Question stringlengths 2 3k | label int64 0 1 |
|---|---|
Ex. 27. An organic compound $(\mathrm{A})(\mathrm{mol} . \mathrm{wt} .=59)$ contains $\mathrm{C}(40 \cdot 68 \%), \mathrm{H}(8 \cdot 48 \%)$, $\mathrm{N}(23 \cdot 72 \%)$ and $\mathrm{O}(27 \cdot 12 \%)$. (A), on distillation with $\mathrm{P}_{2} \mathrm{O}_{5}$ gave (B) which on hydrolysis gave (C), a monobasic acid o... | 1 |
11. For the reaction $\mathrm{A}+2 \mathrm{~B} \rightarrow \mathrm{C}, 5$ moles of A and 8 moles of B will produce
(a) 5 moles of C
(b) 4 moles of C
(c) 8 moles of C
(d) 13 moles of C | 1 |
39. How many optically active stereoisomers are possible for butane-2, 3diol?
(a) 1
(b) 2
(c) 3
(d) 4 | 1 |
Why do some people currently believe the earth is flat? | 0 |
What are some functions of softpub.dll? | 0 |
How do I cure myself from nightfall? | 0 |
What are some lesser known/interesting facts about Delhi Metro? | 0 |
Can you pass a urine test for meth with Suragel? | 0 |
Can Donald Trump really be disqualified for saying there should be a ban for Muslims entering the US? | 0 |
4.116. There are two oscillating circuits (Fig. 4.31) with capacitors of equal capacitances. How must inductances and active resistances of the coils be interrelated for the frequencies and damping of free oscillations in both circuits to be equal? The mutual inductance of coils in the left circuit is negligible. | 1 |
19. Which one of the following does not have a pyramidal shape?
(a) $\left(\mathrm{CH}_{3}\right)_{3} \mathrm{~N}$
(b) $\left(\mathrm{SiH}_{3}\right)_{3} \mathrm{~N}$
(c) $\mathrm{P}\left(\mathrm{CH}_{3}\right)_{3}$
(d) $\mathrm{P}\left(\mathrm{SiH}_{3}\right)$ | 1 |
Why do you fall in love with your best friend? | 0 |
Parish pump politics is also called what? | 0 |
3. A photon of energy $h v$ is absorbed by a free electron of a metal having work function $\varphi<h v$.
(a) The electron is sure to come out.
(b) The electron is sure to come out with a kinetic energy $h v-\varphi$.
(c) Either the electron does not come out or it comes out with a kinetic energy $h v-\varphi$.
(d)... | 1 |
How do we know if we are making the right decision? | 0 |
How does one deal with analysis paralysis? | 0 |
What is the best alternative energy source? | 0 |
Where can I find a good hacker? | 0 |
What is the term for the degree to which an emotional experience feels positive or negative? | 0 |
What would be the expected price of iPhone 7? | 0 |
Why are AAP followers certain that Arvind Kejriwal's model of governance is better than other models? | 0 |
1. If $f: R \rightarrow R$ is a twice differentiable function such that $f^{\prime \prime}(x)>0$ for all $x \in R$, and $f \frac{1}{2}=\frac{1}{2}, f(1)=1$, then
(a) $f^{\prime}(1) \leq 0$
(c) $0<f^{\prime}(1) \leq \frac{1}{2}$
(b) $f^{\prime}(1)>1$
(d) $\frac{1}{2}<f^{\prime}(1) \leq 1$ | 1 |
What was the Hyderabad region known as under the Chalukya dynasty? | 0 |
What languages does Quora plan to support in the future? | 0 |
How high were the buildings in the Mature Indus ? | 0 |
How does the "Rocky Mountain Low" pressure region affect temperatures in eastern Colorado? | 0 |
16. The cost at 5 paise/ kWh of operating an electric motor for 8 hours which takes 15 amp at 110 V is
(a) Rs 66
(b) 66 paise
(c) 37 paise
(d) Rs $6 \cdot 60$
[Hint: W=I.V] | 1 |
11. The transition element that has lowest enthalpy of atomisation is:
(a) $\mathrm{Fe}$
(b) $\mathrm{Cu}$
(c) $\mathrm{V}$
(d) $\mathrm{Zn}$ | 1 |
3. Which one is a vector quantity?
(a) Time
(b) Temperature
(c) Magnetic flux
(d) Magnetic field intensity | 1 |
5. Two stones are thrown up simultaneously from the edge of a cliff $240 \mathrm{~m}$ high with initial speed of $10 \mathrm{~m} / \mathrm{s}$ and $40 \mathrm{~m} / \mathrm{s}$, respectively. Which of the following graph best represents the time variation of relative position of the second stone with respect to the fir... | 1 |
39. A particle of mass $m$ and charge $q$ is projected into a region having a perpendicular magnetic field $B$. Find the angle of deviation (figure 34-E14) of the particle as it comes out of the magnetic field if the width $d$ of the region is very slightly smaller than
(a) $\frac{m v}{q B}$
(b) $\frac{m v}{2 q B}$
(c)... | 1 |
What are useful social skills that can be picked up quickly? | 0 |
8. Let $0<\theta<\frac{\pi}{2}$. If the eccentricity of the hyperbola $\frac{x^{2}}{\cos ^{2} \theta}-\frac{y^{2}}{\sin ^{2} \theta}=1$ is greater than 2 , then the length of its latus rectum lies in the interval
(2019 Main, 9 Jan I)
(a) $1, \frac{3}{2}$
(b) $(3, \infty)$
(c) $\frac{3}{2}, 2$
(d) $(2,3]$ | 1 |
42. The ground state energy of hydrogen atom is $-13.6 \mathrm{eV}$. Consider an electronic state $\psi$ of $\mathrm{He}^{+}$ whose energy, azimuthal quantum number and magnetic quantum number are $-3.4 \mathrm{eV}, 2$ and 0 , respectively. Which of the following statement(s) is (are) true from the state $\psi$ ?
(... | 1 |
*24 A wire is bent into three circular segments, each of radius $r=$ 10 cm , as shown in Fig. 30-48. Each segment is a quadrant of a circle, $a b$ lying in the $x y$ plane, $b c$ lying in the $y z$ plane, and $c a$ lying in the $z x$ plane. (a) If a uniform magnetic field $\vec{B}$ points in the positive $x$ direction,... | 1 |
After an MSc in mathematics, is it possible to go for an MS in computer science from the top universities of the US? | 0 |
Who took Jerusalem in 1187? | 0 |
5. The radius of the second Bohr orbit, in terms of the Bohr radius, $a_{0}$, in $\mathrm{Li}^{2+}$ is:
(a) $\frac{2 a_{0}}{3}$
(b) $\frac{4 a_{0}}{9}$
(c) $\frac{4 a_{0}}{3}$
(d) $\frac{2 a_{0}}{9}$ | 1 |
69. Give reason(s) why elemental nitrogen exists as a diatomic molecule whereas elemental phosphorus as a tetraatomic molecule. | 1 |
11.
$$
\left[\begin{array}{ccc}
1 & 0 & 0 \\
0 & \cos \alpha & \sin \alpha \\
0 & \sin \alpha & -\cos \alpha
\end{array}\right]
$$ | 1 |
73. Spherical rain drop evaporates at a rate proportional to its surface area. The differential equation corresponding to the rate of change of the radius of the rain drop if the constant of proportionality is $K>0$ is
a. $\frac{d r}{d t}+K=0$
b. $\frac{d r}{d t}-K=0$
c. $\frac{d r}{d t}=K r$
d. None of these | 1 |
24. Two identical containers $A$ and $B$ with frictionless pistons contain the same ideal gas at the same temperature and the same volume $V$. The mass of the gas in $A$ is $m_{A}$ and that in $B$ is $m_{B}$. The gas in each cylinder is now allowed to expand isothermally to the same final volume $2 V$. The changes in t... | 1 |
7. Hydrogen peroxide oxidises $\left[\mathrm{Fe}(\mathrm{CN})_{6}\right]^{4-}$ to $\left[\mathrm{Fe}(\mathrm{CN})_{6}\right]^{3-}$ in acidic medium but reduces $\left[\mathrm{Fe}(\mathrm{CN})_{6}\right]^{3-}$ to $\left[\mathrm{Fe}(\mathrm{CN})_{6}\right]^{4-}$ in alkaline medium. The other products formed are respectiv... | 1 |
Why is Donald Trump so obsessed with Mexico? | 0 |
7. Mark the correct statements :
(a) The magnitude of the velocity of a particle is equal to its speed.
(b) The magnitude of average velocity in an interval is equal to its average speed in that interval.
(c) It is possible to have a situation in which the speed of a particle is always zero but the average speed is ... | 1 |
7. $\left[\begin{array}{l}\mathrm{H}_{2}(\mathrm{~g})+\frac{1}{2} \mathrm{O}_{2}(\mathrm{~g})=\mathrm{H}_{2} \mathrm{O}(\mathrm{g}) ; \Delta H=-241 \cdot 8 \mathrm{~kJ} \\ \mathrm{CO}(\mathrm{g})+\frac{1}{2} \mathrm{O}_{2}(\mathrm{~g})=\mathrm{CO}_{2}(\mathrm{~g}) ; \Delta H=-283 \mathrm{~kJ}\end{array}\right]$
The he... | 1 |
What is something that may cause the reception and decoding of the content of a message to be faulty? | 0 |
- 31 SSM WwW The only force acting on a 2.0 kg body as it moves along a positive $x$ axis has an $x$ component $F_{x}=-6 x \mathrm{~N}$, with $x$ in meters. The velocity at $x=3.0 \mathrm{~m}$ is $8.0 \mathrm{~m} / \mathrm{s}$. (a) What is the velocity of the body at $x=4.0 \mathrm{~m}$ ? (b) At what positive value of ... | 1 |
When do colleges start sending admission letters? | 0 |
- 52 For the arrangement of Fig. 25-17, suppose that the battery remains connected while the dielectric slab is being introduced. Calculate (a) the capacitance, (b) the charge on the capacitor plates, (c) the electric field in the gap, and (d) the electric field in the slab, after the slab is in place. | 1 |
6. When a rod moves at a relativistic speed $v$, its mass
(a) must incrase by a factor of $\gamma$
(b) may remain unchanged
(c) may increase by a factor other than $\gamma$
(d) may decrease. | 1 |
What single experience most changed your outlook on life? | 0 |
(ii) -4 | 1 |
5. Number of $\alpha$-hydrogen in given compound are :
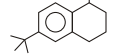
(A) 2
(B) 3
(C) 4
(D) 5 | 1 |
22. Four capacitors are connected as shown in figure to a $30 \mathrm{~V}$ battery. The potential difference between points $a$ and $b$ is
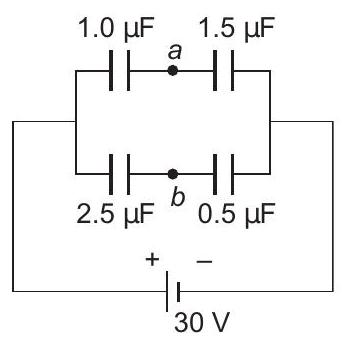
(a) $5 \mathrm{~V}$
(b) $9 \mathrm{~V}$
(c) $10 \m... | 1 |
14. A wire of length 2 units is cut into two parts which are bent respectively to form a square of side $=x$ units and a circle of radius $=r$ units. If the sum of the areas of the square and the circle so formed is minimum, then
(a) $2 x=(\pi+4) r$
(b) $(4-\pi) x=\pi r$
(c) $x=2 r$
(d) $2 x=r$ | 1 |
4. $\int \frac{g(x)}{f(x)} d x$ is equal to
a. $\log \left(e^{x}+e^{-x}\right)+c$
b. $\log \left|e^{x}-e^{-x}\right|+c$
c. $\log \left|e^{2 x}-1\right|+c$
d. None of these | 1 |
12. A mixture of $\mathrm{N}_{2}$ and $\mathrm{H}_{2}$ is caused to react in a closed container to form $\mathrm{NH}_{3}$. The reaction ceases before either reactant has been totally consumed. At this stage, 2.0 moles each of $\mathrm{N}_{2}, \mathrm{H}_{2}$ and $\mathrm{NH}_{3}$ are present. The moles of $\mathrm{N}_{... | 1 |
10. A satellite is revolving round the earth in circular orbit
(a) if mass of earth is made four times, keeping other factors constant, orbital speed of satellite will become two times
(b) corresponding to change in part (a), times period of satellite will remain half
(c) when value of $G$ is made two times orbital ... | 1 |
What are the safety precautions on handling shotguns proposed by the NRA in North Dakota? | 0 |
A book with pictures with no captions that tell a story are called what? | 0 |
16. A battery is connected between two points $A$ and $B$ on the circumference of a uniform conducting ring of radius $r$ and resistance $R$. One of the $\operatorname{arcs} A B$ of the ring subtends an angle $\theta$ at the centre. The value of the magnetic induction at the centre due to the current in the ring is
(... | 1 |
What was the origin of the war? | 0 |
1. The number of real solutions of
$$
\tan ^{-1} \sqrt{x(x+1)}+\sin ^{-1} \sqrt{x^{2}+x+1}=\frac{\pi}{2} \text { is }
$$
(a) zero
(b) one
(c) two
(d) infinite | 1 |
What ways can God interact with the universe? | 0 |
20. If $b>a$, then the equation $(x-a)(x-b)-1=0$ has
a. both roots in $(a, b)$
b. both roots in $(-\infty, a)$
c. both roots in $(b,+\infty)$
d. one root in $(-\infty, a)$ and the other in $(b,+\infty)$
(IIT-JEE, 2000) | 1 |
The first order rate constant for the decomposition of ethyl iodide by the reaction
$$
\mathrm{C}_2 \mathrm{H}_5 \mathrm{I}(\mathrm{~g}) \rightarrow \mathrm{C}_2 \mathrm{H}_4(\mathrm{~g})+\mathrm{HI}(\mathrm{~g})
$$
at 600 K is $1.60 \times 10^{-5} \mathrm{~s}^{-1}$. Its energy of activation is $209 \mathrm{~kJ} / \m... | 1 |
How long will my period last on birth control? | 0 |
What's the best way to make a indie game profitable? | 0 |
What are the differences between torque, moment and couple? | 0 |
2.7 Calculate the wavelength, frequency and wavenumber of a light wave whose period is $2.0 \times 10^{-10} \mathrm{~s}$. | 1 |
2. The following molecule acts as an :
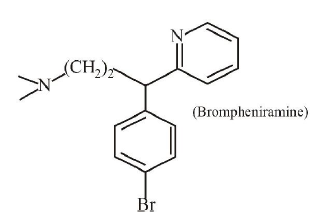
(a) Antiseptic
(b)Anti-depressant
(c) Anti-bacterial
(d) Anti-histamine | 1 |
For Problems 4-6
Let $A$ be the set of all $3 \times 3$ symmetric matrices all of whose entries are either 0 or 1 . Five of these entries are 1 and four of them are 0 .
4. The number of matrices in $A$ is
a. 12
b. 6
c. 9
d. 3
5. The number of matrices $A$ in $A$ for which the system of linear equations
$A\left[\be... | 1 |
What is another benefit of measuring sexuality on two scaless verses just the Kinsey scale? | 0 |
Ex. 29. The density of sodium chloride at $25^{\circ} \mathrm{C}$ is $2.163 \times 10^{3} \mathrm{~kg} \mathrm{~m}^{-3}$. When X -rays from a palladium target having a wavelength of 58.1 pm are used, the (200) reflection of sodium chloride occurs at an angle of $5.90^{\circ}$. How many $\mathrm{Na}^{+}$and $\mathrm{Cl}... | 1 |
Is virtues such as compassion and tolerance is no acceptable, as the past have years show? | 0 |
Give an example of why it's "sometimes better to forget what's legal and do what's right?" | 0 |
What distinction does the city of Thessaloniki have in regards to size in Greece? | 0 |
Ex. 48. A 0.2 m aqueous solution of KCl freezes at $-0.68^{\circ} \mathrm{C}$. Calculate ' $i$ ' and the osmotic pressure at $0^{\circ} \mathrm{C}$. Assume the volume of solution to be that of pure $\mathrm{H}_{2} \mathrm{O}$ and $\mathrm{K}_{f}$ for $\mathrm{H}_{2} \mathrm{O}$ is 1.86 . | 1 |
What is one called that believes he has the one religion, but that the others aren't necessarily wrong? | 0 |
Through what time period did the Berlin Blockade last? | 0 |
How do cats purr? | 0 |
3. Pick the correct statements :
(a) Average speed of a particle in a given time is never less than the magnitude of the average velocity.
(b) It is possible to have a situation in which $\frac{d \vec{v}}{d t} \neq 0$ but $\frac{d}{d t} \vec{v}=0$
(c) The average velocity of a particle is zero in a time interval. It... | 1 |
Which environmental preserve in Hyderabad is known to be a home for migratory birds? | 0 |
What are the best small classes for freshmen at Western Connecticut State? | 0 |
What would gravity be like in the center of the earth? | 0 |
Example 3.4. Solve $16^{\sin ^{2} x}+16^{\cos ^{2} x}=10,0 \leq x<2 \pi$. | 1 |
What does it feel like to be high on caffeine? | 0 |
What is the best Android app for learning Android programming? | 0 |
4. A ring of mass $M$ and radius $R$ is rotating with angular speed $\omega$ about a fixed vertical axis passing through its centre $O$ with two point masses each of mass $\frac{M}{8}$ at rest at $O$. These masses can move radially outwards along two massless rods fixed on the ring as shown in the figure.
$
b. $(1 / 7,2,1 / 9)$
c. $(1,3,-4)$
d. $(1 / 2,1,1 / 3)$ | 1 |
33. The correct order of reactivity of $\mathrm{PhMgBr}$ with
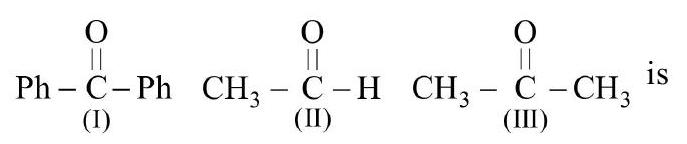
(a) (I) $>$ (II) $>$ (III)
(b) (III) $>$ (II) $>$ (I)
(c) (II) $>$ (III) $>$ (I)
(d) (I) $>$ (III) $>$ (II) | 1 |
23. A force $\mathbf{F}=(3 t \hat{\mathbf{i}}+5 \hat{\mathbf{j}}) \mathrm{N}$ acts on a body due to which its displacement varies as $\mathbf{S}=\left(2 t^{2} \hat{\mathbf{i}}-5 \hat{\mathbf{j}}\right) \mathrm{m}$. Work done by this force in 2 second is
(a) $32 \mathrm{~J}$
(b) $24 \mathrm{~J}$
(c) $46 \mathrm{~J}$
(d)... | 1 |
2. The average molar mass of chlorine is $35.5 \mathrm{~g} \mathrm{~mol}^{-1}$. The ratio of ${ }^{35} \mathrm{Cl}$ to ${ }^{37} \mathrm{Cl}$ in naturally occrring chlorine is close to:
(a) $4: 1$
(b) $3: 1$
(c) $2: 1$
(d) $1: 1$ | 1 |
20. If a polythene sample contains two monodisperse fractions in the ratio $2: 3$ with degree of polymerization 100 and 200, respectively, then its weight average molecular weight will be :
[Main Online April 9, 2013]
(a) 4900
(b) 4600
(c) 4300
(d) 5200 | 1 |
15. The chords of contact of the pair of tangents drawn from each point on the line $2 x+y=4$ to circle $x^{2}+y^{2}=1$ pass through point $\qquad$ .
(IIT-JEE, 1997) | 1 |
Can we create GPS tracking device at home in India? What kind of expertise is needed for the same? | 0 |
36. In the reaction $p$-chlorotoluene with $\mathrm{KNH}_{2}$ in liq. $\mathrm{NH}_{3}$, the major product is:
(a) $o$-toluidine
(b) $m$-toluidine
(c) $p$-toluidine
(d) $p$-chloroaniline. | 1 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.