Question stringlengths 2 3k | label int64 0 1 |
|---|---|
Read the following statement (Assertion) and explanation (Reason) and answer each question as per the options given below :
(a) If both assertion and reason are correct, and reason is the correct explanation of the assertion.
(b) If both assertion and reason are correct, but reason is not the correct explanation of t... | 1 |
| 3 |  | | | | | | 1 |
During what decade was the Lodhi colony built? | 0 |
Why are there more books in the Catholic bible than the Protestant bible? | 0 |
What does it mean if a God is not omnipotent? | 0 |
How do I stop being a material scavenger and start preparing for civil services? | 0 |
14. In a purely resistive $\mathrm{AC}$ circuit,
(a) voltage leads current
(b) voltage lags current
(c) voltage and current are in same phase
(d) nothing can be said | 1 |
King Philip renounced his right to the throne after what treaty? | 0 |
What Did Thomas Edison switch to in 1882? | 0 |
What is the Quora policy on posting photos with answers? | 0 |
Is it worth learning Python if we already know C/C++ and Java? My main aim is to get a Job as software engineer so will knowing python add an advantage? | 0 |
Can you add 5 odd numbers to get 30? | 0 |
19. Two batteries one of the emf $3 \mathrm{~V}$, internal resistance $1 \Omega$ and the other of emf $15 \mathrm{~V}$, internal resistance $2 \Omega$ are connected in series with a resistance $R$ as shown. If the potential difference between points $a$ and $b$ is zero, the resistance $R$ in $\Omega$ is
 ethyne
(b) ethene
(c) benzene
(d) ethane | 1 |
25. A monoatomic ideal gas, initially at temperature $T_{1}$, is enclosed in a cylinder fitted with a frictionless piston. The gas is allowed to expand adiabatically to a temperature $T_{2}$ by releasing the piston suddenly. If $L_{1}$ and $L_{2}$ are the lengths of the gas column before and after expansion respectivel... | 1 |
6. Among the statements (I - IV), the correct ones are :
(I) Be has smaller atomic radius compared to $\mathrm{Mg}$.
(II) Be has higher ionization enthalpy than $\mathrm{Al}$.
(III)Charge/radius ratio of $\mathrm{Be}$ is greater than that of $\mathrm{Al}$.
(IV)Both $\mathrm{Be}$ and $\mathrm{Al}$ form mainly covalent ... | 1 |
Illustration 3.3 $A B C$ is a triangle and $A=(2,3,5)$, $B=(-1,3,2)$ and $C=(\lambda, 5, \mu)$. If the median through $A$ is equally inclined to the axes, then find the value of $\lambda$ and $\mu$. | 1 |
45. Let $f: N \rightarrow N$ defined by $f(x)=x^{2}+x+1, x \in N$, then $f$ is
a. One-one onto
b. Many-one onto
c. One-one but not onto
d. None of these | 1 |
What are suggestions or ideas to improve my English writing skills? | 0 |
2. The redox reaction among the following is:
(a) formation of ozone from atmospheric oxygen in the presence of sunlight
(b) reaction of $\left[\mathrm{Co}\left(\mathrm{H}_{2} \mathrm{O}\right)_{6}\right] \mathrm{Cl}_{3}$ with $\mathrm{AgNO}_{3}$
(c) reaction of $\mathrm{H}_{2} \mathrm{SO}_{4}$ with $\mathrm{NaOH}$
... | 1 |
24. A parallel plate capacitor is made of two circular plates separated by a distance of $5 \mathrm{~mm}$ and with a dielectric of dielectric constant 2.2 between them. When the electric field in the dielectric is $3 \times 10^{4} \mathrm{~V} / \mathrm{m}$, the charge density of the positive plate will be close to
(a) ... | 1 |
(iv) $\{a\} \subset\{a, b, c\}$ | 1 |
39. The best indicator for detection of end point in titration of a weak acid and a strong base is :
(a) methyl orange ( 3 to 4$)$
(b) methyl red (5 to 6 )
(c) bromothymol blue (6 to 7.5 )
(d) phenolphthalein (8 to 9.6 ) | 1 |
Which programming language should I learn? | 0 |
1. 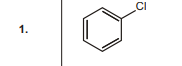 | 1 |
19. There are two identical small holes of area of cross-section $a$ on the opposite sides of a tank containing a liquid of density $\rho$. The difference in height between the holes is $h$. Tank is resting on a smooth horizontal surface. Horizontal force which will has to be applied on the tank to keep it in equilibri... | 1 |
31. Number of non-cyclic isomers of $\mathrm{C}_{4} \mathrm{H}_{8} \mathrm{O}$ are :
(A) 6
(B) 7
(C) 8
(D) 15 | 1 |
Why is the gym busy in June and will it slow down? | 0 |
24. The structure of compound $\mathbf{P}$ is
(a) $\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{CH}_{2} \mathrm{CH}_{2} \mathrm{C} \equiv \mathrm{CH}$
(b) $\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{C} \equiv \mathrm{CCH}_{2} \mathrm{CH}_{3}$
(c)
 | 1 |
20. In the Hofmann bromamide degradation reaction, the number of moles of $\mathrm{NaOH}$ and $\mathrm{Br}_{2}$ used per mole of amine produced are :
(a) Two moles of $\mathrm{NaOH}$ and two moles of $\mathrm{Br}_{2}$.
(b) Four moles of $\mathrm{NaOH}$ and one mole of $\mathrm{Br}_{2}$.
(c) One mole of $\mathrm{Na... | 1 |
40. X mL of $\mathrm{H}_{2}$ gas effuses through a hole in a container in 5 seconds. The time taken for the effusion of the same volume of the gas specified below under identical conditions is
(a) 10 seconds : He
(b) 20 seconds: $\mathrm{O}_{2}$
(c) 25 seconds: CO
(d) 55 seconds: $\mathrm{CO}_{2}$
(IIT 1996) | 1 |
How many people migrated between the newly created countries of India and Pakistan? | 0 |
Can states grant rights to citizens that are not defined by the constitution? | 0 |
4. $\lim _{x \rightarrow 0} \frac{2^{x}-1}{(1+x)^{1 / 2}-1}$ | 1 |
7. Which one is classified as a condensation polymer?
(a) Dacron
(b) Neoprene
(c) Teflon
(d) Acrylonitrile | 1 |
2.38. Draw the approximate plots of isochoric, isobaric, isothermal, and adiabatic processes for the case of an ideal gas, using the following variables:
(a) $p, T$; (b) $V, T$. | 1 |
We're are both "players". Is there a chance we change together? | 0 |
37. How many numbers can be made with the digits $3,4,5,6,7,8$ lying between 3000 and 4000 , which are divisible by 5 while repetition of any digit is not allowed in any number?
a. 60
b. 12
c. 120
d. 24 | 1 |
54. The mirror image of the parabola $y^{2}=4 x$ in the tangent to the parabola at the point $(1,2)$ is
a. $(x-1)^{2}=4(y+1)$
b. $(x+1)^{2}=4(y+1)$
c. $(x+1)^{2}=4(y-1)$
d. $(x-1)^{2}=4(y-1)$ | 1 |
Ex. 32. How much AgCl will be formed by adding 1.70 g of $\mathrm{AgNO}_{3}$ in 200 mL of 5 N HCl solution? $\quad(\mathrm{Ag}=108, \mathrm{~N}=14, \mathrm{O}=16)$ | 1 |
How do you not waste time on the internet? | 0 |
1. Which of the following statements is not true about RNA?
[Main April 12, 2019 (I)]
(a) It controls the synthesis of protein.
(b) It has always double stranded helix structure.
(c) It usually does not replicate.
(d) It is present in the nucleus of the cell. | 1 |
10. The metal that forms nitride by reacting directly with $\mathrm{N}_{2}$ of air, is:
(a) $\mathrm{K}$
(b) $\mathrm{Li}$
(c) $\mathrm{Rb}$
(d) $\mathrm{Cs}$ | 1 |
7. How many structural isomers does $\mathrm{C}_{3} \mathrm{H}_{6} \mathrm{Cl}_{2}$ have?
(A) 2
(B) 3
(C) 4
(D) 5 | 1 |
Example 2.83 If $\cos \theta=\cos \alpha \cos \beta$, prove that $\tan \frac{\theta+\alpha}{2} \tan \frac{\theta-\alpha}{2}=\tan ^{2} \frac{\beta}{2}$. | 1 |
In what region of Western Europe did the pope have influence? | 0 |
What is the ego? | 0 |
32. Number of positional isomers of given compound with 6 membered aromatic ring are :
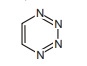
(A) 3
(B) 4
(C) 5
(D) 6 | 1 |
How do I develop a british accent? | 0 |
47. If $f(x)=\left\{\begin{array}{cc}e^{-1 / x^{2}}, & x>0 \\ 0, & x \leq 0\end{array}\right.$, then $f(x)$ is
a. Differentiable at $x=0$
b. Continuous but not differentiable at $x=0$
c. Discontinuous at $x=0$
d. None of these | 1 |
When did Greece go into recession? | 0 |
What are some good books that one must read? | 0 |
Métal hurlant was of what genre? | 0 |
Evaluate the amplitude constant $A$ in Eq. 39-10 for an infinite potential well extending from $x=0$ to $x=L$. | 1 |
Is it possible to make third party API calls from the Parse server side code of my Android app? | 0 |
What are some examples of alternative schools? | 0 |
How do I start a web search from the Yahoo Messenger window? | 0 |
What is common in developed countries? | 0 |
Who are the eleven best bowlers in ODI cricket, as of today? | 0 |
Have you ever been caught while masturbating? | 0 |
18. The range of $f(x)$ is 11
a. $\left[-\frac{\sqrt{3}}{2}, \frac{\sqrt{3}}{2}\right]$
b. $\left[-\frac{\sqrt{5}}{3}, \frac{\sqrt{5}}{3}\right]$
c. $\left[-\frac{\sqrt{5}}{2}, \frac{\sqrt{5}}{2}\right]$
d. None of these | 1 |
Why are some of the comparative graphs shown in shades of the same colour rather than in totally different colours? | 0 |
Which British Prime Minister first supported home rule of Ireland? | 0 |
What dynasty ruled Egypt in this period? | 0 |
4. If $f$ and $g$ are two distinct linear functions defined on $R$ such that they map $[-1,1]$ onto $[0,2]$ and $h: R-\{-1$, $0,1\} \rightarrow R$ defined by $h(x)=\frac{f(x)}{g(x)}$, then show that $\mid h(h(x))$ $+h(h(1 / x)) \mid>2$. | 1 |
What is it like to be an Indian Air Force (IAF) fighter plane pilot? | 0 |
How can I get my love back? | 0 |
What popular uprising occurred in France during this period? | 0 |
I have 90% in 10th grade, 87% in 12th grade and 68% in graduation. How is my profile for any of the IIMs? | 0 |
Which countries began colonizing because they envied the empires of Portugal and Spain? | 0 |
Which city has the largest slum? | 0 |
23. The correct statement with respect to product $Y$ is
a) It gives a positive Tollen's test and is a functional isomer of $X$
(b) It gives a positive Tollen's test and is a geometrical isomer of $X$
(c) It gives a positive iodoform test and is a functional isomer of $X$
(d) It gives a positive iodoform test and i... | 1 |
If Tank battle happens in between India & Pakistan, Will Arjun Tank benefit India? How? | 0 |
What is the best sort of job for a Computer Science student? I have full time hours available and am looking to get the best experience possible. | 0 |
Of the Southeast Asian countries, which country has the largest economy? | 0 |
33. If $\vec{\alpha} \|(\vec{\beta} \times \vec{\gamma})$, then $(\vec{\alpha} \times \vec{\beta}) \cdot(\vec{\alpha} \times \vec{\gamma})$ equals to
a. $|\vec{\alpha}|^{2}(\vec{\beta} \cdot \vec{\gamma})$
b. $|\vec{\beta}|^{2}(\vec{\gamma} \cdot \vec{\alpha})$
c. $|\vec{\gamma}|^{2}(\vec{\alpha} \cdot \vec{\beta})$
d.... | 1 |
How can I take on the Agency of Cadbury? | 0 |
38. The electric potential at a point $(x, y)$ in the $x-y$ plane is given by $V=-k x y$. The field intensity at a distance $r$ in this plane, from the origin is proportional to
(a) $r^{2}$
(b) $r$
(c) $1 / r$
(d) $1 / r^{2}$ | 1 |
What are the best ways to teach kids how to read? | 0 |
Omnipresent gods are a way to watch large groups and enforce what? | 0 |
6. Which bases are strong enough to deprotonate $\mathrm{H}_{2} \mathrm{O}$ ?
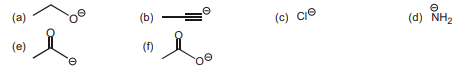
(A) a,b,c,d,e
(B) b,c,d,e,f
(C) a,b,d,e
(D) b,c,d,e | 1 |
20. Which of the following is a set of green house gases?
(a) $\mathrm{CH}_{4}, \mathrm{O}_{3}, \mathrm{~N}_{2}, \mathrm{SO}_{2}$
(b) $\mathrm{O}_{3}, \mathrm{~N}_{2}, \mathrm{CO}_{2}, \mathrm{NO}_{2}$
(c) $\mathrm{O}_{3}, \mathrm{NO}_{2}, \mathrm{SO}_{2}, \mathrm{Cl}_{2}$ | 1 |
32. $\mathrm{AgNO}_{3}(\mathrm{aq})$ was added to an aqueous KCl solution gradually and the conductivity of the solution was measured. The plot of conductance $(G)$ versus the volume of $\mathrm{AgNO}_{3}$ is

[Hint: See Example 6] | 1 |
What is the minimum setup to operate the VIPER12 DIP LED Driver? Which voltage regulator should I use to get 350mA current at 3.2 V? 7805? Or LM317? | 0 |
What's the difference between a tin whistle and a flute? | 0 |
6. Consider the following molecules and statements related to them :
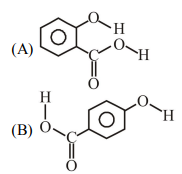
(1) (B) is more likely to be crystalline than (A)
(2) (B) has higher boiling point than (A)
(3) (B) dissolves more readily than (A) in water
Ide... | 1 |
How is the U.S. President elected? | 0 |
5. Two light wires $P$ and $Q$ shown in the figure are made of same material and have radii $r_{P}$ and $r_{Q}$, respectively. The block between them has a mass $m$. When the force $F=\frac{m g}{3}$, then one of the wires breaks. Choose the correct option(s).
(a) $P$ breaks, if $r_{P}=r_{Q}$
(b) $P$ breaks, if $r_{P}... | 1 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.