Question stringlengths 2 3k | label int64 0 1 |
|---|---|
1. If equation $R(x)=x^{2}+a x+.1$ has two distinct real roots, then exhaustive values of $a$ are
a. $(-2,2)$
b. $(-\infty,-2) \cup(2, \infty)$
c. $(-2, \infty)$
d. all real numbers | 1 |
6. Kjeldahl's method cannot be used to estimate nitrogen for which of the following compounds?
(a) $\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{NH}_{2}$
(b) $\mathrm{CH}_{3} \mathrm{CH}_{2}-\mathrm{C} \cdot \mathrm{N}$
(c) $\mathrm{C}_{6} \mathrm{H}_{5} \mathrm{NO}_{2}$
(d) $\mathrm{NH}_{2}-\stackrel{\text { }}{\mathrm... | 1 |
1 A solid sphere of radius $R$ acquires a terminal velocity $v_{1}$ when falling (due to gravity) through a viscous fluid having a coefficient of viscosity $\eta$. The sphere is broken into 27 identical solid spheres. If each of these spheres acquires a terminal velocity, $v_{2}$ when falling through the same fluid, th... | 1 |
Illustration 2.78 For any four vectors, prove that
$$
(\vec{b} \times \vec{c}) \cdot(\vec{a} \times \vec{d})+(\vec{c} \times \vec{a}) \cdot(\vec{b} \times \vec{d})+(\vec{a} \times \vec{b}) \cdot(\vec{c} \times \vec{d})=0
$$ | 1 |
1. A square is inscribed in the circle $x^{2}+y^{2}-2 x+4 y+3$ $=0$. Its sides are parallel to the coordinate axes. The one vertex of the square is
(IIT-JEE, 1980)
a. $(1+\sqrt{2},-2)$
b. $(1-\sqrt{2},-2)$
c. $(1,-2+\sqrt{2})$
d. None of these | 1 |
3.34 Which of the following statements related to the modern periodic table is incorrect?
(a) The $p$-block has 6 columns, because a maximum of 6 electrons can occupy all the orbitals in a p-shell.
(b) The $d$-block has 8 columns, because a maximum of 8 electrons can occupy all the orbitals in a $d$-subshell.
(c) Ea... | 1 |
How many inches are in 12mm? | 0 |
(ii) Ethane to bromoethene | 1 |
How do the brain structures of dragonflies change as they age? How are these structural changes different from human brains? | 0 |
I went for a blind date with this guy I met online. I told him we will split the tab and he did not insist on paying the tab at all. Is that normal? | 0 |
21. Two capacitors $C_{1}=1 \mu \mathrm{F}$ and $C_{2}=3 \mu \mathrm{F}$ each are charged to a potential difference of $100 \mathrm{~V}$ but with opposite polarity as shown in the figure. When the switch $S$ is closed, the new potential difference between the points $a$ and $b$ is
 $\quad r$ : For every real number $x$, either $x>1$ or $x<1$. | 1 |
2. The equation that represents the water-gas shift reaction is:
(a) $\mathrm{CH}_{4}(\mathrm{~g})+\mathrm{H}_{2} \mathrm{O}(\mathrm{g}) \xrightarrow[\mathrm{Ni}]{1270 \mathrm{~K}} \mathrm{CO}(\mathrm{g})+3 \mathrm{H}_{2}(\mathrm{~g})$
(b) $2 \mathrm{C}(\mathrm{s})+\mathrm{O}_{2}(\mathrm{~g})+4 \mathrm{~N}_{2}(\mathr... | 1 |
6. In YDSE, refer to figure, $d \gg \lambda$ and $q=30^{\circ}$. The central bright fringe is obtained at distance $x$ above point $O$, then $x$ is :
(a) $\frac{D}{2}$
(b) $\frac{D}{\sqrt{3}}$
(c) 2 D
(d) zero
 7
(b) 4
(c) 2
(d) 1 | 1 |
Ex. 40. Calculate the emf of the following cell at $25^{\circ} \mathrm{C}$,
$$
\mathrm{H}_{2}(\mathrm{~g})(1 \mathrm{~atm}) / \mathrm{H}^{+}(\mathrm{aq}) \| \mathrm{OH}^{-}(\mathrm{aq}) / \mathrm{O}_{2}(\mathrm{~g})(1 \mathrm{~atm})
$$
from the following data:
(i) $\mathrm{H}_{2}+\frac{1}{2} \mathrm{O}_{2}=\mathrm{H}... | 1 |
1. The minimum value of $\frac{x^{4}+y^{4}+z^{2}}{x y z}$ for positive real
numbers $x, y, z$ is
a. $\sqrt{2}$
b. $2 \sqrt{2}$
c. $4 \sqrt{2}$
d. $8 \sqrt{2}$ | 1 |
How long have you been playing guitar for and can you solo over standard jazz chords to your satisfaction yet? | 0 |
Why does Amazon mostly use Java? | 0 |
If you had an unlimited amount of money, what would you do with your life? | 0 |
What is the best way to impress a girl? | 0 |
In Lazarus' view, what could the cognitive activity be if it was not conscious? | 0 |
How do plants communicate to warn nearby plants of danger? | 0 |
With latest announcement of making Rs 500 and Rs 1000 notes invalid in India, is this step a checkmate to people who have black money? | 0 |
13. The following is one way of writing the structure of the nitrate ion. Draw others. | 1 |
5. The value of $x+y$ is
a. $\frac{2 a c}{a^{2}-b^{2}}$
b.rac{c^{2}-b^{2}}{a^{2}-b^{2}}$
c. $\frac{c^{2}-b^{2}}{a^{2}+b^{2}}$
d none of these | 1 |
135. The condition that one of the straight lines given by the equation $a x^{2}+2 h x y+b y^{2}=0$ may coincide with one of those given by the equation $a^{\prime} x^{2}+2 h^{\prime} x y+b^{\prime} y^{2}=0$ is
a. $\left(a b^{\prime}-a^{\prime} b\right)^{2}=4\left(h a^{\prime}-h^{\prime} a\right)\left(b h^{\prime}-b^{... | 1 |
Who wrote on the topic of architectural ideals contrasted with simple construction? | 0 |
34. Which pair does not represent a pair of resonance structures?
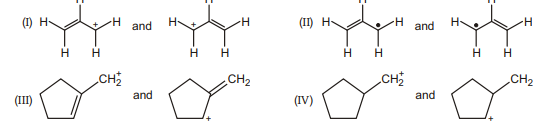
(A) I
(B) II
(C) III
(D) IV
(E) All of these represent pairs of resonance structures. | 1 |
Is gujarat really a developed state? | 0 |
What is base64 encoding? | 0 |
5. Given gas critical temperature/K for
$\mathrm{H}_{2} \mathrm{CH}_{4} \mathrm{CO}_{2} \quad \mathrm{SO}_{2}$ is
$33 \quad 190 \quad 304630$
On the basis of data given above, predict which of the following gases shows least adsorption on a definite amount of charcoal?
(a) $\mathrm{SO}_{2}$
(b) $\mathrm{CH}_{4}$
... | 1 |
What does ASEAN mean? | 0 |
Do anyone know Ritwik Wakankar Of MAMC 2013 batch? | 0 |
What were the most important buildings of the time? | 0 |
What creations brought in the modern industrial economy? | 0 |
22. If 8.3 mL of a sample of $\mathrm{H}_{2} \mathrm{SO}_{4}(36 \mathrm{~N})$ is diluted by 991.7 mL of water, the approximate normality of the resulting solution is
(a) $0 \cdot 4$
(b) 0.2
(c) $0 \cdot 1$
(d) 0.3 | 1 |
What are the odds that Trump will be impeached? | 0 |
12. All the following statements are correct except (for real objects)
(a) the magnification produced by a convex mirror is always less than one
(b) a virtual, erect and same sized image can be obtained using a plane mirror
(c) a virtual, erect, magnified image can be formed using a concave mirror
(d) a real, inver... | 1 |
- Example 19 Light from a discharge tube containing hydrogen atoms falls on the surface of a piece of sodium. The kinetic energy of the fastest photoelectrons emitted from sodium is $0.73 \mathrm{eV}$. The work-function for sodium is $1.82 \mathrm{eV}$. () Find
(a) the energy of the photons causing the photoelectrons ... | 1 |
8. Magnetic field in a cylindrical region of radius $R$ in inward direction is as shown in figure.
(a) an electron will experience no force kept at $(2 R, 0,0)$ if magnetic field increases with time
(b) in the above situation, electron will experience the force in negative $y$-axis
 is/are correct?
(a) If the electric field due to a point charge varies as $r^{-2.5}$ instead of $r^{-2}$, then the Gauss's law will still be valid
(b) The Gauss's law can be used to calculate the field distribution around an electric dipole
(c) If the electric field between tw... | 1 |
11. A physical quantity $A$ is dependent on other four physical quantities $p, q, r$ and $s$ as given by $A=\frac{\sqrt{p q}}{r^{2} s^{3}}$. The percentage error of measurement in $p, q, r$ and $s$ are $1 \%, 3 \%, 0.5 \%$ and $0.33 \%$ respectively, then the maximum percentage error in $A$ is
(a) $2 \%$
(b) $0 \%$
(c)... | 1 |
What fraction of land was under cultivate using the three-field system? | 0 |
Who also profited from Southeast Asia resources? | 0 |
How can I find out if my girlfriend is cheating? | 0 |
3.178. In the circuit shown in Fig. 3.42 the emf of the source is equal to $\mathscr{E}=5.0 \mathrm{~V}$ and the resistances are equal to $R_{1}=4.0 \Omega$ and $R_{2}=6.0 \Omega$. The internal resistance of the source equals $R=$ $=0.10 \Omega$. Find the currents flowing through the resistances $R_{1}$ and $R_{2}$. | 1 |
- $\mathrm{CH}_{3} \mathrm{CH}(\mathrm{OH}) \mathrm{CH}_{3}$
- Functional groups is an alcohol, therefore suffix $=$-ol
- Hydrocarbon structure is an alkane therefore -ane
- The longest continuous chain is $\mathrm{C} 3$ therefore root = prop
- It doesn't matter which end we number from, the alcohol group locant is 2- | 1 |
What does architecture share with other sciences? | 0 |
33. In the formula $X=3 Y Z^{2}, X$ and $Z$ have dimensions of capacitance and magnetic induction, respectively. The dimensions of $Y$ in MKSQ system are_____________. | 1 |
Who would you argue is the greatest all-around athlete of all time? | 0 |
10. The maximum value of $\left(\cos \alpha_{1}\right)\left(\cos \alpha_{2}\right) \cdots\left(\cos \alpha_{n}\right)$, under the restrictions. $0 \leq \alpha_{1}, \alpha_{2}, \ldots, \alpha_{n} \leq \pi / 2$ and $\left(\cot \alpha_{1}\right)\left(\cot \alpha_{2}\right) \cdots\left(\cot \alpha_{n}\right)=1$ is
a. $1 / ... | 1 |
What are the best basic books about fluid power control? | 0 |
How much was George Lucas paid for Rogue One? | 0 |
4. The major product obtained in the photo catalysed bromination of 2methylbutane is:
(a) 1-bromo-2-methylbutane
(b) 1-bromo-3-methylbutane
(c) 2-bromo-3-methylbutane
(d) 2-bromo-2-methylbutane | 1 |
Ex. 36. When $\mathrm{Cl}_{2}$ dissolves in water, it disproportionates, producing $\mathrm{Cl}^{-}$ion and HClO . Find at what $\mathrm{H}_{3} \mathrm{O}^{+}$concentration the potential for the disproportionation changes from a negative value to a positive value, assuming 1.0 atm of pressure and concentration of 1.0 M... | 1 |
What are the job opportunities for Indians in Germany after doing MS in mechanical engineering in Germany itself? | 0 |
3.61. A very long straight thread is oriented at right angles to an infinite conducting plane; its end is separated from the plane by a distance $l$. The thread carries a uniform charge of linear density $\lambda$. Suppose the point $O$ is the trace of the thread on the plane. Find the surface density of the induced ch... | 1 |
How much does a knee replacement surgery cost in Mexico? | 0 |
32. The colour of the $X_{2}$ molecules of group 17 elements changes gradually from yellow to violet down the group. This is due to
(a) The physical state of $X_{2}$ at room temperature changes from gas to solid down the group
(b) Decrease in ionization energy down the group
(c) Decrease in $\pi^{*}-\sigma^{*}$ ga... | 1 |
8. Faraday constant
(a) depends on the amount of the electrolyte
(b) depends on the current in the electrolyte
(c) is a universal constant
(d) depends on the amount of charge passed through the electrolyte. | 1 |
Read the following statement-1(Asseration/Statement) and Statement -2 (Reason/Explanation) and answer as per the options given below :
(a) Statement-1 is True, Statement-2 is True; Statement-2 is a correct explanation for Statement-1.
(b) Statement-1 is True, Statement-2 is True; Statement-2 is NOT a correct explanat... | 1 |
Which two cities benefited greatly from the activities of Crusaders? | 0 |
Which one is the best about men's watch? I need to tell my papa? | 0 |
Where can I watch the new sailor moon redub on my mobile device for free? | 0 |
- Give IUPAC names for the following compounds :
(a)

(c)
(... | 1 |
(a) $\mathrm{P}_4(\mathrm{~s})+\mathrm{OH}^{-}(\mathrm{aq}) \rightarrow \mathrm{PH}_3(\mathrm{~g})+\mathrm{HPO}_2^{-}$(aq) | 1 |
43. The compound $\mathbf{X}$ is
(a) $\mathrm{NaNO}_{3}$
(b) $\mathrm{NaCl}$
(c) $\mathrm{Na}_{2} \mathrm{SO}_{4}$
(d) $\mathrm{Na}_{2} \mathrm{~S}$ | 1 |
11. The pressure of a medium is changed from $1.01 \times 10^{5} \mathrm{~Pa}$ to $1.165 \times 10^{5} \mathrm{~Pa}$ and change in volume is $10 \%$ keeping temperature constant. The bulk modulus of the medium is
(a) $204.8 \times 10^{5} \mathrm{~Pa}$
(b) $102.4 \times 10^{5} \mathrm{~Pa}$
(c) $51.2 \times 10^{5} \math... | 1 |
2. Prove that $\left|\begin{array}{lll}b+c & a-b & a \\ c+a & b-c & b \\ a+b & c-a & c\end{array}\right|=3 a b c-a^{3}-b^{3}-c^{3}$. | 1 |
How long does it take for me to build my credit? | 0 |
7. For any $x, y \in R, x y>0$ then the minimum value of $\frac{2 x}{y^{3}}+\frac{x^{3} y}{3}+\frac{4 y^{2}}{9 x^{4}}$ is. | 1 |
38. In a Young's double slit experiment, the separation between the two slits is $d$ and the wavelength of the light is $\lambda$. The intensity of light falling on slit 1 is four times the intensity of light falling on slit 2 . Choose the correct choice (s).
(a) If $d=\lambda$, the screen will contain only one maximu... | 1 |
What is the difference between wavelengths of sound and light? | 0 |
19. The reaction of propene with $\mathrm{HOCl}\left(\mathrm{Cl}_{2}+\mathrm{H}_{2} \mathrm{O}\right)$ proceeds through the intermediate:
[Main 2016]
(a) $\mathrm{CH}_{3}-\mathrm{CH}(\mathrm{OH})-\mathrm{CH}_{2}^{+}$
(b) $\mathrm{CH}_{3}-\mathrm{CHCl}-\mathrm{CH}_{2}^{+}$
(c) $\mathrm{CH}_{3}-\mathrm{CH}^{+}-\mathr... | 1 |
19. The magnitude of the induced electric field in the orbit at any instant of time during the time interval of the magnetic field change is
(a) $\frac{B R}{4}$
(b) $\frac{-B R}{2}$
(c) $B R$
(d) $2 B R$ | 1 |
What is your definition of Literature? | 0 |
How should I improve my SAT score? | 0 |
Ex. 1. What amount of CaO will be produced by 1 g of calcium? | 1 |
Along with Iceland, Normandy, Ireland and England, what other area did Scandinavians colonize during this period? | 0 |
What are some of the funniest things you've seen? | 0 |
How is uber as a business model in India? Can someone invest in uber as just money making business? | 0 |
What are some lesser-known facts about India Pakistan's partition in 1947? | 0 |
What are the best arguments for or against euthanasia? | 0 |
(ii) Benzene into N, N-dimethylaniline (iii) $\mathrm{Cl}-\left(\mathrm{CH}_{2}\right)_{4}-\mathrm{Cl}$ into hexan-1,6-diamine? | 1 |
If you had 100 billion dollars, what would you do? | 0 |
46. Assertion : Addition of $\mathrm{Br}_{2}$ to 1-butene gives two optical isomers. Reason : The product contains one asymmetric carbon.
[1998 - 2 Marks] | 1 |
What is the most complicated movie you have ever watched? And why? | 0 |
How can we rid the world of religion? | 0 |
Ex. 14. Two elements P and Q form the compounds $\mathrm{P}_{2} \mathrm{Q}_{3}$ and $\mathrm{PQ}_{2}$. If 0.15 mole of $\mathrm{P}_{2} \mathrm{Q}_{3}$ weighs 15.9 g and 0.15 mole of $\mathrm{PQ}_{2}$ weighs 9.3 g , find the atomic weights of P and Q . | 1 |
53. Show that: $2 \cdot 303 \log K=-\frac{\Delta H^0}{R T}+\frac{\Delta S^0}{R}$
[Hint: Use equations 12 and 22 (Ch. 14)] | 1 |
Should I buy Galaxy Note 4 now? | 0 |
What are some theories as to what could've precipitated the Big Bang? | 0 |
10. 32 g of oxygen and 3 g of hydrogen are mixed and kept in a vessel of 760 mm pressure and $0^{\circ} \mathrm{C}$. The total volume occupied by the mixture will be nearly
(a) 22.4 litres
(b) 33.6 litres
(c) 56 litres
(d) 44.8 litres | 1 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.