Question stringlengths 2 3k | label int64 0 1 |
|---|---|
When entrusted funds are stolen, it is called what? | 0 |
42. In the following monobromination reaction, the number of possible chiral products is
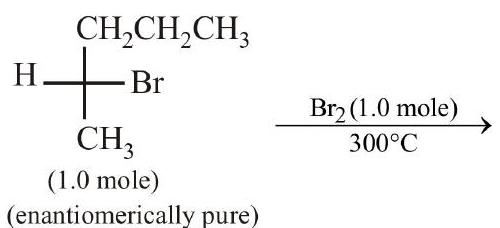
( 1.0 mole $)$
(enantiomerically pure) | 1 |
1. A circuit connected to an AC source of emf $e=e_{0} \sin (100 t)$ with $t$ in seconds, gives a phase difference of $\frac{\pi}{4}$ between the emf $e$ and current $i$. Which of the following circuits will exhibit this?
(a) $R C$ circuit with $R=1 \mathrm{k} \Omega$ and $C=1 \mu \mathrm{F}$
(b) $R L$ circuit with... | 1 |
23. Vectors $\vec{a}=-4 \hat{i}+3 \hat{k} ; \vec{b}=14 \hat{i}+2 \hat{j}-5 \hat{k}$ are laid off from one point. Vector $\vec{d}$, which is being laid off from the same point dividing the angle between vectors $\vec{a}$ and $\vec{b}$ in equal halves and having the magnitude $\sqrt{6}$, is
a. $\hat{i}+\hat{j}+2 \hat{k}$... | 1 |
Ex. 17. The vapour pressure of pure benzene at $88^{\circ} \mathrm{C}$ is 957 mm and that of toluene at the same temperature is 379.5 mm . Calculate the composition of a benzene-toluene mixture boiling at $88^{\circ} \mathrm{C}$. | 1 |
What do architecture schools look at more and more nowadays? | 0 |
9.13 What is the density of water at a depth where pressure is 80. 0 atm , given that its density at the surface is $1.03 \times
103 \mathrm{~kg} \mathrm{~m}^{-3}$ ? | 1 |
What is it like to work with Barack Obama? | 0 |
How can I be a singer like Enrique? | 0 |
If you have crossbred some peppers before, what did you think of them? | 0 |
35. The nuclides ${ }_{18}^{40} \mathrm{Ar}$ and ${ }_{19}^{41} \mathrm{~K}$ are
(a) isotopes
(b) isobars
(c) isotones
(d) none of these | 1 |
28. Amongst the compounds given, the one that would form a brilliant colored dye on treatment with $\mathrm{NaNO}_{2}$ in dil. $\mathrm{HCl}$ followed by addition to an alkaline solution of $\beta$-naphthol is
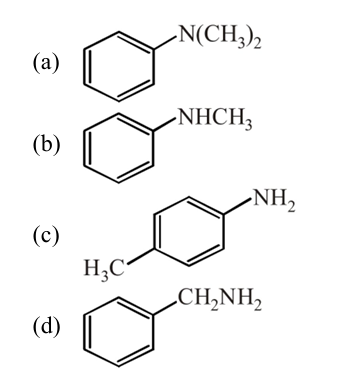 | 1 |
In what yer was the Battle of Manzikert fought? | 0 |
What is your review of Monterey, CA? | 0 |
22. Which one of the following exhibits the large number of oxidation states?
(a) $\mathrm{Ti}(22)$
(b) $\mathrm{V}(23)$
(c) $\operatorname{Cr}(24)$
(d) $\mathrm{Mn}(25)$ | 1 |
What are some of the real life application of Deque? | 0 |
24. Compare resonance Energy of following
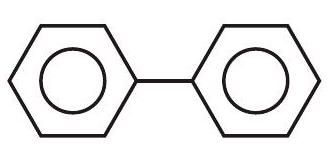
(a)
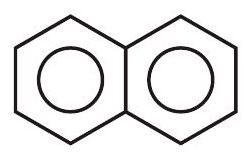
(b)
 the same, with 2, 0 and 1 lone pairs of electrons respectively
(b) the same, with 1,1 and 1 lone pairs of electrons respectively
(c) different, with 0,1 and 2 lone pairs of electrons respectively
... | 1 |
62. In a game a coin is tossed $2 n+m$ times and a player wins if he does not get any two consecutive outcomes same for atleast $2 n$ times in a row. The probability that player wins the game is
a. $\frac{m+2}{2^{2 n}+1}$
b. $\frac{2 n+2}{2^{2 n}}$
c. $\frac{2 n+2}{2^{2 n+1}}$
d. $\frac{m+2}{2^{2 n}}$ | 1 |
3. The shortest wavelength of $\mathrm{H}$ atom in the Lyman series is $\lambda_{1}$. The longest wavelength in the Balmer series is $\mathrm{He}^{+}$ is :
(a) $\frac{36 \lambda_{1}}{5}$
(b) $\frac{5 \lambda_{1}}{9}$
(c) $\frac{9 \lambda_{1}}{5}$
(d) $\frac{27 \lambda_{1}}{5}$ | 1 |
70. $A B C D$ is a convex quadrilateral and $3,4,5$ and 6 points are marked on the sides $A B, B C, C D$ and $D A$, respectively. The number of triangles with vertices on different sides is
a. 270
b. 220
c. 282
d. 342 | 1 |
12. A curve $C$ passes through $(2,0)$ and the slope at $(x, y)$ as $\frac{(x+1)^{2}+(y-3)}{x+1}$. Find the equation of the curve. Find the area bounded by curve and $x$-axis in fourth quadrant. | 1 |
3. 2 litres of ammonia at $13^{\circ} \mathrm{C}$ and 0.90 atmospheric pressure is neutralised by 134 mL of $\mathrm{H}_{2} \mathrm{SO}_{4}$ solution. Find the normality of the acid.
(0.57 N) | 1 |
Which is the oldest university in Asia? | 0 |
Why don't Asians talk about racism against them? | 0 |
12. A fair coin is tossed repeatedly. If the tail appears on first four tosses, then the probability of the head appearing on the fifth toss equals
a. 1/2
b. 1/32
c. $31 / 32$
d. $1 / 5$ | 1 |
Will the increasing amount of pseudoscience in America become detrimental? | 0 |
How should I stop being insecure? | 0 |
Spinoza believed that the universe was made up of one substance which is? | 0 |
What did Tanselle say about textual criticism? | 0 |
What does Drivers of Change promote? | 0 |
What was the first name of Sony Music Entertainment, Inc? | 0 |
(ix) 1-Chlorobutane to n-octane | 1 |
What were Sathvik Reddy Erla's marks in AIIMS (AIR 1)? | 0 |
In what year was Epic Records created? | 0 |
What was the social reform movement centered in Bengal in the 19th to early 20th century? | 0 |
1. 
(1) $\mathrm{W}(\mathrm{VI})$ is more stable than $\mathrm{Cr}(\mathrm{VI})$.
(2) in the presence of $\mathrm{HCl}$, permanganate titrations provide satisfactory results.
(3) some... | 1 |
How can I lose 70 pounds? | 0 |
What was the magnitude of the notable 2015 earthquake that shook New Delhi? | 0 |
What is the weather like in Kalkan, Turkey? | 0 |
In what year was Croatia admitted to the European Union? | 0 |
What are some of the best ways to gain weight in a healthy way? | 0 |
24. When steam condenses to water at $90^{\circ} \mathrm{C}$, the entropy of the system decreases. What must be true if the second law of thermodynamics is to be satisfied?
(a) Entropy of the universe also decreases.
(b) Entropy of the surroundings also decreases.
(c) Entropy of the surroundings increases to the same e... | 1 |
2. Sketch the graph of $y=|x+3|$ and evaluate $\int_{-6}^{0}|x+3| d x$. | 1 |
2. $\tan (A-B)=\frac{\tan A-\tan B}{1+\tan A \tan B}$ | 1 |
How do I install the required software packages and configure the old Linux command rcp? | 0 |
Why aren't books being added to the Bible in modern day? | 0 |
Which Indianised kingdom was based in eastern java? | 0 |
11. The acidic, basic and amphoteric oxides, respectively, are:
[Main Jan. 09, $2020(\mathrm{I})]$
(a) $\mathrm{Na}_2 \mathrm{O}, \mathrm{SO}_3, \mathrm{Al}_2 \mathrm{O}_3$
(b) $\mathrm{Cl}_2 \mathrm{O}, \mathrm{CaO}, \mathrm{P}_4 \mathrm{O}_{10}$
(c) $\mathrm{N}_2 \mathrm{O}_3, \mathrm{Li}_2 \mathrm{O}, \mathrm{Al}_2 ... | 1 |
(d) $\cos \omega t+\cos 3 \omega t+\cos 5 \omega t$ | 1 |
16. Which of the following curves represents a first order reaction?
(a)
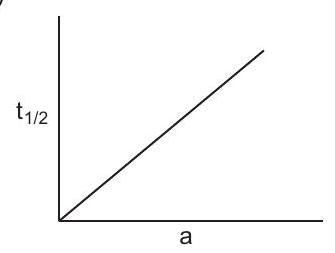
(b)
 only ionic
(b) covalent and coordinate
(c) only covalent
(d) covalent and ionic | 1 |
Are the Illuminati real? | 0 |
- Example 16 Consider the sets X and Y of Example 14. Find $\mathrm{X} \cap \mathrm{Y}$. | 1 |
I feel so lonely, how do I stop feeling this way? | 0 |
How many national parks does Hyderabad have? | 0 |
53. A slide projector has to project a $35 \mathrm{~mm}$ slide $(35 \mathrm{~mm} \times 23 \mathrm{~mm})$ on a $2 \mathrm{~m} \times 2 \mathrm{~m}$ screen at a distance of $10 \mathrm{~m}$ from the lens. What should be the focal length of the lens in the projector? | 1 |
Ex. 1. The degree of dissociation of acetic acid in a 0.1 M solution is $1.32 \times 10^{-2}$.
Find out the dissociation constant of the acid and its $\mathrm{p} K_{\mathrm{a}}$ value. | 1 |
Example 10.25 Solve $(d y / d x)=e^{x-y}\left(e^x-e^y\right)$. | 1 |
10. The diameters of circles (in mm ) drawn in a design are given below:
| Diameters | $33-36$ | $37-40$ | $41-44$ | $45-48$ | $49-52$ |
| :--- | :---: | :---: | :---: | :---: | :---: |
| No. of circles | 15 | 17 | 21 | 22 | 25 |
Calculate the standard deviation and mean diameter of the circles. | 1 |
26. A TV transmission tower has a height of $140 \mathrm{~m}$ and the height of the receiving antenna is $40 \mathrm{~m}$. What is the maximum distance upto which signals can be broadcasted from this tower in LOS (Line of Sight) mode? (Take, radius of earth $=6.4 \times 10^{6} \mathrm{~m}$ ).
(a) $65 \mathrm{~km}$
... | 1 |
What dynasty controlled Hyderabad until the early 18th century? | 0 |
How can I get free silver coins in India? | 0 |
8. A platinum resistance thermometer reads $0^{\circ}$ when its resistance is $80 \Omega$ and $100^{\circ}$ when its resistance is $90 \Omega$. Find the temperature at the platinum scale at which the resistance is $86 \Omega$. | 1 |
Ex. 63. What is the $\left[\mathrm{CO}_{3}^{2-}\right]$ in a $0.001 \mathrm{M} \mathrm{Na}_{2} \mathrm{CO}_{3}$ solution after the hydrolysis reactions have come to an equilibrium? $\mathrm{K}_{\mathrm{a}}\left(\mathrm{HCO}_{3}^{-}\right)=4.7 \times 10^{-11}$ | 1 |
1. A man (mass $=50 \mathrm{~kg}$ ) and his son (mass $=20 \mathrm{~kg}$ ) are standing on a frictionless surface facing each other. The man pushes his son, so that he starts moving at a speed of $0.70 \mathrm{~ms}^{-1}$ with respect to the man. The speed of the man with respect to the surface is
(a) $0.28 \mathrm{~m... | 1 |
I am an Indian commerce student studying in 12th. After my 12th, if I want to learn Android app developing, website making and online business, which career option should I choose? | 0 |
What are some of the rarely mentioned facts of the Roman empire? | 0 |
Illustration 3.27 Find the condition if lines $x=a y+b, z=c y+d$ and $x=a^{\prime} y+b^{\prime}, z=c^{\prime} y+d$ are perpendicular. | 1 |
What are some small things that makes you happy? | 0 |
What does MIH stand for? | 0 |
Example 2.33 Evaluate $\lim _{x \rightarrow 2} \frac{x^{2}-x-2}{x^{2}-2 x-\sin (x-2)}$. | 1 |
What were Vitruvius' three principles for a well made building? | 0 |
2. A particle moves along the $X$-axis as $x=u(t-2 \mathrm{~s})+a(t-2 \mathrm{~s})^{2}$.
(a) the initial velocity of the particle is $u$
(b) the acceleration of the particle is $a$
(c) the acceleration of the particle is $2 a$
(d) at $t=2 \mathrm{~s}$ particle is at the origin. | 1 |
How can I lose my weight quickly ? | 0 |
Can I charge a laptop battery outside my laptop? | 0 |
What are the top 10 custom ROMs? | 0 |
17. Correct order of Basic Strength ( $K_{\mathrm{b}}$ order) is :
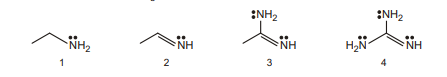
(A) $4>3>2>1$
(B) $1>2>3>4$
(C) $4>3>1>2$
(D) $1>4>3>2$ | 1 |
9. A gas is contained in a metallic cylinder fitted with a piston. The piston is suddenly moved in to compress the gas and is maintained at this position. As time passes the pressure of the gas in the cylinder
(a) increases
(b) decreases
(c) remains constant
(d) increases or decreases depending on the nature of the g... | 1 |
What other forms of writing materials were used during this period? | 0 |
42. In Fig. 12-7 and the associated sample problem, let the coefficient of static friction ms between the ladder and the pavement | 1 |
7. Aqueous solution of which salt will not contain ions with the electronic configuration $1 s^{2} 2 s^{2} 2 p^{6} 3 s^{2} 3 p^{6}$ ?
(a) $\mathrm{NaF}$
(b) $\mathrm{KBr}$
(c) $\mathrm{NaCl}$
(d) $\mathrm{CaI}_{2}$ | 1 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.