm and area of cross-section about $10^{-3} \mathrm{~m}^2$. The maximum current-carrying capacity of a given coil of wire is 15 A and the number of turns per unit ... | 1 |
What is %A0 transfer in SBI? Amount has been debited from my account without my permission. | 0 |
How will the India-US relationship change if Hillary Clinton becomes the President of the US ? | 0 |
33. If $a, b, c$ are positive and are the $p^{\text {th }}, q^{\text {ll }}$ and $r^{\text {th }}$ terms, respectively, of a G.P., then $\Delta=\left|\begin{array}{lll}\log a & p & 1 \\ \log b & q & 1 \\ \log c & r & 1\end{array}\right|$ is
a. 0
b. $\log (a b c)$
c. $-(p+q+r)$
d. none of these | 1 |
60. If $a, b \in R, a \neq 0$ and the quadratic equation $a x^{2}-b x+1=0$ has imaginary roots then $(a+b+1)$ is
a. positive
b. negative
c. zero
d. dependent on the sign of $b$ | 1 |
What is the best phone I can buy under the price of 15000? | 0 |
6. The chemical nature of hydrogen peroxide is:
(a) Oxidising agent in acidic medium, but not in basic medium.
(b) Reducing agent in basic medium, but not in acidic medium.
(c) Oxidising and reducing agent in acidic medium, but not in basic medium
(d) Oxidising and reducing agent in both acidic and basic medium. | 1 |
Is it possible that the islands of the Caribbean will form a united country? | 0 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.