Question stringlengths 2 3k | label int64 0 1 |
|---|---|
5. The density of a material in the shape of a cube is determined by measuring three sides of the cube and its mass. If the relative errors in measuring the mass and length are respectively $1.5 \%$ and $1 \%$, the maximum error in determining the density is
(a) $6 \%$
(b) $2.5 \%$
(c) $3.5 \%$
(d) $4.5 \%$ | 1 |
What was the best ground-attack aircraft of World War 2 (on either Allied or Axis side)? | 0 |
What do the low corruption countries have solidly established? | 0 |
Which are the best CA firms for articleship in kolkata? | 0 |
What is best alternative for Kat.al? | 0 |
6. Identify the correct order of reactivity in electrophilic substitution reactions of the following compounds
=\log _{x}(\log x)$, then $f^{\prime}(x)$ at $x=e$ is
$(1985,2 M)$ | 1 |
27. What is the maximum number of electrons in an atom in which the last electron, filled, has the following quantum numbers?
(a) $n=3$
(b) $n=3$, and $l=1$
(c) $n=3, l=1$ and $m=-1$
(d) $n=3, l=1, m=-1$ and $s=+\frac{1}{2}$
[(a) 30,
(b) 18
(c) 16
(d) 13] | 1 |
73. The sum to 50 terms of the series $\frac{3}{1^{2}}+\frac{5}{1^{2}+2^{2}}+\frac{7}{1^{2}+2^{2}+3^{2}}$ $+\cdots$ is
a. $\frac{100}{17}$
b. $\frac{150}{17}$
c. $\frac{200}{51}$
d. $\frac{50}{17}$ | 1 |
Will there be change in gravity and time dilation if the masses of all our planets are considered to be the same? What is the case when angular velocities are also considered the same? | 0 |
25. How is boron obtained from borax? Give chemical equations with reaction conditions. Write the structure of $\mathrm{B}_{2} \mathrm{H}_{6}$ and its reaction with $\mathrm{HCl}$. | 1 |
Can I transfer to Indian university from a Australian university for masters? | 0 |
9. Solve $\tan \left(\frac{\pi}{2} \cos \theta\right)=\cot \left(\frac{\pi}{2} \sin \theta\right)$. | 1 |
39. $K_{\text {sp }}$ of AgCl at $25^{\circ} \mathrm{C}$ is $1.782 \times 10^{-10}$. At $35^{\circ} \mathrm{C}, K_{s p}$ is $4.159 \times 10^{10}$. What are $\Delta H^{0}$ and $\Delta S^{0}$ for the reaction: $\mathrm{AgCl}(\mathrm{s})=\mathrm{Ag}^{+}(\mathrm{aq})+\mathrm{Cl}^{-}(\mathrm{aq}) ? \quad\left(\Delta H^{0}=... | 1 |
3.39 Considering the elements $\mathrm{B}, \mathrm{C}, \mathrm{N}, \mathrm{F}$, and Si , the correct order of their non-metallic character is :
(a) $\mathrm{B}>\mathrm{C}>\mathrm{Si}>\mathrm{N}>\mathrm{F}$
(b) Si $>$ C $>$ B $>$ N $>$ F
(c) $\mathrm{F}>\mathrm{N}>\mathrm{C}>\mathrm{B}>\mathrm{Si}$
(d) $\mathrm{F}>\math... | 1 |
What sort of programming experience does one need to start learning and become an AWS Certified Solutions architect? | 0 |
Example 10 Find the ratio in which the line segment joining the points $(4,8,10)$ and $(6,10,-8)$ is divided by the YZ-plane. | 1 |
42 An object, with mass $m$ and speed $v$ relative to an observer, explodes into two pieces, one three times as massive as the other; the explosion takes place in deep space. The less massive piece stops relative to the observer. How much kinetic energy is added to the system during the explosion, as measured in the ob... | 1 |
How old is Lake Manasarovar and Mount Kailash? | 0 |
How many feet are in 60 inches? | 0 |
What are some cool first prints with a 3D printer? | 0 |
9. The number of isomers for the compound with molecular formula $\mathrm{C}_{2} \mathrm{BrClFI}$ is [2001S]
(a) 3
(b) 4
(c) 5
(d) 6 | 1 |
35. Pure water freezes at 273 K and 1 bar. The addition of 34.5 g of ethanol to 500 g of water changes the freezing point of the solution. Use the $f . p$. depression constant of water as $2 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1}$. The figures shown below represents plot of vapour pressure (V.P.) versus temperatur... | 1 |
4. Keeping the number of moles, volume and temperature the same, which of the following are the same for all ideal gases?
(a) Rms speed of a molecule
(b) Density
(c) Pressure
(d) Average magnitude of momentum. | 1 |
What was the majority of the population of the western half of Bengal? | 0 |
92. (i) Write the chemical reactions involved in the extraction of metallic silver from argentite.
(ii) Write the balanced chemical equation for developing photographic films | 1 |
16. The IUPAC name of the compound
$$
\mathrm{CH}_{2}=\mathrm{CH}-\mathrm{CH}\left(\mathrm{CH}_{3}\right)_{2} \text { is }
$$
(a) 1, 1-dimethyl-2-propene
(b) 3-methyl-1-butene
(c) 2-vinylpropane
(d) 1-isopropylethylene | 1 |
Are all infinite languages uncomputable? Why? Why not? | 0 |
What language was spoken in the Byzantine Empire? | 0 |
(ii) $\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{CHBrCH}_{2} \mathrm{CH}\left(\mathrm{CH}_{3}\right) \mathrm{CHO}$ | 1 |
18. A rectangular sheet of tin 45 cm by 24 cm is to be made into a box without top, by cutting off square from each corner and folding up the flaps. What should be the side of the square to be cut off so that the volume of the box is maximum ? | 1 |
Can I get a good job in a Salesforce domain while switching from testing? | 0 |
7. When electricity is passed through a solution of $\mathrm{AlCl}_{3}, 13 \cdot 5 \mathrm{~g}$ of Al is deposited. The number of faradays must be
(a) 1.0
(b) 1.5
(c) 0.5
(d) 2.0 | 1 |
What were Vladimir Lenin's decisions caused by? | 0 |
3. $\sin (a x+b)$ | 1 |
What is the reason for the separation of the waters of the Indian and Atlantic Oceans? | 0 |
Is there any way to read Quora offline? | 0 |
What was the period of European empires competing to control Africa called? | 0 |
17. A square gate of size $1 \mathrm{~m} \times 1 \mathrm{~m}$ is hinged at its mid point. A fluid of density $\rho$ fills the space to the left of the gate. The force $F$ required to hold the gate stationary is
 of $\mathrm{Ag}_{2} \mathrm{CrO}_{4}$ in a $0.1 \mathrm{M} \mathrm{AgNO}_{3}$ solution is
(a) $1.1 \times 10^{-11}$
(b) $1.1 \times 10^{-10}$
(c) $1.1 \times 10^{-... | 1 |
What did the Guptas promote in their reigns? | 0 |
3.. From 6 different novels and 3 different dictionaries, 4 novels and 1 dictionary are to be selected and arranged in a row on a shelf, so that the dictionary is always in the middle. The number of such arrangements is
(2018 Main)
(a) atleast 1000
(b) less than 500
(c) atleast 500 but less than 750
(d) atleast 75... | 1 |
22. In terms of potential difference $V$, electric current $I$, permittivity $\varepsilon_{0}$, permeability $\mu_{0}$ and speed of light $c$, the dimensionally correct equations is/are
(a) $\mu_{0} I^{2}=\varepsilon_{0} V^{2}$
(b) $\varepsilon_{0} I=\mu_{0} V$
(c) $I=\varepsilon_{0} c V$
(d) $\mu_{0} c I=\vareps... | 1 |
8. A bimetallic strip is formed out of two identical strips- one of copper and the other of brass. The coefficients of linear expansion of the two metals are $\alpha_{C}$ and $\alpha_{B}$. On heating, the temperature of the strip goes up by $\Delta T$ and the strip bends to form an arc of radius of curvature $R$. Then,... | 1 |
7. Assertion : Motional induced emf $e=B v l$ can be derived from the relation $e=-\frac{d \varphi}{d t}$.
Reason : Lenz's law is a consequence of law of conservation of energy. | 1 |
59. If maximum distance of any point on the ellipse $x^{2}+2 y^{2}+2 x y=1$ from its centre be $r$, then $r$ is equal to
a. $3+\sqrt{3}$
b. $2+\sqrt{2}$
c. $\frac{\sqrt{2}}{\sqrt{3}-\sqrt{5}}$
d. $\sqrt{2-\sqrt{2}}$ | 1 |
62. For a first-order reaction $A \rightarrow P, t_{1 / 2}$ is 10 days. The time required for $\frac{1}{4}$ th conversion of A (in days) is: $(\ln 2=0.692, \ln 3=1.1)$
(a) 3.2
(b) 4.1
(c) 5
(d) 2.5 | 1 |
What are challenged to an unpopular political party? | 0 |
What does this sentence mean in Spanish? La copa mundial de fantasía es un juego de fútbol de mentiras, donde puedes entrenar a jugadores que juegan en la copa del mundo. | 0 |
9. Two capacitors $A$ and $B$ with capacities $3 \mu \mathrm{F}$ and $2 \mu \mathrm{F}$ are charged to a potential difference of $100 \mathrm{~V}$ and $180 \mathrm{~V}$, respectively. The plates of the capacitors are connected as shown in figure with one wire of each capacitor free. The upper plate of $A$ is positive a... | 1 |
How can I pass the Oracle 1Z0-520 exam? | 0 |
What pleasure do people get from answering questions on Quora? | 0 |
36 A beam of light of a single wavelength is incident perpendicularly on a double-slit arrangement, as in Fig. 35-10. The slit widths
are each $46 \mu \mathrm{m}$ and the slit separation is 0.30 mm . How many complete bright fringes appear between the two first-order minima of the diffraction pattern? | 1 |
The son of the security guard in our society is not willing to go to school. Even after many attempts to persuade him he denies it saying he has to work and earn, how do I get him to study? | 0 |
26. The outermost electronic configuration of the most electronegative element is
(a) $n s^{2} n p^{3}$
(b) $n s^{2} n p^{4}$
(c) $n s^{2} n p^{5}$
(d) $n s^{2} n p^{6}$ | 1 |
7. The gas evolved on heating $\mathrm{CaF}_{2}$ and $\mathrm{SiO}_{2}$ with concentrated $\mathrm{H}_{2} \mathrm{SO}_{4}$, on hydrolysis gives a white gelatinous precipitate. The precipitate is:
(a) hydrofluosilicic acid
(b) silica gel
(c) silicic acid
(d) calciumfluorosilicate | 1 |
13. A string vibrates in 5 segments to a frequency of $480 \mathrm{~Hz}$. The frequency that will cause it to vibrate in 2 segments will be
(a) $96 \mathrm{~Hz}$
(b) $192 \mathrm{~Hz}$
(c) $1200 \mathrm{~Hz}$
(d) $2400 \mathrm{~Hz}$ | 1 |
When was the Sino-British Joint Declaration? | 0 |
14. Given that $\log (2)=0.3010 \ldots$, the number of digits in the number $2000^{2000}$ is
a. 6601
b. 6602
c. 6603
d. 6604 | 1 |
Which region manufactures textiles, heavy industrial products & high-tech electronic goods? | 0 |
What label for the Greek market were artists on under the Sony umbrella? | 0 |
1. The sum of series $1+\frac{1^{3}+2^{3}}{1+2}+\frac{1^{3}+2^{3}+3^{3}}{1+2+3}+\ldots$ $+\frac{1^{3}+2^{3}+3^{3}+\ldots+15^{3}}{1+2+3+\ldots+15}-\frac{1}{2}(1+2+3+\ldots+15)$ is equal to
(2019 Main, 10 April II)
(a) 620
(b) 660
(c) 1240
(d) 1860 | 1 |
What is the difference between Android Lollipop 5.1.1 and Marshmallow 6.0? | 0 |
What is A-GPS? | 0 |
12. Which of the following statements is false?
a. vectors $\vec{P}, \vec{A}$ and $\vec{P} \times \vec{B}$ are linearly dependent.
b. vectors $\vec{P}, \vec{B}$ and $\vec{P} \times \vec{B}$ are linearly independent.
c. $\vec{P}$ is orthogonal to $\vec{B}$ and has length $1 / \sqrt{2}$.
d. none of the above. | 1 |
13. When water droplets merge to form a bigger drop
(a) energy is liberated
(b) energy is absorbed
(c) energy is neither liberated nor absobred
(d) energy may either be liberated or absorbed depending on the nature of the liquid. | 1 |
64. Among the following, the correct statement is
(a) Between $\mathrm{NH}_{3}$ and $\mathrm{PH}_{3}, \mathrm{NH}_{3}$ is a better electron donor because the lone pair of electrons occupies spherical $s$-orbital and is less directional
(b) Between $\mathrm{NH}_{3}$ and $\mathrm{PH}_{3}, \mathrm{PH}_{3}$ is a better e... | 1 |
How many years ago did Homo sapiens reached the Southeast Asia region? | 0 |
Which are the places with communal discord in India? | 0 |
When were architectural writings first made? | 0 |
6. In Column I, some statements are given related to work done by a force on an object while in Column II the sign and information about value of work done is given. Match the entries of Column I with the entries of Column II.
\begin{tabular}{l|l}
\hline \multicolumn{1}{c|}{ Column I } & \multicolumn{1}{c}{ Column II ... | 1 |
When did Egypt stop being a British client state? | 0 |
27. If the equation $z^{4}+a_{1} z^{3}+a_{2} z^{2}+a_{3} z+a_{4}=0$, where $a_{1}, a_{2}, a_{3}$, $a_{4}$ are real coefficients different from zero, has a purely imaginary root, then the expression $a_{3} /\left(a_{1} a_{2}\right)+\left(a_{1} a_{4}\right) /\left(a_{2} a_{3}\right)$ has the value equal to
a. 0
b. 1
c. -... | 1 |
What ruler opposed the British use of permits and ended up at war? | 0 |
1. Mark the correct statements:
(a) Equations of special relativity are not applicable for small speeds.
(b) Equations of special relativity are applicable for all speeds.
(c) Nonrelativistic equations give exact result for small speeds.
(d) Nonrelativistic equations never give exact result. | 1 |
What is the count of Tech Startup in 2012 and 2013 in Silicon Valley? | 0 |
What were said to have been caused by government policy failures? | 0 |
Why shouldn't one commit suicide? | 0 |
11. If $\alpha+\beta=\pi / 2$ and $\beta+\gamma=\alpha$, then $\tan \alpha$ equals
a. $2(\tan \beta+\tan . \gamma)$
b $\tan \beta+\tan \gamma$
c. $\tan \beta+2 \tan \gamma$
d. $2 \tan \beta+\tan \gamma$
(IIT-JEE, 2001) | 1 |
What are the best Tamil movies of all time? | 0 |
30. Find the ratio of currents as measured by ammeter in two cases when the key is open and when the key is closed
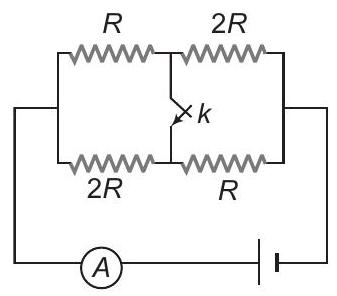
(a) $9 / 8$
(b) $10 / 11$
(c) $8 / 9$
(d) None of these | 1 |
In a ticket booked for one couple, if one passenger gets a confirmed berth, and the other is still waitlisted after chart preparation, can the waitlisted passenger board the train? | 0 |
31. The major product of the following reaction is
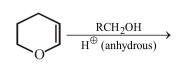
(a) a hemiacetal
(b) an acetal
(c) an ether
(d) an ester | 1 |
What fictional character reminds you most of yourself? | 0 |
Does light have kinetic energy? | 0 |
Is there a possibility that there is already a cure for cancer? | 0 |
How is a mass shooting like the one at the Fort Lauderdale airport possible with today's security upgrades? | 0 |
Which treaty had given Hong Kong to Britain? | 0 |
Can I ever get a free domain name? | 0 |
11. If $E$ and $F$ are events with $P(E) \leq P(F)$ and $P(E \cap F)>0$, then
a. occurrence of $E \Rightarrow$ occurrence of $F$
b. occurrence of $F \Rightarrow$ occurrence of $E$
c. non-occurrence of $E \Rightarrow$ non-occurrence of $F$
d. none of the above implications holds | 1 |
1. What compound is represented by the Newman projection shown?
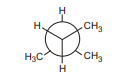
(A) $\mathrm{CH}_{3}\mathrm{CH}_{2}\mathrm{CH}_{3}$
(B) $\left(\mathrm{CH}_{3}\right)_{2} \mathrm{CHCH}_{2} \mathrm{CH}_{3}$
(C) $\mathrm{CH}_{3} \mathrm... | 1 |
6. A deuteron (the nucleus of an isotope of hydrogen) has a mass of $3.34 \times 10^{-27} \mathrm{~kg}$ and a charge of $+e$. The deuteron travels in a circular path with a radius of $6.96 \mathrm{~mm}$ in a magnetic field with magnitude $2.50 \mathrm{~T}$.
(a) Find the speed of the deuteron.
(b) Find the time requir... | 1 |
What sort of response does the stimulus cause? | 0 |
Being stuck in YouTube buffering is very irritating. What can one do to keep oneself busy and entertained at the same time? | 0 |
Why does Legendary Indian cricketer MS Dhoni has so many haters in his own country? | 0 |
What should everyone know about finance? | 0 |
Why do young female teachers prefer murga punishment to boys and spare girls by warning only? | 0 |
22. Which of the following would decrease the pH of $25 \mathrm{~cm}^3$ of a 0.01 M solution of HCl ?
(a) The addition of Mg
(b) The addition of $25 \mathrm{~cm}^3$ of 0.02 M HCl
(c) The addition of $25 \mathrm{~cm}^3$ of 0.005 M HCl
(d) None of these | 1 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.