Question stringlengths 2 3k | label int64 0 1 |
|---|---|
What well known socialist adopted the usage of this term in Germany? | 0 |
40. The gas-phase decomposition of NOBr is second order in [NOBr], with $k=0.81 \mathrm{M}^{-1} \cdot \mathrm{~s}^{-1}$ at $10^{\circ} \mathrm{C}$. Initial concentration of NOBr in the flask at $10^{\circ} \mathrm{C}$ $=4.00 \times 10^{-3} \mathrm{M}$. In how many seconds does it take up $1.50 \times 10^{-3} \mathrm{M}... | 1 |
7. In the following reaction,
$$
\mathrm{CH}_{3} \mathrm{NH}_{2}+\mathrm{CHCl}_{3}+\mathrm{KOH}
$$
$\rightarrow$ Nitrogen containing compound $+\mathrm{KCl}+\mathrm{H}_{2} \mathrm{O}$.
The nitrogen containing compound is
(a) $\mathrm{CH}_{3}-\mathrm{NH}-\mathrm{CH}_{3}$
(b) $\mathrm{CH}_{3}-\mathrm{C} \equiv \math... | 1 |
4. I represents intensity of sound wave, $A$ the amplitude and $r$ the distance from the source. Then, match the following two columns.
| Column I | Column II |
| :--- | :--- |
| (a) Intensity due to a point source. | (p) proportional to $r^{-1 / 2}$ |
| (b) Amplitude due to a point source. | (q) proportional to $r^{-... | 1 |
What are North Korea and China examples of? | 0 |
7. A bullet of mass $m$ and charge $Q$ is fired towards a solid uniformly charged sphere of radius $R$ and total charge $Q$. If it strikes the surface of sphere with speed $v$, the minimum speed $v$ so that it can penetrate through the sphere :
(a) $\frac{Q}{\sqrt{2 \pi \epsilon_{0} m R}}$
(b) $\frac{Q}{\sqrt{6 \pi \ep... | 1 |
37. Which of the following is paramagnetic?
(a) $\mathrm{O}_{2}^{-}$
(b) $\mathrm{CN}^{-}$
(c) $\mathrm{CO}$
(d) $\mathrm{NO}^{+}$ | 1 |
Can a persons sexual interest change over different times in their life? | 0 |
What is the recommendation movie? | 0 |
26. The density of solid AgCl is $5.56 \mathrm{~g} / \mathrm{cc}$. The solid is made up of a cubic array of alternate $\mathrm{Ag}^{+}$and $\mathrm{Cl}^{-}$ions at a spacing of $2.773 \AA$ between centres. From these data calculate the Avogadro constant.
$$
\left(6.04 \times 10^{23}\right)
$$ | 1 |
Who guides the students in a formal environment? | 0 |
5. It was observed that after some time emission of photoelectrons from the sphere stopped. Charge on the sphere when the photon emission stops is
(a) $16 \pi \varepsilon_{0} r$ coulomb
(b) $8 \pi \varepsilon_{0} r$ coulomb
(c) $15 \pi \varepsilon_{0} r$ coulomb
(d) $20 \pi \varepsilon_{0} r$ coulomb | 1 |
14. Positive deviation from ideal behaviour takes place because of
(a) Molecular interaction between atoms and $P V / n R T>1$
(b) Molecular interaction between atoms and $P V / n R T<1$
(c) Finite size of atoms and $P V / n R T>1$
(d) Finite size of atoms and $P V / n R T<1$ | 1 |
How can we get job in abroad? | 0 |
   | 1 |
What entity controls the schools in Hyderabad? | 0 |
15. The rate constant, the activation energy and the Arrhenius parameter of a chemical reaction at $25^{\circ} \mathrm{C}$ are $3.0 \times 10^{-4} \mathrm{~s}^{-1}, 104.4 \mathrm{~kJ} \mathrm{~mol}^{-1}$ and 6.0 $\times 10^{14} \mathrm{~s}^{-1}$ respectively. The value of the rate constant as $T \rightarrow \infty$ is,... | 1 |
18. Which of the following would you expect to be aromatic?
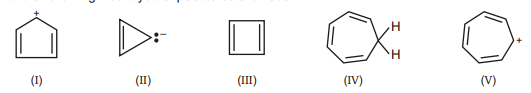
(A) I
(B) II
(C) III
(D) IV
(E) $\mathrm{V}$ | 1 |
What is going to happen to the old denominations of 500 and 1000? | 0 |
Why are all the institutes of national importance in India related to engineering, medical and science? | 0 |
What is the the difference between value and growth stocks? | 0 |
18. A student appears for tests I, II and III. The student is successful if he passes either in tests I and II or tests I and III. The probabilities of the student passing in tests I, II and III are $p, q$ and $\frac{1}{2}$, respectively. If the probability that the student is successful, is $\frac{1}{2}$, then
(a) $p=... | 1 |
What are best ways to refinish wood floors? | 0 |
64. Let $X$ be a set with exactly 5 elements and $Y$ be a set with exactly 7 elements. If $\alpha$ is the number of one-one functions from $X$ to $Y$ and $\beta$ is the number of onto functions from $Y$ to $X$, then the value of $\frac{1}{5 !}(\beta-\alpha)$ is___________. | 1 |
3. Find the angle at which the parabolas $y^{2}=4 x$ and $x^{2}=32 y$ intersect. | 1 |
16. A small source of sound moves on a circle as shown in figure (16-Q1) and an observer is sitting at $O$. Let $v_{1}, \nu_{2}, v_{3}$ be the frequencies heard when the source is at $A, B$ and $C$ respectively.
(a) $v_{1}>v_{2}>v_{3}$
(b) $v_{1}=v_{2}>v_{3}$
(c) $v_{2}>v_{3}>v_{1}$
(d) $v_{1}>v_{3}>v_{2}$
 What maximum height above ground level is reached by... | 1 |
84. $\cos ^{3} x \sin 2 x=\sum_{x=0}^{n} a_{r} \sin (r x) \forall x \in R$, then
a. $n=5, a_{1}=1 / 2$
b. $n=5, a_{1}=1 / 4$
c. $n=5, a_{2}=1 / 8$
d. $n=5, a_{2}=1 / 4$ | 1 |
4. Three charged particles are in equilibrium under their electrostatic forces only. Then,
(a) the particles must be collinear
(b) all the charges cannot have the same magnitude
(c) all the charges cannot have the same sign
(d) the equilibrium is unstable | 1 |
31. The reaction conditions leading to the best yields of $\mathrm{C}_{2} \mathrm{H}_{5} \mathrm{Cl}$ are :
(a) $\mathrm{C}_{2} \mathrm{H}_{6}$ (excess) $+\mathrm{Cl}_{2} \xrightarrow{\text { uv light }}$
(b) $\mathrm{C}_{2} \mathrm{H}_{6}+\mathrm{Cl}_{2} \xrightarrow[\text { room temperature }]{\text { dark }}$
(... | 1 |
What would modern society look like if the Internet was never invented? | 0 |
What are the skills needed to become a system analyst? | 0 |
(xii) But-1-ene to n-butyliodide | 1 |
How many prisoners of war were executed in the sack of Delhi? | 0 |
7. Assertion : At some given instant $I_{1}$ and $I_{2}$ both are $2 \mathrm{~A}$ each. Then, $I$ at this instant should be zero.
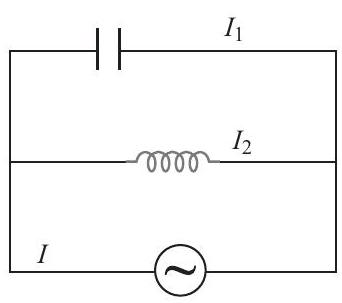
Reason : There is a phase difference of $\pi$ between $I_... | 1 |
The greatest integer is
a. divisible by 7
b. divisible by 11
c. divisible by 9
d. none of these | 1 |
Where on Earth is the highest marine activity recorded? | 0 |
How can I lose weight safely as a teenage girl? | 0 |
What is another name for roundworms? | 0 |
Is it possible to construct a triangle with the side lengths of 7, 8, and 15? | 0 |
4. Two blocks of masses $m_{1}$ and $m_{2}$ are placed in contact with each other on a horizontal platform as shown in figure. The coefficient of friction between $m_{1}$ and platform is $2 \mu$ and that between block $m_{2}$ and platform is $\mu$. The platform moves with an acceleration $a$. The normal reaction betwee... | 1 |
What problems can arise due to translation issues? | 0 |
18. If $z_{1}$ and $z_{2}$ are two complex numbers such that $\left|z_{1}\right|<1<\left|z_{2}\right|$, then prove that $\left|\left(1-z_{1} \bar{z}_{2}\right) /\left(z_{1}-z_{2}\right)\right|<1$. | 1 |
2. Which pair of points lies on the same side of $3 x-8 y$ $-7=0$ ?
a. $(0,-1)$ and $(0,0)$
b. $(4,-3)$ and $(0,1)$
c. $(-3,-4)$ and $(1,2)$
d. $(-1,-1)$ and $(3,7)$ | 1 |
7. A triode is operated in the linear region of its characteristics. If the plate voltage is slightly increased, the dynamic plate resistance will
(a) increase
(b) decrease
(c) remain almost the same
(d) become zero. | 1 |
Example 19 Let R be a relation on the set A of ordered pairs of positive integers defined by $(x, y) \mathrm{R}(u, v)$ if and only if $x v=y u$. Show that R is an equivalence relation. | 1 |
Which IIT is best for M.Tech? | 0 |
18. The equation $x^{3}-\frac{3}{4} x=-\frac{\sqrt{3}}{8}$ is satisfied by
a. $x=\cos \left(\frac{5 \pi}{18}\right)$
b. $x=\cos \left(\frac{7 \pi}{18}\right)$
c. $x=\cos \left(\frac{23 \pi}{18}\right)$
d. $x=\cos \left(\frac{17 \pi}{18}\right)$ | 1 |
5. The slope of the tangent to the curve $y=\sqrt{4-x^{2}}$ at the point, where the ordinate and the abscissa are equal, is
a. -1
b. 1
c. 0
d. None of these | 1 |
55. The nitrogen containing compound produced in the reaction of $\mathrm{HNO}_{3}$ with $\mathrm{P}_{4} \mathrm{O}_{10}$
(a) can also be prepared by reaction of $\mathrm{P}_{4}$ and $\mathrm{HNO}_{3}$
(b) is diamagnetic
(c) contains one $\mathrm{N}-\mathrm{N}$ bond
(d) reacts with Na metal producing a brown gas | 1 |
What were the 3 main causes for the Fall of Rome? | 0 |
If a 90% marginal tax rate was reinstated in the USA with no loopholes, and capital gains were taxed at the same rate, what would happen to the US economy? | 0 |
9. What quantity (in $\mathrm{mL}$ ) of a $45 \%$ acid solution of a mono-protic strong acid must be mixed with a $20 \%$ solution of the same acid to produce $800 \mathrm{~mL}$ of a $29.875 \%$ acid solution?
(a) 320
(b) 325
(c) 316
(d) 330 | 1 |
What actions by the Indian kingdoms gave the European traders the opportunity to acquire lands and influence? | 0 |
Does every state have the same laws? | 0 |
Example 1.46 Solve $|x-1|+|x-2| \geq 4$. | 1 |
14. If a function satisfies $(x-y) f(x+y)-(x+y) f(x-y)=2\left(x^{2} y-y^{3}\right) \forall x, y \in R$ and $f(1)=2$, then
(a) $f(x)$ must be polynomial function
(b) $f(3)=12$
(c) $f(0)=0$
(d) $f(x)$ may not be differentiable | 1 |
What factors did the Young Bengal movement say were needed for civil conduct? | 0 |
32. Pure ammonia is placed in a vessel at a temperature where its dissociation constant $(\alpha)$ is appreciable. At equilibrium:
(a) $K_{p}$ does not change significantly with pressure.
(b) $\alpha$ does not change with pressure.
(c) concentration of $\mathrm{NH}_{3}$ does not change with pressure.
(d) concentr... | 1 |
(c) An applied voltage signal consists of a superposition of a dc voltage and an ac voltage of high frequency. The circuit consists of an inductor and a capacitor in series.
Show that the dc signal will appear across C and the ac signal across L . | 1 |
Example 2.60 Prove that $\cos 18^{\circ}-\sin 18^{\circ}=\sqrt{2} \sin 27^{\circ}$. | 1 |
10. If $a^{2}+b^{2}+c^{2}=1$, then $a b+b c+c a$ lies in the interval
a. $\left[\frac{1}{2}, 2\right]$
b. $[-1,2]$
$$
\text { c. }\left[-\frac{1}{2}, 1\right] \quad \text { d. }\left[-1, \frac{1}{2}\right]
$$
(IIT-JEE, 1984) | 1 |
50. Locus of the image of the point $(2,3)$ in the line $(x-2 y+3)+\lambda(2 x-3 y+4)=0$ is $(\lambda \in R)$
a. $x^{2}+y^{2}-3 x-4 y-4=0$
b. $2 x^{2}+3 y^{2}+2 x+4 y-7=0$
c. $x^{2}+y^{2}-2 x-4 y+4=0$
d. none of these | 1 |
The infant brain contains more of what type of cells in the brain than the adult brain? | 0 |
How is the word 'chronometer' used in a sentence? | 0 |
What's a good processor speed for a laptop? | 0 |
8. A student is performing the experiment of resonance column. The diameter of the column tube is 4 cm . The frequency of the tuning fork is 512 Hz . The air temperature is $38^{\circ} \mathrm{C}$ in which the speed of sound is $336 \mathrm{~m} / \mathrm{s}$. The zero of the meter scale coincides with the top end of th... | 1 |
Who was the head of CBS at the time? | 0 |
Example 4 The distance between two point sources of light is $24 \mathrm{~cm}$. Find out where would you place a converging lens of focal length $9 \mathrm{~cm}$, so that the images of both the sources are formed at the same point. | 1 |
13. A water sample has ppm level concentration of following anions $\mathrm{F}^{-}=10 ; \mathrm{SO}_{4}^{-}=100 ; \mathrm{NO}_{3}=50$
the anion/anions that make/makes the water sample unsuitable for drinking is/are :
(a) only $\mathrm{NO}_{3}$
(b) both $\mathrm{SO}_{4}^{-}$and $\mathrm{NO}_{3}$
(c) only $\mathrm{... | 1 |
5. A boy's catapult is made of rubber cord which is $42 \mathrm{~cm}$ long, with $6 \mathrm{~mm}$ diameter of cross-section and of negligible mass. The boy keeps a stone weighing $0.02 \mathrm{~kg}$ on it and stretches the cord by $20 \mathrm{~cm}$ by applying a constant force. When released the stone flies off with a ... | 1 |
8. Among the following, the compound that can be most readily sulphonated is
(a) benzene
(b) nitrobenzene
(c) toluene
(d) chlorobenzene | 1 |
What hotel in Allahabad would be safe for unmarried couples, without the harassment of police, hotel staff, and moral police? | 0 |
4. Write the IUPAC name of the following compound :
(A) ethyl-2-(chlorocarbonyl) benzoate
(B) ethyl-2-(chlorocarbonyl) hexanoate
(C) 2-(ethoxycarbonyl) benzoyl chloride
(D) None of the... | 1 |
In what form is the information or content sent in a simple model? | 0 |
1. Two wires $A$ and $B$ are carrying currents $I_{1}$ and $I_{2}$ as shown in the figure. The separation between them is $d$. A third wire $C$ carrying a current $I$ is to be kept parallel to them at a distance $x$ from $A$ such that the net force acting on it is zero. The possible values of $x$ are
 $\mathrm{H}^{+}$in the first step
(b) $\mathrm{Cl}^{+}$in the first step
(c) $\mathrm{OH}^{-}$in the first step
(d) $\mathrm{Cl}^{+}$and $\mathrm{OH}^{-}$in a single step | 1 |
What is the difference between a cell phone a mobile phone and a smartphone? | 0 |
How did South Korea become a developed nation? | 0 |
4.196. Find the mean Poynting vector (S) of a plane electromagnetic wave $\mathbf{E}=\mathbf{E}_{m} \cos (\omega t-\mathbf{k r})$ if the wave propagates in vacuum. | 1 |
4. If radius of earth is increased, without change in its mass, will the length of day increase, decrease or remain same? | 1 |
What was the name of the school of thought that combined theology and philosophy? | 0 |
For each of the following compounds, write a condensed formula and also their bond-line formula.
(a) $\mathrm{HOCH}_2 \mathrm{CH}_2 \mathrm{CH}_2 \mathrm{CH}\left(\mathrm{CH}_3\right) \mathrm{CH}\left(\mathrm{CH}_3\right) \mathrm{CH}_3$
 | 1 |
2. Why it is so much easier to perform interference experiments with a laser than with an ordinary light source? | 1 |
(vi) pentanol. | 1 |
Is there any way how to improve my life? | 0 |
22. What is the correct structure for 5-hydroxy-2-phenyl hexan-3-one?
(A)
(B)
}}{x}$ is
(1991, 2M)
(a) 1
(b) -1
(c) 0
(d) None of these | 1 |
9. The eccentricity of the hyperbola whose length of the latusrectum is equal to 8 and the length of its conjugate axis is equal to half of the distance between its foci, is
(a) $\frac{4}{3}$
(b) $\frac{4}{\sqrt{3}}$
(c) $\frac{2}{\sqrt{3}}$
(d) $\sqrt{3}$ | 1 |
I want to try for VNIT (preferably CS, Mechanical) . How many marks should I aim for in Maharashtra board and JEE Mains to get that branch? | 0 |
62 Suppose 2.00 mol of a diatomic gas is taken reversibly around the cycle shown in the $T$ $S$ diagram of Fig. 20-35, where $S_{1}=6.00 \mathrm{~J} / \mathrm{K}$ and $S_{2}=8.00 \mathrm{~J} / \mathrm{K}$. The molecules do not rotate or oscillate. What is the energy transferred as heat $Q$ for (a) path $1 \rightarrow 2... | 1 |
What should I do in order to improve my digestive system? | 0 |
Where is the English word God derived from? | 0 |
Were you impressed with the way Chris Wallace, moderator of the third Presidential debate performed? | 0 |
What percentage of brain capacity do humans use? | 0 |
What are your favorite high carb and protein recipes for a mass gaining diet? | 0 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.