Question stringlengths 2 3k | label int64 0 1 |
|---|---|
21. The correct decreasing order of basic strength of the following species is . $\qquad$
$\mathrm{H}_{2} \mathrm{O}, \mathrm{NH}_{3}, \mathrm{OH}^{-}, \mathrm{NH}_{2}^{-}$
(A) $\mathrm{NH}_{2}^{-}>\mathrm{OH}^{-}>\mathrm{NH}_{3}>\mathrm{H}_{2} \mathrm{O}$
(B) $\mathrm{OH}^{-}>\mathrm{NH}_{2}^{-}>\mathrm{H}_{2} \math... | 1 |
12. Let $P(x)$ and $Q(x)$ be two polynomials. Suppose that $f(x)=P\left(x^{3}\right)+x Q\left(x^{3}\right)$ is divisible by $x^{2}+x+1$, then
a. $P(x)$ is divisible by $(x-1)$ but $Q(x)$ is not divisible by $x-1$
b. $Q(x)$ is divisible by $(x-1)$ but $P(x)$ is not divisible by $x-1$
c. Both $P(x)$ and $Q(x)$ are divis... | 1 |
What are some ways to start a business? | 0 |
21. Let $f(x)=\frac{3}{4} x+1$, and $f^{n}(x)$ be defined as $f^{2}(x)=f(f(x))$, and for $n \geq 2, f^{n+1}(x)=f\left(f^{n}(x)\right)$. If $\lambda=\lim _{n \rightarrow \infty} f^{n}(x)$, then
a. $\lambda$ is independent of $x$
b. $\lambda$ is a linear polynomial in $x$
c. the line $y=\lambda$ has slope 0
d. the line... | 1 |
How are the different types of African drums utilized? | 0 |
Who is the most inspirational person and how? | 0 |
48 Four particles are in the insulated box of Fig. 20-17. What are (a) the least multiplicity, (b) the greatest multiplicity, (c) the least entropy, and (d) the greatest entropy of the four-particle system? | 1 |
How is the word 'sparse' used in a sentence? | 0 |
2. Show that the function $f: \mathbf{R} \rightarrow \mathbf{R}$ given by $f(x)=x^{3}$ is injective. | 1 |
Where do I buy kimchi in PA? | 0 |
What are the best ways to enabled Gzip compression for a website? | 0 |
What are the proofs that aliens really exist? | 0 |
How many keywords are there in FOXPRO Programming Language in the latest version? | 0 |
3. Acceleration-time graph of a particle in SHM is as shown in figure. Match the following two columns.
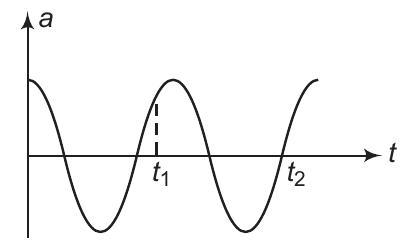
| | Column I | Column II | |
| :--- | :--- | :--- | :---: |
| (a) Displacement of p... | 1 |
3. A mixture of gases $\mathrm{O}_{2}, \mathrm{H}_{2}$ and $\mathrm{CO}$ are taken in a closed vessel containing charcoal. The graph that represents the correct behaviour of pressure with time is:
(a)
 If both assertion and reason are CORRECT, and reason is the CORRECT explanation of th... | 1 |
4.
| Column I | Column II |
| :--- | :--- |
| a. The length of the common chord of | |
| two circles of radii 3 and 4 units which | p. 1 |
| intersect orthogonally is $\frac{k}{5}$, then $k$ | |
| equals to | |
| | The circumference of the circle $x^{2}+y^{2}$ <br> $+4 x+12 y+p=0$ is bisected by the <br> circle... | 1 |
Is it true that Bangladeshi Muslims are more liberal than Pakistani muslims? | 0 |
Can I get a IRCTC tatkal ticket booking in this festive season easily? What tips should I follow to get a confirmed ticket? | 0 |
4. Let $\vec{\alpha}=3 \hat{\mathbf{i}}+\hat{\mathbf{j}}$ and $\vec{\beta}=2 \hat{\mathbf{i}}-\hat{\mathbf{j}}+3 \hat{\mathbf{k}}$ If $\vec{\beta}=\vec{\beta}_{1}-\vec{\beta}_{2}$, where $\vec{\beta}_{1}$ is parallel to $\vec{\alpha}$ and $\vec{\beta}_{2}$ is perpendicular to $\vec{\alpha}$, then $\vec{\beta}_{1} \time... | 1 |
What are some gift ideas for my wife on Christmas? | 0 |
18. During Searle's experiment, zero of the vernier scale lies between $3.20 \times 10^{-2} \mathrm{~m}$ and $3.25 \times 10^{-2} \mathrm{~m}$ of the main scale. The 20th division of the vernier scale exactly coincides with one of the main scale divisions. When an additional load of $2 \mathrm{~kg}$ is applied to the w... | 1 |
9. In which one of the following equilibria, $\mathrm{K}_{\mathrm{p}} \neq \mathrm{K}_{\mathrm{c}}$ ?
(a) $2 \mathrm{C}(\mathrm{s})+\mathrm{O}_{2}(\mathrm{~g}) \rightleftharpoons 2 \mathrm{CO}(\mathrm{g})$
(b) $2 \mathrm{HI}(\mathrm{g}) \rightleftharpoons \mathrm{H}_{2}(\mathrm{~g})+\mathrm{I}_{2}(\mathrm{~g})$
(c... | 1 |
31. How much amount of $\mathrm{NaCl}$ should be added to $600 \mathrm{~g}$ of water ( $\mathrm{r}=$ $1.00 \mathrm{~g} / \mathrm{mL}$ ) to decrease the freezing point of water to $-0.2^{\circ} \mathrm{C}$ ? (The freezing point depression constant for water $=2 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1}$ ) | 1 |
Example 3.49 Find the sum of the following series: 5 $+55+555+\cdots$ to $n$ terms. | 1 |
43. The equilibrium constant of the reaction $A_{2}(\mathrm{~g})+B_{2}(\mathrm{~g}) \rightleftharpoons 2 A B(\mathrm{~g})$
at $100^{\circ} \mathrm{C}$ is 50 . If a one litre flask, containing one mole of $A_{2}$ is connected to a two litre flask, containing two mole of $B_{2}$, how many mole of $A B$ will be formed at... | 1 |
32. Values of $a$ for which $f(x)=0$ has exactly one positive root.
a. $(0,1)$
b. $(-\infty, 1)$
c. $(-1,1)$
d. $(1, \infty)$ | 1 |
What sucks about Stack Overflow? | 0 |
How do I get into Ivy League colleges? I have not cared about college until my junior year of high school, so my grades weren’t good in the past. | 0 |
How does it feel to give a footjob(to a girl)? Does girls only do it for sake of boy happiness? | 0 |
4. Find the values of $1 / x$ for the following values of $x$ :
(i) $(2,5)$
(ii) $[-5,-1)$
(iii) $(3, \infty)$
(iv) $(-\infty,-2]$
(v) $[-3,4]$ | 1 |
What determines what literary structure a literary author uses for expression of ideas? | 0 |
27. The reaction :
[1985 - 1 Mark
$1 / 2 \mathrm{H}_{2}(\mathrm{~g})+\mathrm{AgCl}(\mathrm{s}) \rightarrow \mathrm{H}^{+}(\mathrm{aq})+\mathrm{Cl}^{-}(\mathrm{aq})+\mathrm{Ag}(\mathrm{s})$
occurs in the galvanic cell
(a) $\mathrm{Ag}|\mathrm{AgCl}(\mathrm{s})| \mathrm{KCl}$ (soln) $\mid \mathrm{AgNO}_{3}$ (soln) $\... | 1 |
What are expenses other than academic and hostel and mess in vit? | 0 |
18. Which one of the following ores is best concentrated by froth floatation method?
(a) Galena
(b) Malachite (c)Magnetite
(d) Siderite | 1 |
What does NARTH consider itself to be? | 0 |
9. The plane $4 x+7 y+4 z+81=0$ is rotated through a right angle about its line of intersection with the plane $5 x+3 y+10 z=25$. The equation of the plane in its new position is
(a) $x-4 y+6 z=106$
(b) $x-8 y+13 z=103$
(c) $x-4 y+6 z=110$
(d) $x-8 y+13 z=105$ | 1 |
What is the meaning of the word neuron in Greek? | 0 |
41. As two trains move along a track, their conductors suddenly notice that they are headed toward each other. Figure 2-31 gives their velocities $v$ as functions of time $t$ as the conductors slow the trains. The figure's vertical scaling is set by

$$ | 1 |
2. The $x$ - $t$ graph of a particle undergoing simple harmonic motion is shown below. The acceleration of the particle at $t=4 / 3 \mathrm{~s}$ is
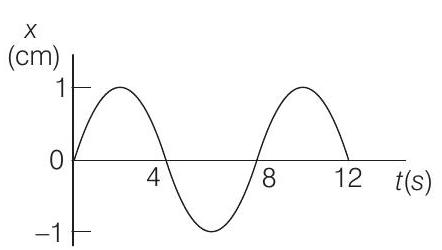
(a) $\frac{\sqrt{3}}{32} \pi^{2} \mathrm... | 1 |
What is the Tour de France in detail? | 0 |
44. $\Delta U$ is equal to
(a) isochoric work
(b) isoboric work
(c) adiabatic work
(d) isothermal work
(IIT 2017 Main) | 1 |
38. If $[x]$ and $\{x\}$ represent the integral and fractional parts of $x$, respectively, then the value of $\sum_{r=1}^{2000} \frac{\{x+r\}}{2000}$ is
a. $x$
b. $[x]$
c. $\{x\}$
d. $x+2001$ | 1 |
How many components are used in assessment? | 0 |
1. "A lift is descending with increasing speed". What are the directions of velocity and acceleration in the given statement? | 1 |
4.146. A capacitor with capacitance $C$ and a coil with active resistance $R$ and inductance $L$ are connected in parallel to a source of sinusoidal voltage of frequency $\omega$. Find the phase difference between the current fed to the circuit and the source voltage. | 1 |
(a) a solution of $\mathrm{Na}_2 \mathrm{CO}_3$ is alkaline ? | 1 |
Where can I get a list of family offices in Europe? | 0 |
Who was the cahir of the national gay and lesbian task force in the 1970s? | 0 |
7.23 Why are halogens strong oxidising agents? | 1 |
I'm currently in Class XII. I'll be giving NDA-2 2016 in september. Will I be call for SSB if I clear the written test? | 0 |
19. A spherical black body with a radius of $12 \mathrm{~cm}$ radiates $450 \mathrm{~W}$ power at $500 \mathrm{~K}$. If the radius were halved and the temperature doubled, the power radiated in watt would be
(a) 225
(b) 450
(c) 900
(d) 1800 | 1 |
86. The final solution contains
(a) $\mathrm{Pb}\left(\mathrm{NH}_{3}\right)_{4}^{2}$ and $\mathrm{CoCl}_{4}^{2}$
(b) $\mathrm{Al}\left(\mathrm{NH}_{3}\right)_{4}{ }^{3}$ and $\mathrm{Cu}\left(\mathrm{NH}_{3}\right)_{4}{ }^{2}$
(c) $\mathrm{Ag}\left(\mathrm{NH}_{3}\right)_{2}$ and $\mathrm{Cu}\left(\mathrm{NH}_{3}\r... | 1 |
Have any two satellites ever collided with each other? | 0 |
How would a war between the US and China play out? | 0 |
Is there a high risk that President Hillary Clinton will initiate a war with Russia? | 0 |
I am not able to install any apps other than from the Google Play Store. Why is this happening? | 0 |
What makes up Indigenous education? | 0 |
Neurons that release the same chemicals are following what rule? | 0 |
3.230. A uniform current of density $j$ flows inside an infinite plate of thickness $2 d$ parallel to its surface. Find the magnetic induction induced by this current as a function of the distance $x$ from the median plane of the plate. The magnetic permeability is assumed to be equal to unity both inside and outside t... | 1 |
How do I salvage all the messages that I may have received during the 13-month time I was absent on Whatsapp through a number? | 0 |
Will Moto G3 receive the Android N update? If yes, when? | 0 |
4. $x y+y^{2}=\tan x+y$ | 1 |
How can you overcome a writer's block? (songwriting) | 0 |
6. Which of the following quantities is the same for all ideal gases at the same temperature?
(a) The kinetic energy of 1 mole
(b) The kinetic energy of $1 \mathrm{~g}$
(c) The number of molecules in 1 mole
(d) The number of molecules in $1 \mathrm{~g}$ | 1 |
2. A gaseous oxide contains $30 \cdot 4 \%$ of nitrogen, one molecule of which contains one nitrogen atom. The density of the oxide relative to oxygen is
(a) 0.94
(b) 1.44
(c) 1.50
(d) 3.0 | 1 |
Example 7.5 Why does $\mathrm{NO}_2$ dimerise ? | 1 |
7. A spherically symmetric gravitational system of particles has a mass density $\rho=\begin{gathered}\rho_{0} \text { for } r \leq R \\ 0 \text { for } r>R\end{gathered}$, where $\rho_{0}$ is a constant. A test mass can undergo circular motion under the influence of the gravitational field of particles. Its speed $v$ ... | 1 |
89. An athlete needs to lose weight and decides to do it by "pumping iron." (a) How many times must an 80.0 kg weight be lifted a distance of 1.00 m in order to burn off 1.00 lb of fat, assuming that that much fat is equivalent to 3500 Cal? (b) If the weight is lifted once every 2.00 s, how long does the task take? | 1 |
What are businesses with a high percentage of its cost as fixed cost? | 0 |
What second period did the rise of the small kingdoms show? | 0 |
What is Fidel Castro's official title? | 0 |
How do you measure the pollution rate in a city with a population of 6 million people? | 0 |
How effective is scrapping 500 and 1000 rupee notes? Will it reduce black money? | 0 |
7. Identify the incorrect statement :
(a) The $\mathrm{S}-\mathrm{S}-\mathrm{S}$ bond angles in the $\mathrm{S}_{8}$ and $\mathrm{S}_{6}$ rings are the same.
(b) Rhombic and monoclinic sulphur have $\mathrm{S}_{8}$ molecules.
(c) $\mathrm{S}_{2}$ is paramagnetic like oxygen
(d) $\mathrm{S}_{8}$ ring has a crown sh... | 1 |
36. The radius of the orbit of an electron in a Hydrogen-like atom is $4.5 a_{0}$ where $a_{0}$ is the Bohr radius. Its orbital angular momentum is $\frac{3 h}{2 \pi}$. It is given that $h$ is Planck constant and $R$ is Rydberg constant. The possible wavelength $(s)$, when the atom de-excites, is (are)
(a) $\frac{9}... | 1 |
Ex. 44. The solubilities of AgCl in water, $0.01 \mathrm{M} \mathrm{CaCl}_{2}, 0.01 \mathrm{M} \mathrm{NaCl}$ and $0.05 \mathrm{M} \mathrm{AgNO}_{3}$ are denoted by $S_{1}, S_{2}, S_{3}$ and $S_{4}$ respectively. Which of the following relationships is correct?
(a) $S_{1}>S_{2}>S_{3}>S_{4}$
(b) $S_{1}>S_{3}>S_{2}>S_{4}... | 1 |
What percentage of the population in Southeast Asia practice Islam? | 0 |
1. If a person is suffering from the deficiency of noradrenaline,
what kind of drug can be suggested?
(a) Anti-inflammatory
(b) Antidepressant
(c) Antihistamine
(d) Analgesic | 1 |
10. A variable name in certain computer language must be either an alphabet or an alphabet followed by a decimal digit. The total number of different variable names that can exist in that language is equal to
a. 280
b. 290
c. 286
d. 296 | 1 |
8. Given that the drawn ball from $\mathrm{U}_{2}$ is white, the probability that head appeared on the coin is
a. $\frac{17}{23}$
b. $\frac{11}{23}$
c. $\frac{15}{23}$
d. $\frac{12}{23}$ | 1 |
- Example 14 A liquid of density $\rho$ is filled upto a height of ' $h$ ' in a container as shown in figure. Base of the container is a square of side L. Ignoring the atmospheric pressure find
 $\frac{a}{b}$
(b) $\frac{b}{a}$
(c) $\frac{a^{2}}{b^{2}}$
(d) $\frac{b^{2}}{a^{2}}$ | 1 |
32. Among the following compounds, the strongest acid is
(a) $\mathrm{HC} \equiv \mathrm{CH}$
(b) $\mathrm{C}_{6} \mathrm{H}_{6}$
(c) $\mathrm{C}_{2} \mathrm{H}_{6}$
(d) $\mathrm{CH}_{3} \mathrm{OH}$ | 1 |
What is a suitable solar panel installation provider near San Francisco, California CA? | 0 |
Example 6.69 Prove that
$$
\frac{1}{n+1}=\frac{{ }^{n} C_{1}}{2}-\frac{2\left({ }^{n} C_{2}\right)}{3}+\frac{3\left({ }^{n} C_{3}\right)}{4}-+(-1)^{n+1} \frac{n\left({ }^{n} C_{n}\right)}{n+1}
$$ | 1 |
What transformed a large part of the world in the 19th century? | 0 |
How can I build a 99.9999% uptime system? I have 40 servers, data base, deployment machine; mirrored to a second data center. How do I handle patches? | 0 |
1.116. A train of mass $m=2000$ tons moves in the latitude $\varphi=$ $=60^{\circ}$ North. Find:
(a) the magnitude and direction of the lateral force that the train exerts on the rails if it moves along a meridian with a velocity $v=$ $=54 \mathrm{~km}$ per hour;
(b) in what direction and with what velocity the train... | 1 |
What is a 360° video or a photo? | 0 |
19. When a $p$-n junction is reverse-biased, the current becomes almost constant at $25 \mu \mathrm{A}$. When it is forwardbiased at $200 \mathrm{mV}$, a current of $75 \mu \mathrm{A}$ is obtained. Find the magnitude of diffusion current when the diode is
(a) unbiased, (b) reverse-biased at $200 \mathrm{mV}$ and (c) fo... | 1 |
How would be the economic relationship between India and America if Hillary Clinton becomes the President? | 0 |
Is female masturbation harmful? Does it has any effect on childbirth or fertility? | 0 |
11. The heats of combustion of carbon and carbon monoxide are -393.5 and $-283.5 \mathrm{~kJ} \mathrm{~mol}^{-1}$, respectively. The heat of formation (in $\mathrm{kJ}$ ) of carbon monoxide per mole is :
(a) -676.5
(b) -110.5
(c) 110.5
(d) 676.5 | 1 |
15. If $\left|\begin{array}{ccc}y z-x^{2} & z x-y^{2} & x y-z^{2} \\ x z-y^{2} & x y-z^{2} & y z-x^{2} \\ x y-z^{2} & y z-x^{2} & z x-y^{2}\end{array}\right|=\left|\begin{array}{ccc}r^{2} & u^{2} & u^{2} \\ u^{2} & r^{2} & u^{2} \\ u^{2} & u^{2} & r^{2}\end{array}\right|$, then
a. $r^{2}=x+y+z$
b. $r^{2}=x^{2}+y^{2}+z^... | 1 |
Which form of writing extended before the invention of paper? | 0 |
22. A sonometer wire supports a $4 \mathrm{~kg}$ load and vibrates in fundamental mode with a tuning fork of frequency $416 \mathrm{~Hz}$. The length of the wire between the bridges is now doubled. In order to maintain fundamental mode, the load should be changed to
(a) $1 \mathrm{~kg}$
(b) $2 \mathrm{~kg}$
(c) $8 \mat... | 1 |
What book did Will Eisner create in 1985? | 0 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.