Question stringlengths 2 3k | label int64 0 1 |
|---|---|
If 1 year back iPhone 5s has upgrade or not? | 0 |
(b) Find the speed of water coming out when half of water has leaked out. | 1 |
When will Apple release the new MacBook Pro in 2016? | 0 |
5. If the equations $4 x^{2}-x-1=0$ and $3 x^{2}+(\lambda+\mu) x+\lambda-\mu=0$ have a root common then the rational values of $\lambda$ and $\mu$ are
a. $\lambda=\frac{-3}{4}$
b. $\lambda=0$
c. $\mu=\frac{3}{4}$
d. $\mu=0$ | 1 |
Who introduced the first major model for communication in 1949? | 0 |
16. If $y=f(x)=\frac{x+2}{x-1}$, then
(1984, 3M)
(a) $x=f(y)$
(b) $f(1)=3$
(c) $y$ increases with $x$ for $x<1$
(d) $f$ is a rational function of $x$ | 1 |
1. When Neils Bohr shook hand with Werner Heisenberg, what kind of force they exerted?
(a) Gravitational
(b) Electromagnetic
(c) Nuclear
(d) Weak. | 1 |
26. A plastic rod has been formed into a circle of radius $R$. It has a positive charge $+Q$ uniformly distributed along one-quarter of its circumference and a negative charge of $-6 Q$ uniformly distributed along the rest of the circumference (figure). With $V=0$ at infinity, what is the electric potential
(a) at the... | 1 |
What is the difference between freedom of whim and will? | 0 |
8. A liquid of density $\rho$ and surface tension $\sigma$ rises in a capillary tube of inner radius $R$. The angle of contact between the liquid and the glass is $\theta$. The point $A$ lies just below the meniscus in the tube and the point $B$ lies at the outside level of liquid in the beaker as shown in figure. The ... | 1 |
What is the best program to refactor DLL files? | 0 |
Which is the best IIT coaching centre in Kolkata? | 0 |
What type of clergy were monks? | 0 |
What was the 2004-05 State Domestic Product of Delhi? | 0 |
116. A bag contains four one-rupee coins, two twenty-five paisa coins and five ten-paisa coins. In how many ways can an amount, not less than Re 1 be taken out from the bag? (Consider coins of the same denominations to be identical.)
a. 71
b. 72
c. 73
d. 80 | 1 |
Will the launch of Google Pixel affect the Apple iPhone 7 market? | 0 |
Who is allowed to use the car park at Google? | 0 |
Along with light cavalry, what military arm was used with increasing frequency in the Late Middle Ages? | 0 |
What was the range of the impact of the new accounting rules on the Greek fiscal figures for the years 1997 to 1999? | 0 |
7.58 The solubility of $\mathrm{Sr}(\mathrm{OH})_{2}$ at 298 K is $19.23 \mathrm{~g} / \mathrm{L}$ of solution. Calculate the concentrations of strontium and hydroxyl ions and the pH of the solution. | 1 |
31. 18 g of glucose $\left(\mathrm{C}_{6} \mathrm{H}_{12} \mathrm{O}_{6}\right)$ is added to 178.2 g of water. The vapour pressure of water (in torr.) for this aqueous solution is
(a) 76.0
(b) 752.4
(c) 759.0
(d) 7.6
(IIT 2016 Main) | 1 |
What are the warning signs of cancer? | 0 |
Has KTM Duke 125 launched in India or is it going to be launched in 2016? | 0 |
86 In Fig. 22-66, particle 1 (of charge +2.00 pC ), particle 2 (of charge -2.00 pC ), and particle 3 (of charge +5.00 pC ) form an equilateral triangle of edge length $a=9.50 \mathrm{~cm}$. (a) Relative to the positive direction of the $x$ axis, determine the direction of the force $\vec{F}_{3}$ on particle 3 due to th... | 1 |
How can I pass IBM C2090-559 exam? | 0 |
20. For a given reaction the logarithm of the concentration of the reactant plotted against time gave a straight line with negative slope. The order of the reaction is
(a) 3
(b) 2
(c) 1
(d) 0 | 1 |
After completing a BTech from normal local colleges, how can I get placed in top companies like Google, Microsoft, Facebook, etc. with good packages? | 0 |
What nationality were the traders that came to archipelago? | 0 |
1
| Column I | Column II |
| :--- | :--- |
| a. Number of straight lines joining any two of 10 <br> points of which four points are collinear | p. 30 |
| b. Maximum number of points of intersection of <br> 10 straight lines in the plane | q. 60 |
| c. Maximum number of points of intersection of <br> six circles in the... | 1 |
Would you drive home after having a glass of wine with dinner? | 0 |
After how many days did is the merit list declared after conducting the SSB interview? | 0 |
18. A professor reads a greeting card received on his 50th birthday with $+2.5 \mathrm{D}$ glasses keeping the card $25 \mathrm{~cm}$ away. Ten years later, he reads his farewell letter with the same glasses but he has to keep the letter $50 \mathrm{~cm}$ away. What power of lens should he now use? | 1 |
5. The major product in the following reaction is :
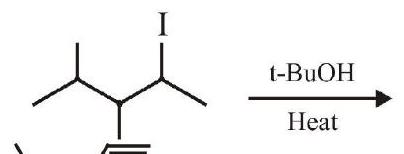
(a)
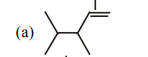
(b)
$. | 1 |
Why is Saltwater taffy candy imported in Switzerland? | 0 |
7. The increasing order of basicity of the following compounds is
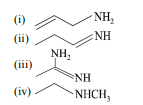
$\mathrm{NH}$
(iv)[^0]
(a) (i) $<$ (ii) $<$ (iii) $<$ (iv)
(b) (ii) $<$ (i) $<$ (iii) $<$ (iv)
(c) (ii) $<$ (i) $<$ (iv) $<$ (iii)
(d) (iv) $<$ (... | 1 |
How is that LPG can be used only on Petrol engine vehicles whereas CNG is being used on both Petrol and Diesel engine vehicles? [By using appropriate conversion kits in both cases? | 0 |
8. A simple pendulum is made of a string of length $l$ and a bob of mass $m$, is released from a small angle $\theta_{0}$. It strikes a block of mass $M$, kept on a horizontal surface at its lowest point of oscillations, elastically. It bounces back and goes up to an angle $\theta_{1}$. Then, $M$ is given by
(a) $m \... | 1 |
Why does my computer not shut down? I use windows 10. | 0 |
How do I rid of dandruff permanently? | 0 |
What was the span of time fro the Ahom Kingdom? | 0 |
11. The acid having $\mathrm{O}-\mathrm{O}$ bond is
(a) $\mathrm{H}_{2} \mathrm{~S}_{2} \mathrm{O}_{3}$
(b) $\mathrm{H}_{2} \mathrm{~S}_{2} \mathrm{O}_{6}$
(c) $\mathrm{H}_{2} \mathrm{~S}_{2} \mathrm{O}_{8}$
(d) $\mathrm{H}_{2} \mathrm{~S}_{4} \mathrm{O}_{6}$ | 1 |
7. Percentage error in the measurement of mass and speed are $2 \%$ and $3 \%$ respectively. The error in the measurement of kinetic energy obtained by measuring mass and speed will be
(a) $12 \%$
(b) $10 \%$
(c) $8 \%$
(d) $5 \%$ | 1 |
Problem 7.8
13.8 g of $\mathrm{N}_{2} \mathrm{O}_{4}$ was placed in a 1L reaction vessel at 400 K and allowed to attain equilibrium
$\mathrm{N}_{2} \mathrm{O}_{4}(\mathrm{~g}) \rightleftharpoons 2 \mathrm{NO}_{2}(\mathrm{~g})$
The total pressure at equilbrium was found to be 9.15 bar. Calculate $K_{c}, K_{p}$ and par... | 1 |
How can I increase the independence of my left and right hands while playing keyboard? | 0 |
35. Figure 2-27 shows a red car and a green car that move toward each other. Figure 2-28 is a graph of their motion, showing the positions $x_{g 0}=270 \mathrm{~m}$ and $x_{r 0}=-35.0 \mathrm{~m}$ at time $t=0$. The green car has a constant speed of $20.0 \mathrm{~m} / \mathrm{s}$ and the red car begins from rest. Wha... | 1 |
5. The correct IUPAC name for the molecule is :
(A) 1-furoic acid
(B) furanyl carboxylic acid
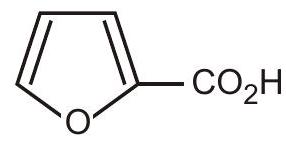
(C) 2-furoic acid
(D) 3-furoic acid | 1 |
Should I got to Purdue or University of Michigan for an undergraduate mechanical engineering program (I'm an international student)? | 0 |
3. | Column I: Functions | Column II: Values of $x$ for which both the functions $5+Y$, <br> in any option of the column I are identical |
| :---: | :---: |
| a. $f(x)=\tan ^{-1}\left(\frac{2 x}{1-x^{2}}\right), g(x)=2 \tan ^{-1} x$ | $\boldsymbol{p} x \in\{-1,1\}$ |
| b $f(x)=\sin ^{-1}(\sin x)$ and $g(x)=\sin \left(\... | 1 |
(a) Show that the work done by the
applied force does not exceed 40 J.
. | 1 |
What replaced agriculture as the focus of European economy with the establishment of overseas colonies? | 0 |
10. There are four machines and it is known that exactly two of them are faulty. They are tested, one by one, in a random order till both the faulty machines are identified. Then the probability that only two tests are needed is
a. $1 / 3$
b. $1 / 6$
c. $1 / 2$
d. $1 / 4$ | 1 |
Which variant of Greek is being used in Alexandros Pallis’ translation of the Iliad? | 0 |
How might the presence of intergalactic dust as an agent for reddening and dimming the light from ever more distant galaxies affect the determination of galactic distances using the redshift associated with the expansion of the cosmos? | 0 |
11. Particle $A$ makes a head on elastic collision with another stationary particle $B$. They fly apart in opposite directions with equal speeds. The mass ratio will be
(a) $\frac{1}{3}$
(b) $\frac{1}{2}$
(c) $\frac{1}{4}$
(d) $\frac{2}{3}$ | 1 |
15. $\mathrm{H}_{2} \mathrm{O}_{2}+\mathrm{KMnO}_{4}=\mathrm{MnO}_{2}+\mathrm{KOH}+\mathrm{O}_{2}+\mathrm{H}_{2} \mathrm{O}$ | 1 |
Who is responsible for the membership fees and expenses? | 0 |
What is the first thing that comes to your mind when you are very happy? | 0 |
Where does most of Greece's renewable energy come from? | 0 |
Fill in the blanks
1. A spherical rain drop evaporates at a rate proportional to its surface area at any instant $t$. The differential equation giving the rate of change of the radius of the rain drop is | 1 |
What does Golden Corral serve for Thanksgiving? How much does their Thanksgiving dinner cost? | 0 |
10. Given a real-valued function $f$ such that
$f(x)=\left\{\begin{array}{cl}\frac{\tan ^{2}\{x\}}{\left(x^{2}-[x]^{2}\right)} & \text { for } x>0 \\ 1 & \text { for } x=0, \text { where }[x] \text { is the integral } \\ \sqrt{\{x\} \cot \{x\}} & \text { for } x<0\end{array}\right.$
part and $\{x\}$ is the fractional... | 1 |
84. (a) Calculate the rate at which body heat is conducted through the clothing of a skier in a steady-state process, given the following data: the body surface area is $1.8 \mathrm{~m}^{2}$, and the clothing is 1.0 cm thick; the skin surface temperature is $33^{\circ} \mathrm{C}$ and the outer surface of the clothing ... | 1 |
How can a recent graduate get a job without experience? | 0 |
Shall we say "many thanks" or "much thanks"? | 0 |
How will the scrapping of Rs 500 and Rs 1000 notes help in reducing black money and corruption? | 0 |
What is the best embarrassing moment of your life? | 0 |
39. Amongst the following elements (whose electronic configurations are given below), the one having the highest ionization energy is :
(a) $[\mathrm{Ne}] 3 s^2 3 p^1$
(b) $[\mathrm{Ne}] 3 s^2 3 p^3$
(c) $[\mathrm{Ne}] 3 s^2 3 p^2$
(d) $[\mathrm{Ne}] 3 d^{10} 4 s^2 4 p^3$ | 1 |
18. When 100 volt DC source is applied across a coil, a current of $1 \mathrm{~A}$ flows through it. When $100 \mathrm{~V}$ AC source of $50 \mathrm{~Hz}$ is applied to the same coil, only 0.5 A current flows. Calculate the inductance of the coil.
(a) $(\pi / \sqrt{3}) \mathrm{H}$
(b) $(\sqrt{3} / \pi) \mathrm{H}$
(c) ... | 1 |
What are some landmarks in Mongolia? | 0 |
How do I make a good PowerPoint presentation? | 0 |
6. The minimum area bounded by the tangent and the coordinate axes is
a. 1
b. $\frac{1}{3}$
c. $\frac{1}{2}$
d. $\frac{1}{4}$ | 1 |
3. If $\lim _{x \rightarrow 1} \frac{x^{4}-1}{x-1}=\lim _{x \rightarrow k} \frac{x^{3}-k^{3}}{x^{2}-k^{2}}$, then $k$ is
(a) $\frac{4}{3}$
(b) $\frac{3}{8}$
(c) $\frac{3}{2}$
(2019 Main, 10 April I) | 1 |
10. Let a three-dimensional vector $\vec{V}$ satisfy the condition, $2 \vec{V}+\vec{V} \times(\hat{i}+2 \hat{j})=2 \hat{i}+\hat{k}$. If $3|\vec{V}|=\sqrt{m}$. Then find the value of $m$. | 1 |
Which career should I choose as per my natal chart? Should it be technical or Administrative field? I am confused and need guidance. | 0 |
Why is China empowering Pakistan? | 0 |
110. Three integers are chosen at random from the set of first 20 natural numbers. The chance that their product is a multiple of 3 is
a. $194 / 285$
b. $1 / 57$
c. $13 / 19$
d. $3 / 4$ | 1 |
16. If $\vec{\alpha}+\vec{\beta}+\vec{\gamma}=a \vec{\delta}$ and $\vec{\beta}+\vec{\gamma}+\vec{\delta}=b \vec{\alpha}, \vec{\alpha}$ and $\vec{\delta}$ are non-collinear, then $\vec{\alpha}+\vec{\beta}+\vec{\gamma}+\vec{\delta}$ equals
a. $a \vec{\alpha}$
b. $b \vec{\delta}$.
c. 0
d. $(a+b) \gamma$ | 1 |
7. A capacitor consists of two stationary plates shaped as a semicircle of radius $R$ and a movable plate made of dielectric with relative permittivity $\in$ and capable of rotating about an axis $O$ between the stationary plates (figure). The thickness of the movable plate is equal to $d$ which is practically the sepa... | 1 |
What are the opinions of the expert when it comes to where the USD to INR exchange rate may get to in next 2 to 3 years time? | 0 |
Is 5 feet 6.5 inches a good enough height for men? | 0 |
How do I get my grandfather's military records in the US? | 0 |
42. At $90^{\circ} \mathrm{C}$, pure water has $\left[\mathrm{H}_{3} \mathrm{O}^{+}\right] 10^{-6} \mathrm{~mole}$ litre ${ }^{-1}$. What is the value of $K_{\mathrm{w}}$ at $90^{\circ} \mathrm{C}$ ?
(a) $10^{-6}$
(b) $10^{-12}$
(c) $10^{-14}$
(d) $10^{-8}$ | 1 |
3.11. A system consists of a thin charged wire ring of radius $R$ and a very long uniformly charged thread oriented along the axis of the ring, with one of its ends coinciding with the centre of the ring. The total charge of the ring is equal to $q$. The charge of the thread (per unit length) is equal to $\lambda$. Fin... | 1 |
15. The compound which reacts fastest with Lucas reagent at room temperature is
(a) butan-1-ol
(b) butan-2-ol
(c) 2-methylpropan-1-ol
(d) 2-methylpropan-2-ol | 1 |
How will Donald Trump benefit India? | 0 |
26. The pair of compounds in which both the compounds give positive test with Tollen's reagent is
(a) Glucose and Sucrose
(b) Fructose and Sucrose
(c) Acetophenone and Hexanal
(d) Glucose and Fructose | 1 |
What did the Vijayanagara Empire promote to unify Indian culture? | 0 |
What do you mean by mechanical engineering? | 0 |
8. A $\operatorname{rod} A B$ moves with a uniform velocity $v$ in a uniform magnetic field as shown in figure (38-Q7).
(a) The rod becomes electrically charged.
(b) The end $A$ becomes positively charged.
(c) The end $B$ becomes positively charged.
(d) The rod becomes hot because of Joule heating.
 d x-x y d y=0(x \neq 0)$ is (where, $C$ is a constant of integration)
(a) $y^{2}-2 x^{2}+C x^{3}=0$
(b) $y^{2}+2 x^{3}+C x^{2}=0$
(c) $y^{2}+2 x^{2}+C x^{3}=0$
(d) $y^{2}-2 x^{3}+C x^{2}=0$ | 1 |
\section*{Problem 3.9}
Are the oxidation state and covalency of Al in $\left[\mathrm{AlCl}\left(\mathrm{H}_{2} \mathrm{O}\right)_{5}\right]^{2+}$ same? | 1 |
What is another term for the Avignon Papacy? | 0 |
Why is change so difficult to implement within an organization? | 0 |
3. The position vectors of the point $A, B, C$ and $D$ are $3 \hat{i}-2 \hat{j}-\hat{k}, 2 \hat{i}+3 \hat{j}-4 \hat{k},-\hat{i}+\hat{j}+2 \hat{k}$ and $4 \hat{i}+5 \hat{j}+\lambda \hat{k}$, respectively. If the points $A, B, C$ and $D$ lie on a plane, find the value of $\lambda$. | 1 |
Is it possible to overdose on marijuana? | 0 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.