Question stringlengths 2 3k | label int64 0 1 |
|---|---|
What was Albania's largest foreign investor in 2013? | 0 |
Which British Prime Minister signed the Atlantic Charter? | 0 |
How can I improve my English pronunciation? | 0 |
Did the Rockingham Whigs ever regain power as a opposition to the government? | 0 |
26. Which of the following atoms has the highest first ionization energy?
(a) $\mathrm{K}$
(b) $\mathrm{Sc}$
(c) $\mathrm{Rb}$
(d) $\mathrm{Na}$ | 1 |
2.  | 1 |
3. Minimum velocity of the particle is
(a) $4 \sqrt{\frac{g L}{3}}$
(b) $2 \sqrt{\frac{g L}{3}}$
(c) $\sqrt{\frac{g L}{3}}$
(d) $3 \sqrt{\frac{g L}{3}}$
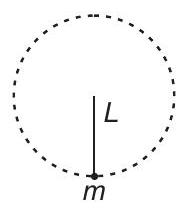 | 1 |
68. $\tan \left(\frac{\pi}{4}+\frac{1}{2} \cos ^{-1} x\right)+\tan \left(\frac{\pi}{4}-\frac{1}{2} \cos ^{-1} x\right), x \neq 0$, is equal to
a. $x$
b. $2 x$
c. $\frac{2}{x}$
d none of these | 1 |
(i) $\int \frac{x+2}{2 x^{2}+6 x+5} d x$ | 1 |
16. A weak base BOH of concentration 0.02 mole/litre has a pH value of $10 \cdot 45$. If 100 mL of this base is mixed with 10 mL of $0 \cdot 1 \mathrm{M} \mathrm{HCl}$, what will be the pH of the mixture? | 1 |
How do I edit a question I asked on Quora? | 0 |
What must happen to a statute for it to become unconstitutional? | 0 |
What is another term used for Special Education Services? | 0 |
(ii) Chlorine gas is passed into a solution of NaI in water. | 1 |
- Example 10.4 A small block of mass $100 \mathrm{~g}$ moves with uniform speed in a horizontal circular groove, with vertical side walls of radius $25 \mathrm{~cm}$. If the block takes $2.0 \mathrm{~s}$ to complete one round, find the normal constant force by the side wall of the groove. | 1 |
Can I change the stream Unix to Java during the ILP at TCS? | 0 |
How can I stop hating someone? | 0 |
Which problems money can not solve? | 0 |
72. The sum to 50 terms of the series $1+2\left(1+\frac{1}{50}\right)+3\left(1+\frac{1}{50}\right)^{2}$
$+\cdots$ is given by
a. 2500
b. 2550
c. 2450
d. none of these | 1 |
103 Light containing a mixture of two wavelengths, 500 and 600 nm , is incident normally on a diffraction grating. It is desired (1) that the first and second maxima for each wavelength appear at $\theta \leq 30^{\circ}$, (2) that the dispersion be as high as possible, and (3) that the third order for the 600 nm light ... | 1 |
What does TDM stand for? | 0 |
What is "Lack Thereof"? | 0 |
20. The centre of a wheel rolling on a plane surface moves with a speed $v_0$. A particle on the rim of the wheel at the same level as the centre will be moving at speed
(a) zero
(b) $v_0$
(c) $\sqrt{ } 2 v_0$
(d) $2 v_0$. | 1 |
Which caste does the surname "Das" belong to? | 0 |
In addition to being practically useful what other aspects did a building need in order to be considered functional? | 0 |
12.17 Explain the terms Inductive and Electromeric effects. Which electron displacement effect explains the following correct orders of acidity of the carboxylic acids?
(a) $\mathrm{Cl}_{3} \mathrm{CCOOH}>\mathrm{Cl}_{2} \mathrm{CHCOOH}>\mathrm{ClCH}_{2} \mathrm{COOH}$
(b) $\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{COO... | 1 |
Is vodka and whiskey a good drink? | 0 |
7. When a sphere rolls down an inclined plane, then identity the correct statement related to the work done by friction force
(a) The friction force does positive translational work
(b) The friction force does negative rotational work
(c) The net work done by friction is zero
(d) All of the above | 1 |
15. A particle of mass $m=3 \mathrm{~kg}$ moves along a straight line $4 y-3 x=2$ where $x$ and $y$ are in metre, with constant velocity $v=5 \mathrm{~ms}^{-1}$. The magnitude of angular momentum about the origin is
(a) $12 \mathrm{~kg} \mathrm{~m}^{2} \mathrm{~s}^{-1}$
(b) $6.0 \mathrm{~kg} \mathrm{~m}^{2} \mathrm{~s}... | 1 |
17. When 1 mole of a solute is dissolved in 1 kg of $\mathrm{H}_{2} \mathrm{O}$, boiling point of solution was found to be $100 \cdot 5^{\circ} \mathrm{C}$. $K_{b}$ for $\mathrm{H}_{2} \mathrm{O}$ is
(a) 0.5
(b) 100
(c) $100 \cdot 5$
(d) 95.5 | 1 |
What is the Pomodoro Technique? | 0 |
Ex. 3. What will be the number of oxygen atoms in 1 mole of $\mathrm{O}_{2}$ ? | 1 |
7. Let $A$ be a set of $n(\geq 3)$ distinct elements. The number of triplets $(x, y, z)$ of the $A$ elements in which at least two coordinates is equal to
a. ${ }^{1} \mathrm{P}_{3}$
b. $n^{3}-{ }^{n} \mathrm{P}_{3}$
c. $3 n^{2}-2 n$
d. $3 n^{2}(n-1)$ | 1 |
12. For any two vectors $\vec{u}$ and $\vec{v}$, prove that
a. $(\vec{u} \cdot \vec{v})^{2}+|\vec{u} \times \vec{v}|^{2}=|\vec{u}|^{2}|\vec{v}|^{2}$ and
b. $\left(\overrightarrow{1}+|\vec{u}|^{2}\right)\left(\overrightarrow{1}+|\vec{v}|^{2}\right)=(1-\vec{u} \cdot \vec{v})^{2}+|\vec{u}+\vec{v}+(\vec{u} \times \vec{v})|... | 1 |
What is tension and compression mean in bridges? | 0 |
114. A man has three friends. The number of ways he can invite one friend everyday for dinner on six successive nights so that no friend is invited more than three times is
a. 640
b. 320
c. 420
d. 510 | 1 |
How do I get banned on Quora? | 0 |
Example 5 Find the coordinates of the focus, axis, the equation of the directrix and latus rectum of the parabola $y^{2}=8 x$. | 1 |
Does Instagram save my login information even after I delete the app and reinstall? | 0 |
How can we get rid of studies? | 0 |
During their life time do plants produce more oxygen than they use at nights, winter, cloudy days etc? | 0 |
How should I get girls to hug me? | 0 |
Example 24 If a machine is correctly set up, it produces $90 \%$ acceptable items. If it is incorrectly set up, it produces only $40 \%$ acceptable items. Past experience shows that $80 \%$ of the set ups are correctly done. If after a certain set up, the machine produces 2 acceptable items, find the probability that t... | 1 |
Which is the only stste without reception statutes? | 0 |
How can I become more attractive to men? | 0 |
4. Two poles standing on a horizontal ground are of heights $5 \mathrm{~m}$ and $10 \mathrm{~m}$, respectively. The line joining their tops makes an angle of $15^{\circ}$ with the ground. Then, the distance (in $\mathrm{m}$ ) between the poles, is
(a) $5(\sqrt{3}+1)$
(b) $\frac{5}{2}(2+\sqrt{3})$
(c) $10(\sqrt{3}-1)$
(... | 1 |
7. The distance between the cathode (filament) and the target in an X-ray tube is $1.5 \mathrm{~m}$. If the cutoff wavelength is $30 \mathrm{pm}$, find the electric field between the cathode and the target. | 1 |
Geologically what is Indonesia's region most active for? | 0 |
Why do some people decide to join groups like ISIS? | 0 |
29. With respect to graphite and diamond, which of the statement(s) given below is (are) correct?
(a) Graphite is harder than diamond.
(b) Graphite has higher electrical conductivity than diamond.
(c) Graphite has higher thermal conductivity than diamond.
(d) Graphite has higher $\mathrm{C}-\mathrm{C}$ bond order... | 1 |
24. Why do we use the carbon dating to calculate the age of the fossil?
(a) Rate of exchange of carbon between atmosphere and living is slower than decay of $\mathrm{C}^{14}$
(b) It is not appropriate to use $\mathrm{C}^{14}$ dating to determine age
(c) Rate of exchange of $\mathrm{C}^{14}$ between atmosphere and li... | 1 |
How do you avoid wasting time? | 0 |
114. If $\alpha, \beta(\beta>\alpha)$ are the roots of $g(x)=a x^{2}+b x+c=0$ and $f(x)$ is an even function, then $\int_{\alpha}^{\beta} \frac{e^{f\left(\frac{g(x)}{x-\alpha}\right)} d x}{e^{f\left(\frac{g(x)}{x-\alpha}\right)}+e^{f\left(\frac{g(x)}{x-\beta}\right)}}$ is equal to
a. $\left|\frac{b}{2 a}\right|$
b. $\f... | 1 |
4. The set of all points, where the function $f(x)=\frac{x}{1+|x|}$ is differentiable is
a. $(-\infty, \infty)$
b. $[0, \infty)$
c. $(-\infty, 0) \cup(0, \infty)$
d. $(0, \infty)$ | 1 |
Example 3 Find the mean deviation about the median for the following data:
$$
3,9,5,3,12,10,18,4,7,19,21
$$ | 1 |
How can I get into a top university? | 0 |
What are some video tutorials that I can use to start learning data structures and algorithms from scratch? | 0 |
What are biomorphism and zoomorphic architecture based after design wise? | 0 |
How can I stop hoarding eBooks? | 0 |
25. For a sparingly soluble salt $A_{\infty} B$, the relationship of its solubility product $\left(L_{S}\right)$ with its solubility $(S)$ is
$$
\text { (a) } L_{S}=S^{p+q} \cdot p^{p} \cdot q^{q}
$$
(b) $L_{S}=S^{p+q} \cdot p^{q} \cdot q^{p}$
(c) $L_{S}=S^{p q} \cdot p^{p} \cdot q^{q}$
(d) $L_{S}=S^{p q} \cdot(p... | 1 |
12. If a reaction with $t_{1 / 2}=69.3$ seconds, has a rate constant value of $10^{-2}$ per second, the order is
(a) 0
(b) 1
(c) 2
(d) 3 | 1 |
How can I determine pump the operating pump head? | 0 |
47. The thermal dissociation of equilibrium of $\mathrm{CaCO}_{3}(\mathrm{~s})$ is studied under different conditions
$$
\mathrm{CaCO}_{3}(\mathrm{~s}) \rightleftharpoons \mathrm{CaO}(\mathrm{~s})+\mathrm{CO}_{2}(\mathrm{~g})
$$
For this equilibrium, the correct statement(s) is(are)
(a) $\Delta H$ is dependent on tem... | 1 |
Where's a good place to take a date in Philadelphia? | 0 |
What are the best web development companies in Kolkata? | 0 |
Example 15 Find real $\theta$ such that
$$
\frac{3+2 i \sin \theta}{1-2 i \sin \theta} \text { is purely real. }
$$ | 1 |
Como conectar a Quora en español? | 0 |
Which is a suitable inpatient drug and alcohol rehab center in Burke County GA? | 0 |
Which is the best torrent site for bollywood movies? | 0 |
27. Find equivalent capacitance between points $A$ and $B$. [Assume each conducting plate is having same dimensions and neglect the thickness of the plate, $\frac{\varepsilon_{0} A}{d}=7 \mu \mathrm{F}$, where $A$ is area of plates]

shown. The reading of the spring balance (in N ) is :
(a... | 1 |
16. The effective resistance between points $P$ and $Q$ of the electrical circuit shown in the figure is
$(2002,2 \mathrm{M})$
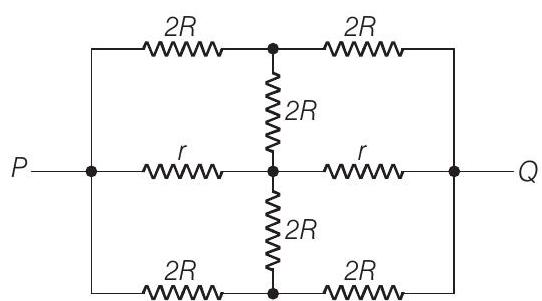
(a) $\frac{2 R r}{R+r}$
(b) $\frac{8 R(R+r)}{3 R+r}$
(c) $2 ... | 1 |
New Delhi shares the position as the most targeted retail destination in Asia-Pacific markets with what other city? | 0 |
During which scientific period did vertebrates appear? | 0 |
What is the salary of iit professors? | 0 |
If you cooled an ice cube to the temperature of absolute zero and put it in a glass of water, would that water freeze instantly? | 0 |
4. $\sin ^{-1}(x \sqrt{x}), 0 \leq x \leq 1$ | 1 |
What makes you feel lonely? | 0 |
What does LCPS stand for? | 0 |
What is the correct way to say "go" in German? | 0 |
What did women acknowledge who considered themselves as lesbian or bisexual? | 0 |
10.3 Why are alkali metals not found in nature ? | 1 |
1. For uranium nucleus how does its mass vary with volume?
(JEE 2003)
(a) $m \propto V$
(b) $m \propto 1 / V$
(c) $m \propto \sqrt{V}$
(d) $m \propto V^{2}$ | 1 |
What is the chemical equation for iron and sulphuric acid? | 0 |
1. Arrange the following solutions in the decreasing order of $\mathrm{pOH}$ :
(A) $0.01 \mathrm{M} \mathrm{HCl}$
(B) $0.01 \mathrm{M} \mathrm{NaOH}$
(C) $0.01 \mathrm{M} \mathrm{CH}_{3} \mathrm{COONa}$
(D) $0.01 \mathrm{M} \mathrm{NaCl}$
(a) $(\mathrm{A})>(\mathrm{C})>(\mathrm{D})>(\mathrm{B})$
(b) $(\mathrm{A})... | 1 |
26. The equilibrium $\mathrm{SO}_2 \mathrm{Cl}_2(\mathrm{~g}) \rightleftharpoons \mathrm{SO}_2(\mathrm{~g})+\mathrm{Cl}_2(\mathrm{~g})$ is attained at $25^{\circ} \mathrm{C}$ in a closed container and an inert gas, helium, is introduced. Which of the following statements are correct?
(a) Concentrations of $\mathrm{SO}_... | 1 |
11.38 If the starting material for the manufacture of silicones is $\mathrm{RSiCl}_3$, write the structure of the product formed. | 1 |
How do tachyons move faster than the speed of light? | 0 |
How long can humans keep wasting natural resources before they all run out? | 0 |
What do you want to do before you die? | 0 |
What are the best hotels in Marseille, France? | 0 |
Why do so many people find a high IQ level so important? Why is it necessary to test people on their IQ levels? Why do you feel so lonely when your IQ is far above the average? | 0 |
What is a notable cross made during this era? | 0 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.