Question stringlengths 2 3k | label int64 0 1 |
|---|---|
What are benefits of drinking ones own urine? | 0 |
What is the average lifespan of a medical marijuana dispensary? | 0 |
What do you call someone who neither supports nor is opposed to LGBTIQ rights? | 0 |
In what year was the First Crusade proclaimed? | 0 |
Zari is a type of what art that is a major Rajasthani export? | 0 |
48. The equation of the curve is $y=f(x)$. The tangents at $[1$, $f(1)],[2, f(2)]$ and $[3, f(3)]$ make angle $\frac{\pi}{6}, \frac{\pi}{3}$ and $\frac{\pi}{4}$, respectively, with the positive direction of $x$-axis, then the value of $\int_{2}^{3} f^{\prime}(x) f^{\prime \prime}(x) d x+\int_{1}^{3} f^{\prime \prime}(x... | 1 |
Which is the best laptop to buy under INR 50K? | 0 |
8. The order of the reaction $2 \mathrm{~A}+\mathrm{B}+\mathrm{C} \rightarrow$ Product, is found to be 1,2 and 0 w.r.t. $\mathrm{A}, \mathrm{B}$ and C respectively. If the concentration of each reactant is increased by two times, what will be the effect on the rate of the reaction?
[Hint: See solved example 15]
(8 time... | 1 |
What is the minimum amount of people that can be involved in a bribe? | 0 |
43. Two reactions have identical values for energy of activation. Does this ensure that they will have the same rate constant if run at the same temperature? (No, $k \propto \mathrm{~A}$ ) | 1 |
Which is the best refrigerator in 250 litres with 5 star ratings? | 0 |
What are some other ways to say "thank you for your consideration"? | 0 |
1. An aeroplane is to along straight line from A to B , and back again. The relative speed with respect to wind is V . The wind blows perpendicular to line AB with speed $v$. The distance between A and B is $\ell$. The total time for the round trip is:
(a) $\frac{2 \ell}{\sqrt{V^{2}-v^{2}}}$
(b) $\frac{2 v \ell}{V^{2}-... | 1 |
(b) Select three metals that can show disproportionation reaction. | 1 |
11. In a meter bridge, the wire of length 1 m has a non-uniform crosssection such that, the variation $\frac{d R}{d l}$ of its resistance $R$ with length $\ell$ is $\frac{d R}{d l} \propto \frac{\ell}{\sqrt{l}}$. Two equal resistances are connected as shown in the figure. The galvanometer has zero deflection when the j... | 1 |
9. $L, C$ and $R$ represent the physical quantities inductance, capacitance and resistance respectively. Which of the following combinations have dimensions of frequency?
(a) $\frac{1}{R C}$
(b) $\frac{R}{L}$
(c) $\frac{1}{\sqrt{L C}}$
(d) $C / L$. | 1 |
If a bank has to open a new branch then who does it asks for the permission? | 0 |
11. The value of $\frac{\tan ^{2} \frac{\pi}{7}+\tan ^{2} \frac{2 \pi}{7}+\tan ^{2} \frac{3 \pi}{7}}{\cot ^{2} \frac{\pi}{7}+\cot ^{2} \frac{2 \pi}{7}+\cot ^{2} \frac{3 \pi}{7}}$ is
a. 7
b. $35 / 3$
c. $21 / 5$
d. none of these | 1 |
11. The point(s) on the curve $y^{3}+3 x^{2}=12 y$, where the tangent is vertical, is (are)
(a) $\pm \frac{4}{\sqrt{3}},-2$
(b) $\pm \sqrt{\frac{11}{3}}, 0$
(c) $(0,0)$
(d) $\pm \frac{4}{\sqrt{3}}, 2$ | 1 |
When was the Atlantic Charter signed? | 0 |
Where we go after death? | 0 |
19. In an oscillating $L-C$ circuit in which $C=4.00 \mu \mathrm{F}$, the maximum potential difference across the capacitor during the oscillations is $1.50 \mathrm{~V}$ and the maximum current through the inductor is $50.0 \mathrm{~mA}$.
(a) What is the inductance $L$ ?
(b) What is the frequency of the oscillations?... | 1 |
24. Sulfide ores are common for the metals
(a) $\mathrm{Ag}, \mathrm{Cu}$ and $\mathrm{Pb}$
(b) $\mathrm{Ag}, \mathrm{Mg}$ and $\mathrm{Pb}$
(c) $\mathrm{Ag}, \mathrm{Cu}$ and $\mathrm{Sn}$
(d) $\mathrm{Al}, \mathrm{Cu}$ and $\mathrm{Pb}$ | 1 |
What vegetarian recipes are your favourite? | 0 |
Which is the best earphone under Rs 2500? | 0 |
30. If a chemical reaction is at equilibrium, which of the following is not correct?
(a) $\Delta G^{0}=0$
(b) $K_{p}=1$
(c) $K_{c}=1$
(d) $\Delta G^{0}=1$ | 1 |
On what date did Timur beat the Delhi Sultan? | 0 |
26. The $\mathrm{pH}$ of $0.1 \mathrm{M}$ solution of the following salts increases in the order.
(a) $\mathrm{NaCl}<\mathrm{NH}_{4} \mathrm{Cl}<\mathrm{NaCN}<\mathrm{HCl}$
(b) $\mathrm{HCl}<\mathrm{NH}_{4} \mathrm{Cl}<\mathrm{NaCl}<\mathrm{NaCN}$
(c) $\mathrm{NaCN}<\mathrm{NH}_{4} \mathrm{Cl}<\mathrm{NaCl}<\mathrm{... | 1 |
8. If $F(x)$ is continuous at $x=-1$, then
a. $a+b=-2$
b. $a-\dot{b}=3$
c. $a+b=5$
d. None of these | 1 |
1. The figure that is not a direct manifestation of the quantum nature of atoms is :
(a)
(b)
(c)
(b)
 is
(a) $802 \mathrm{~nm}$
(b) $823 \mathrm{~nm}$
(c) $1882 \mathrm{~nm}$
(d) $1648 \mathrm{~nm}$ | 1 |
88 At a certain point, two waves produce pressure variations given by $\Delta p_{1}=\Delta p_{m} \sin \omega t$ and $\Delta p_{2}=\Delta p_{m} \sin (\omega t-\phi)$. At this point,
what is the ratio $\Delta p_{r} / \Delta p_{m}$, where $\Delta p_{r}$ is the pressure amplitude of the resultant wave, if $\phi$ is (a) 0 ,... | 1 |
In what educational hub would one find AIIMS? | 0 |
How many types of triangles are there, along with their conditions? | 0 |
Corruption in what compromises the rule of law? | 0 |
Is it too late for me as 25 years old to go to part-time university program CS course? | 0 |
What was the role of Mary Magdalene in Jesus Christ's life? | 0 |
What is the difference between earthing, grounding and neutral? | 0 |
19. As a wave propagates
(a) the wave intensity remains constant for a plane wave
(b) the wave intensity decreases as the inverse of the distance from the source for a spherical wave
(c) the wave intensity decreases as the inverse square of the distance from the source for a spherical wave
(d) total intensity of th... | 1 |
What is Elastic demand? | 0 |
How is the living condition in SVNIT hostels? | 0 |
Why is the frictional force considered to be a centripetal force? | 0 |
Do you lie ok? | 0 |
56. Let $f(\theta)=\frac{\cot \theta}{1+\cot \theta}$ and $\alpha+\beta=\frac{5 \pi}{4}$, then the value $f(\alpha) f(\beta)$ is
a. $\frac{1}{2}$
b. $-\frac{1}{2}$
c. 2
d none of these | 1 |
What is the average number of tests a college student takes in a year? | 0 |
What are some nice places to visit in Chennai? | 0 |
Non-Disclosure Agreements: For how long is the average NDA in effect? | 0 |
What does a black mole near right ear of a man indicates? | 0 |
16. For positive integers $n_{1}, n_{2}$ the value of expression $(1+i)^{n_{1}}+\left(1+i^{3}\right)^{n_{1}}+\left(1+i^{5}\right)^{n_{2}}+\left(1+i^{7}\right)^{n_{2}}$, here $i=\sqrt{-1}$ is a real number, if and only if
$(1996,2 \mathrm{M})$
(a) $n_{1}=n_{2}+1$
(b) $n_{1}=n_{2}-1$
(c) $n_{1}=n_{2}$
(d) $n_{1}>0, n_{2... | 1 |
What is the meaning of life? | 0 |
4. $\int_{-1}^1 \frac{\sin x-x^2}{3-|x|} d x$ | 1 |
Does it seem like a lot of breakthroughs (in medicine and in energy technology) get very quickly and "conveniently" forgotten shortly after? Is there an organization dedicated to keeping a good record of such findings? Is it ethical? | 0 |
What was the Meija era marked by? | 0 |
When did Crusaders invade Constantinople? | 0 |
17. $p$-T diagram of one mole of an ideal monatomic gas is shown. Processes $A B$ and $C D$ are adiabatic. Work done in the complete cycle is
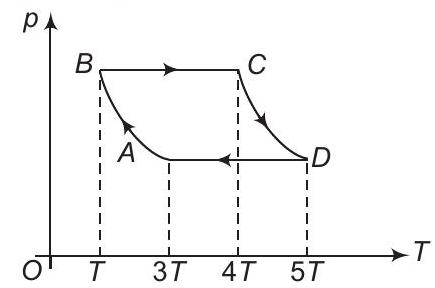
(a) $2.5 R T$
(b) $-2 R T$
(c) $1.5 R T$
(d) $-3... | 1 |
Which startups in India are hiring freshers as Android developers? | 0 |
What obsession can do to someone? | 0 |
How can a person lose a government job in India? | 0 |
How do I become a wildlife photographer? | 0 |
34. The rate constant of a reaction depends on
(a) temperature
(b) initial concentration of the reactants
(c) time of reaction
(d) extent of reaction | 1 |
10. According to molecular orbital theory, which of the following is true with respect to $\mathrm{Li}_{2}^{+}$ and $\mathrm{Li}_{2}^{-}$?
(a) $\mathrm{Li}_{2}^{+}$ is unstable and $\mathrm{Li}_{2}^{-}$ is stable
(b) $\mathrm{Li}_{2}^{+}$ is stable and $\mathrm{Li}_{2}^{-}$ is unstable
(c) B... | 1 |
Which country attacked British Malaya in 1941? | 0 |
How can I train my brain to forget someone? | 0 |
What skills are required to become a good freelancer? | 0 |
How do I get into a really good college? | 0 |
In what country is Kindergarten a term used for primary education? | 0 |
Is there such a job as an army architect, and how do I become one? | 0 |
41. Complete and balance the following chemical reactions :
(iii) $\mathrm{NH}_{3}+\mathrm{NaOCl} \rightarrow \ldots \ldots . .+\ldots \ldots$. | 1 |
The parents of my girlfriend are not allowing me to marry her because she belongs to a "South Indian pure Brahmin" family and I to a "North Indian non Brahmin" family. How do I convince her parents to let us marry? | 0 |
What is the Labour party? | 0 |
In the James-Lange theory, what causes emotions? | 0 |
The level of corruption can vary in richer democracies in regards to the what of those officials making decisions? | 0 |
Who created this group? | 0 |
23. Given that the abundances of isotopes ${ }^{54} \mathrm{Fe},{ }^{56} \mathrm{Fe}$ and ${ }^{57} \mathrm{Fe}$ are $5 \%, 90 \%$ and $5 \%$, respectively, the atomic mass of $\mathrm{Fe}$ is
(a) 55.85
(b) 55.95
(c) 55.75
(d) 56.05 | 1 |
How can Set up Email to a Friend in Magento 2? | 0 |
(c) the net charge appearing on one of the coated surfaces. | 1 |
3. Choose the correct options.
(a) By gamma radiations atomic number is not changed
(b) By gamma radiations mass number is not changed
(c) By the emission of one $\alpha$ and two $\beta$ particles isotopes are produced
(d) By the emission of one $\alpha$ and four $\beta$ particles isobars are produced | 1 |
17. A man running round a race course notes that the sum of the distances of two flagposts from him is always $10 \mathrm{~m}$ and the distance between the flag posts is $8 \mathrm{~m}$. Then the area of the path he encloses in square metres is
a. $15 \pi$
b. $20 \pi$
c. $27 \pi$
d. $30 \pi$ | 1 |
What are some of the best IAS coaching institutions in Delhi? | 0 |
13. $m$-Chlorobromobenzene is an isomer of $m$-bromochlorobenzene. | 1 |
(iii) $\int_{1}^{2} \frac{x d x}{(x+1)(x+2)}$ | 1 |
What were the problems in ancient Egypt? | 0 |
39. A fixed mass $m$ of a gas is subjected to transformation of states from $K$ to $L$ to $M$ to $N$ and back to $K$ as shown in the figure.
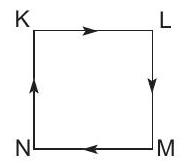
The successive operations that enable this trans... | 1 |
If atheists don't believe in God, but aren't sure he doesn't exist, what do they think caused the universe? | 0 |
How can the heat of combustion of methanol be calculated? | 0 |
How do I stop hating people that I don't even know? | 0 |
10. 0.90 g of a solid organic compound (molecular weight $=90$ ) containing $\mathrm{C}, \mathrm{H}$ and O was heated with oxygen corresponding to a volume of 224 mL at STP. After the combustion the total volume of the gases was 560 mL at STP. On treatment with KOH the volume decreased to 112 mL . Determine the molecul... | 1 |
33. The correct statement(s) about the following sugars $\mathbf{X}$ and $\mathbf{Y}$ is (are)
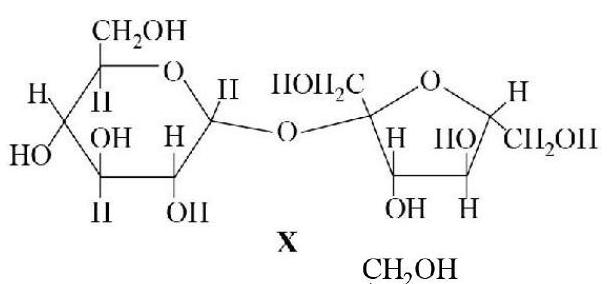
[2009S]
$$
\text { }
$$
(a) $\mathbf{X}$ is a reducing sugar and $\mathbf{Y}$ is a non-reduc... | 1 |
Which religions were well established in the Malay peninsula by the beginning of the 1st century CE? | 0 |
90. The value $\sum_{r=0}^{20} r(20-r)\left({ }^{20} C_{.}\right)^{2}$ is equal to
a. $400{ }^{39} C_{20}$
b. $400{ }^{40} \mathrm{C}_{19}$
c. $400{ }^{39} C_{19}$
d. $400{ }^{38} C_{20}$ | 1 |
Do you feel that reading Quora is a good substitute for reading books? | 0 |
What would wages look like if there was no minimum wage? | 0 |
What was included in this quest for knowledge of the belief system? | 0 |
Catholics: how do I stop watching internet porn? | 0 |
15. Tautomer of which of the following can show geometrical isomerism :
(A) $\mathrm{CH}_{3}-\mathrm{CHO}$
(B) $\mathrm{CH}_{3} \mathrm{CH}_{2}-\mathrm{CHO}$
(C) $\left(\mathrm{CH}_{3}\right)_{2} \mathrm{CH}-\mathrm{CH}=\mathrm{O}$
(D) 
(b)
![](https://cdn.mathpix.com/cropped/2024_06_11_79acd2f562b0071... | 1 |
What part of the area did Jayapala lose? | 0 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.