Question stringlengths 2 3k | label int64 0 1 |
|---|---|
21. If $I_{1}=\int_{-100}^{101} \frac{d x}{\left(5+2 x-2 x^{2}\right)\left(1+e^{2-4 x}\right)}$ and $I_{2}=\int_{-100}^{101} \frac{d x}{5+2 x-2 x^{2}}$, then $\frac{I_{1}}{I_{2}}$ is
a. 2
b. $\frac{1}{2}$
c. 1
d. $-\frac{1}{2}$ | 1 |
The study and following of classical writers was said to be a solution to what issue? | 0 |
3. At a given temperature $\mathrm{T}$, gases $\mathrm{Ne}, \mathrm{Ar}, \mathrm{Xe}$ and $\mathrm{Kr}$ are found to deviate from ideal gas behaviour. Their equation of state is given as
$$
\mathrm{P}=\frac{\mathrm{RT}}{\mathrm{V}-\mathrm{b}} \text { at } \mathrm{T}
$$
Here, $\mathrm{b}$ is the van der Waals constant... | 1 |
When was the Balfour Declaration issued? | 0 |
4. Which of the given is most stable?
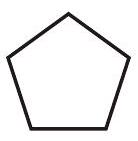
(I)
(II)
=\log _{3+x}\left(x^{2}-1\right)$ is
$$
\begin{aligned}
& \text { a. }(-3,-1) \cup(1, \infty) \\
& \text { b. }[-3,-1) \cup[1, \infty)
\end{aligned}
$$
c. $(-3,-2) \cup(-2,-1) \cup(1, \infty)$
d. $[-3,-2) \cup(-2,-1) \cup[1, \infty)$ | 1 |
27. Highly pure dilute solution of sodium in liquid ammonia
(a) shows blue colour
(b) exhibits electrical conductivity
c) produces sodium amid
(d) produces hydrogen gas. | 1 |
11. The foci of ellipse are
a. $(0, \pm \sqrt{3})$
b. $(0, \pm 2 / 3)$
c. $( \pm \sqrt{3} / 2,0)$
d. none of these | 1 |
What is included in the first part of the critical apparatus? | 0 |
How many openings in the digestive chamber do animals in the Bilateria group have? | 0 |
How to make green beans out of a can? | 0 |
How is life like in New Zealand? | 0 |
85 A golf ball is launched at an angle of $20^{\circ}$ to the horizontal, with a speed of $60 \mathrm{~m} / \mathrm{s}$ and a rotation rate of $90 \mathrm{rad} / \mathrm{s}$. Neglecting air drag, determine the number of revolutions the ball makes by the time it reaches maximum height. | 1 |
Are Indo-Russian relations deteriorating? | 0 |
Which service provider is better: Vodafone or Tata Docomo? | 0 |
12. Four identical capacitors are connected in series with a $10 \mathrm{~V}$ battery as shown in the figure. The point $N$ is earthed. The potentials of points $A$ and $B$ are
(a) $10 \mathrm{~V}, 0 \mathrm{~V}$
(b) $7.5 \mathrm{~V},-2.5 \mathrm{~V}$
(c) $5 \mathrm{~V},-5 \mathrm{~V}$
 values of the half cells $\mathrm{Mg}^{2+} / \mathrm{Mg}$ and $\mathrm{Cl}_{2} / \mathrm{Cl}^{-}$are respectively -2.36 V and +1.36 V . The $E^{0}$ value of the cell $\mathrm{Mg}\left|\mathrm{Mg}^{2+} \| \mathrm{Cl}_{2}\right| \mathrm{Cl}^{-}$is
(a) 3.72 V
(b) 1 V
(c) 0.18 V
(d) 2.64 V | 1 |
(i) 2-Chloro-3-methylpentane | 1 |
How is the method of educational psychology have some relation to medicine and biology? | 0 |
(v) Benzyl chloride to 2-phenylethanamine | 1 |
What are the best episodes of Dragon Ball Z Kai? | 0 |
Is it healthy to eat bread every day? | 0 |
4. A stone is dropped from a height $h$, simultaneously another stone is thrown up from the ground which at a height $4 h$, the two stones cross each other after time
(a) $\sqrt{\frac{h}{2 g}}$
(b) $\sqrt{\frac{h}{8 g}}$
(c ) $\sqrt{8 h g}$
(d) $\sqrt{2 h g}$ | 1 |
62. $\mathrm{H}_{2} \mathrm{SO}_{3}+\mathrm{Cr}_{2} \mathrm{O}_{7}^{2-}+\mathrm{H}^{+}=\mathrm{HSO}_{4}^{-}+\mathrm{Cr}^{3+}+\mathrm{H}_{2} \mathrm{O}$ | 1 |
What cultural event showcases dancers and musicians from all over India? | 0 |
What are the different parts of a thesis? | 0 |
1. A transparent cube of side $d$, made of a material of refractive index $\mu_{2}$, is immersed in a liquid of refractive index $\mu_{1}\left(\mu_{1}<\mu_{2}\right)$. A ray is incident on the face $A B$ at an angle $\theta$ (shown in the figure). Total internal reflection takes place at point $E$ on the face $B C$.
!... | 1 |
70. If the value of the determinant $\left|\begin{array}{lll}a & 1 & 1 \\ 1 & b & 1 \\ (a, b, c>0) \\ 1 & 1 & c\end{array}\right|$ is positive, then
a. $a b c>1$
b. $a b c>-8$
c. $a b c<-8$
d. $a b c>-2$ | 1 |
Example 5.30 How many different numbers of 4 digits can be formed from the digits $0,1,2, \ldots, 9$ if repetition is (i) allowed, (ii) not allowed. | 1 |
When do I know whether or not I should throw food away if it has fungus on it? | 0 |
24. Rate of diffusion of a gas is :
[1985 - 1 Mark
(a) directly proportional to its density.
(b) directly proportional to its molecular weight. (c) directly proportional to the square root of its molecular weight.
(d) inversely proportional to the square root of its molecular weight. | 1 |
Who still could handle the structural calculations for designing at that time? | 0 |
What are some tips to concentrate when reading or at work? I find my mind wanders and I know I am not being as productive as I should be. | 0 |
1. If $\lim _{x \rightarrow 0} \frac{\sqrt{1+x^{2}} \tan \sin \tan ^{-1} x+2 \sqrt{1-x^{2}} \sin \tan ^{-1} \sin ^{-1} x-3 x}{x^{p}}=\mathrm{L}$ then choose the correct option
(a) $p=3, \mathrm{~L}=\frac{-31}{60}$
(b) $p=5, \mathrm{~L}=\frac{-29}{60}$
(c) $p=5, \mathrm{~L}=\frac{-31}{60}$
(d) None of these | 1 |
What type of creature has a ganglion at the back end of the nerve cord as well? | 0 |
What is it like to be a maladaptive daydreamer? | 0 |
4. If $\alpha$ and $\beta$ are the roots of the equation $x^{2}-2 x+2=0$, then the least value of $n$ for which $\frac{\alpha}{\beta}{ }^{n}=1$ is
(a) 2
(b) 5
(2019 Main, 8 April I)
(c) 4
(d) 3 | 1 |
11. Draw most stable resonating structure :
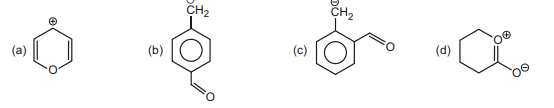 | 1 |
23. A particle is attached to a vertical spring and is pulled down a distance $4 \mathrm{~cm}$ below its equilibrium and is released from rest. The initial upward acceleration is $0.5 \mathrm{~ms}^{-2}$. The angular frequency of oscillation is
(a) $3.53 \mathrm{rad} / \mathrm{s}$
(b) $0.28 \mathrm{rad} / \mathrm{s}$
(c... | 1 |
49. The following reaction is performed at 298 K
$$
2 \mathrm{NO}(\mathrm{~g})+\mathrm{O}_{2}(\mathrm{~g}) \rightleftharpoons 2 \mathrm{NO}_{2}(\mathrm{~g})
$$
The standard free energy of formation of $\mathrm{NO}(\mathrm{g})$ is $86.6 \mathrm{~kJ} \mathrm{~mol}^{-1}$ at 298 K . What is the standard free energy of f... | 1 |
If given a choice, what would be the ONE thing from your past that you would change/remove? | 0 |
7. $\sec ^{-1}\left(\frac{2}{\sqrt{3}}\right) \quad$ | 1 |
3. A ball is thrown vertically up (taken as $+Z$-axis) from the ground. The correct momentum-height $(p-h)$ diagram is
(a)
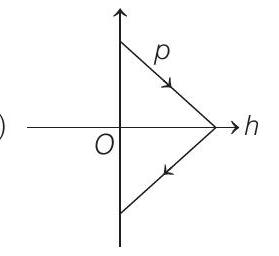
(c)
 } & \text { Self reduction } & \text { (p) } & \text { Lead } \\
\text { (B) } & \text { Carbon reduction } & \text { (q) } & \text { Silver } \\
... | 1 |
What did the 2008 OECD report show the average tourist expenditure while in Greece was? | 0 |
15. If $\mathrm{A}=\left[\begin{array}{rrr}2 & -3 & 5 \\ 3 & 2 & -4 \\ 1 & 1 & -2\end{array}\right]$, find $\mathrm{A}^{-1}$. Using $\mathrm{A}^{-1}$ solve the system of equations
$$
\begin{aligned}
2 x-3 y+5 z & =11 \\
3 x+2 y-4 z & =-5 \\
x+y-2 z & =-3
\end{aligned}
$$ | 1 |
14. In an experiment to determine the acceleration due to gravity $g$, the formula used for the time period of a periodic motion is $T=2 \pi \sqrt{\frac{7(R-r)}{5 g}}$. The values of $R$ and $r$ are measured to be $(60 \pm 1) \mathrm{mm}$ and $(10 \pm 1) \mathrm{mm}$, respectively. In five successive measurements, the ... | 1 |
Along with LeDoux, who was a notable neurological brain researcher? | 0 |
The Peace Pagoda built in New Delhi by the Nipponzan Myohoji contained what type of artifacts? | 0 |
3. Mark the correct options.
(a) The luminous efficiency of a monochromatic source is always greater than that of a white light source of same power.
(b) The luminous efficiency of a monochromatic source of wavelength $555 \mathrm{~nm}$ is always greater than that of a white light source of same power.
(c) The illum... | 1 |
65. The number of solutions of $[\sin x+\cos x]=3+[-\sin x]+[-\cos x]$ (where [.] denotes the greatest integer function), $x \in[0,2 \pi]$, is
a. 0
b. 4
c. infinite
d 1 | 1 |
28. Figure (17-E5) shows three equidistant slits being illuminated by a monochromatic parallel beam of light. Let $B P_{0}-A P_{0}=\lambda / 3$ and $D \gg \lambda$. (a) Show that in this case $d=\sqrt{2 \lambda D / 3}$. (b) Show that the intensity at $P_{0}$ is three times the intensity due to any of the three slits in... | 1 |
The passed Home Rule Bill wasn't implemented because of which war? | 0 |
I study in Ashutosh College in Kolkata. Can I study in any other college in India in my 2nd year? | 0 |
How can I get a perfect score on the new SAT? | 0 |
20. Let $O(0,0), P(3,4), Q(6,0)$ be the vertices of the triangle $O P Q$. The point $R$ inside the triangle $O P Q$ is such that the triangle $O P R, P Q R, O Q R$ are of equal area. The coordinates of $R$ are
a. $(4 / 3,3)$
b. $(3,2 / 3)$
c. $(3,4 / 3)$
d. $(4 / 3,2 / 3)$ | 1 |
What is the best multi disc DVD player? | 0 |
Why should I believe in horoscopes? | 0 |
Example 2.10 Evaluate $\lim _{x \rightarrow 0} \frac{5 \sin x-7 \sin 2 x+3 \sin 3 x}{x^{2} \sin x}$. | 1 |
Who would women rather be: an attractive girl who receives a lot of attention including the unwanted kind, or an average girl who doesn't get much attention? | 0 |
What is shell gas? | 0 |
42. In the depression of freezing point experiment, it is found that the
(a) vapour pressure of the solution is less than that of pure solvent
(b) vapour pressure of the solution is more than that of pure solvent
(c) only solute molecules solidify at the freezing point
(d) only solvent molecules solidify at the f... | 1 |
How can I learn American accent fluently? | 0 |
How can I remove the wage garnishments? | 0 |
10. A cycle pump becomes hot near the nozzle after a few quick strokes even if they are smooth because
(a) the volume of air decreases
(b) the number of air molecules increases
(c) the compression is adiabatic
(d) collision between air particles increases | 1 |
About how many millions of people lived in Europe in the year 1000? | 0 |
What empire was destroyed by the Sack of Constantinople? | 0 |
In what year was Russia declared an empire? | 0 |
What was the Malleus Maleficarum? | 0 |
20. A pendulum clock keeping correct time is taken to high
altitudes,
(a) it will keep correct time
(b) its length should be increased to keep correct time
(c) its length should be decreased to keep correct time
(d) it cannot keep correct time even if the length is
changed. | 1 |
2.229. How does the mean free path $\lambda$ and the number of collisions of each molecule per unit time $v$ depend on the absolute temperature of an ideal gas undergoing
(a) an isochoric process:
(b) an isobaric process?
. | 1 |
What does the overseas development institute want to do? | 0 |
18. The value of the parameter $a$ such that the area bounded by $y=a^{2} x^{2}+a x+1$, coordinate axes and the line $x=1$ attains its least value, is equal to
a. $-\frac{1}{4}$ sq. units
c. $-\frac{3}{4}$ sq. units
d. -1 sq. units
h. $-\frac{1}{2}$ sq. units | 1 |
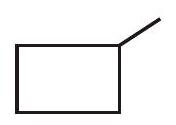
12.15 What is the relationship between the members of following pairs of structures ? Are they structural or geometrical isomers or resonance contributors ?
 | 1 |
21. If $z=\frac{\sqrt{3}}{2}+\frac{i}{2}^{5}+\frac{\sqrt{3}}{2}-\frac{i}{2}^{5}$, then
(a) $\operatorname{Re}(z)=0$
(b) $\operatorname{Im}(z)=0$
(c) $\operatorname{Re}(z)>0, \operatorname{Im}(z)>0$
(d) $\operatorname{Re}(z)>0, \operatorname{Im}(z)<0$
$(1982,2 \mathrm{M})$ | 1 |
Does negative energy exist? If so, what is it? | 0 |
What is the difference between sore throat and STREP throat? What causes strep throat suddenly? | 0 |
40. If in the expansion of $(a-2 b)^{\text {n' }}$, the sum of $5^{\text {lh }}$ and $6^{\text {lh }}$ terms is0, then the values of $a l b=$
a. $\frac{n-4}{5}$
b. $\frac{2(n-4)}{5}$
c. $\frac{5}{n-4}$
d. $\frac{5}{2(n-4)}$ | 1 |
15. The ore that contains both iron and copper is:
(a) copper pyrites
(b) malachite
(c) dolomite
(d) azurite | 1 |
What is best book to read for begginer? | 0 |
Is it "Profit and loss" or "profit or loss" in the income statement? | 0 |
Which is the best QuickBooks Tech Support Number in Texas? | 0 |
What did the Pan American Health organization say these treatments violated? | 0 |
4. A mixture of 2 moles of helium gas (atomic mass $=4 \mathrm{amu}$ ) and 1 mole of argon gas (atomic mass $=$ $40 \mathrm{amu})$ is kept at 300 K in a container. The ratio of the rms speeds $\left(\frac{v_{\text {rms }}(\text { helium })}{v_{\text {rms }}(\text { argon })}\right)$ is
(a) 0.32
(b) 0.45
(c) 2.24
(d... | 1 |
5. Which $\mathrm{C}-\mathrm{N}$ bond having more bond strength.
    in a short time | 1 |
What will be the impact of banning Rs. 500, Rs. 1000 notes on Indian economy and Businesses? | 0 |
12. Which is wrong with respect to our responsibility as a human being to protect our environment
(a) Restricting the use of vehicles
(b) Avoiding the use of floodlighted facilities
(c) Setting up compost tin in gardens.
(d) Using plastic bags. | 1 |
5. The number of values of $r$ satisfying the equation ${ }^{69} C_{3 r-1}-{ }^{69} C_{r^{2}}$ $={ }^{69} C_{r^{2}-1}-{ }^{69} C_{3 r}$ is
a. 1
b. 2
c. 3
d. 7 | 1 |
17. Total number of words that can be formed using all letters of the word 'BRIJESH' that neither begins with 'I' nor ends with ' $\mathrm{B}$ ' is equal to
a. 3720
b. 4920
c. 3600
d. 4800 | 1 |
38. The derivative of $y=(1-x)(2-x) \ldots(n-x)$ at $x=1$ is
a. 0
b. $(-1)(n-1)$ !
c. $n!-1$
d. $(-1)^{n-1}(n-1)$ ! | 1 |
10. Hydrogen peroxide acts both as an oxidising and as a reducing agent depending upon the nature of the reacting species. In which of the following cases $\mathrm{H}_{2} \mathrm{O}_{2}$ acts as a reducing agent in acid medium?
(a) $\mathrm{MnO}_{4}^{-}$
(b) $\mathrm{Cr}_{2} \mathrm{O}_{7}^{2-}$
(c) $\mathrm{SO}_{... | 1 |
2. Five moles of an ideal gas at 1 bar and $298 \mathrm{~K}$ is expanded into vacuum to double the volume. The work done is :
(a) $\mathrm{C}_{\mathrm{v}}\left(\mathrm{T}_{2}-\mathrm{T}_{1}\right)$
(b) $-\mathrm{RT}\left(\mathrm{V}_{2}-\mathrm{V}_{1}\right)$
(c) $-\mathrm{RT} \ln \mathrm{V}_{1} / \mathrm{V}_{1}$
... | 1 |
40. For the following cell
$$
\mathrm{Zn}(\mathrm{~s})\left|\mathrm{ZnSO}_{4}(\mathrm{aq}) \| \mathrm{CuSO}_{4}(\mathrm{aq})\right| \mathrm{Cu}(\mathrm{~s})
$$
when $\left[\mathrm{Zn}^{2+}\right]$ is ten times of $\left[\mathrm{Cu}^{2+}\right]$, the expression $\Delta G\left(\right.$ in $\left.\mathrm{J} \mathrm{mol}... | 1 |
20. The solution of $\frac{d v}{d t}+\frac{k}{m} v=-g$ is
a. $v=c e^{-\frac{k}{m} t}-\frac{m g}{k}$
b. $v=c-\frac{m g}{k} e^{-\frac{k}{m} t}$
c. $v e^{-\frac{k}{m} t}=c-\frac{m g}{k}$
d. $v e^{\frac{k}{m} t}=c-\frac{m g}{k}$ | 1 |
Most of the space in the brain is made up of what structures? | 0 |
Which book will be best for iit? | 0 |
According to you, what is Love? | 0 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.