Question stringlengths 2 3k | label int64 0 1 |
|---|---|
Arabia has a strong influence in which Southeast Asian country? | 0 |
What was the name of the company founded by Hoggart in 1964? | 0 |
What are strategies to make money from fx forwards? | 0 |
How can I clear my KCS exams without coaching centers? | 0 |
Can you suggest names for a new NGO whose objective is to help in guiding parents of children with special needs? Preferably Indian names, but a good English name is also welcome. | 0 |
If local corruption did not exist, there would be enough what to help the people in these countries needing medical help? | 0 |
What Rajasthani national park is known for its tigers? | 0 |
What organization is responsible for awarding Indira Gandhi International Airport as Best Improved Airport in the Asia-Pacific Region? | 0 |
135. You throw a ball from a cliff with an initial velocity of $15.0 \mathrm{~m} / \mathrm{s}$ at an angle of $20.0^{\circ}$ below the horizontal. Find (a) its horizontal displacement and (b) its vertical displacement 2.30 s later. | 1 |
How do I overcome the fear of judgement? | 0 |
What is the principal sense of night-active mammals? | 0 |
In what type of settlement did most Jews live? | 0 |
What causes light to be light? | 0 |
1. The following hydro carbon has an unusually high large dipole moment, explain why
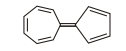 | 1 |
12. Which of the following parameters are the same for all hydrogen-like atoms and ions in their ground states?
(a) Radius of the orbit
(b) Speed of the electron
(c) Energy of the atom
(d) Orbital angular momentum of the electron | 1 |
What can make a person believe they know someone elses sexual orientation? | 0 |
61. If the bond length of $\mathrm{CO}$ bond in carbon monoxide is $1.128 \AA$, then what is the value of $\mathrm{CO}$ bond length in $\mathrm{Fe}(\mathrm{CO})_{5}$ ?
$[2006-5 M,-1]$
(a) $1.15 \AA$
(b) $1.128 \AA$
(c) $1.13 \AA$
(d) $1.118 \AA$ | 1 |
9. A body floats in a liquid contained in a beaker. The whole system as shown in figure falls freely under gravity. The upthrust on the body is
(1982, 3M)
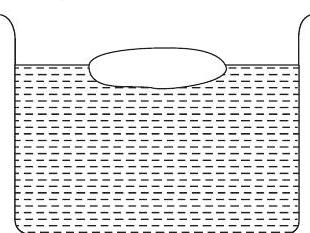
(a) zero
(b) equal to the weig... | 1 |
My guy friend hasn't been texting me for about a week now. Should I panic? | 0 |
Why is it that pigs are considered smarter than dogs? | 0 |
Is the stock market “rigged”? | 0 |
39. Which of the following statements about buffer solutions is wrong?
(a) Weak acids and their salts are better as buffers for $\mathrm{pH}<7$.
(b) Weak bases and their salts are better as buffers for $\mathrm{pH}>7$.
(c) A buffer solution has generally lost its usefulness when one component of the buffer pair is less... | 1 |
What is the best way to make double cream? | 0 |
Where can I get quality support in Sydney for purchasing a property? | 0 |
3. The equation that is incorrect is
(a) $\left(\Lambda_{\mathrm{m}}^{0}\right)_{\mathrm{NaBr}}-\left(\Lambda_{\mathrm{m}}^{0}\right)_{\mathrm{NaCl}}=\left(\Lambda_{\mathrm{m}}^{0}\right)_{\mathrm{KBr}}-\left(\Lambda_{\mathrm{m}}^{0}\right)_{\mathrm{KCl}}$
(b) $\left(\Lambda_{\mathrm{m}}^{0}\right)_{\mathrm{KCl}}-\lef... | 1 |
13. A given quantity of an ideal gas is at pressure $p$ and absolute temperature $T$. The isothermal bulk modulus of the gas is
(1998, 2M)
(a) $\frac{2}{3} p$
(b) $p$
(c) $\frac{3}{2} p$
(d) $2 p$ | 1 |
Illustration 3.32 Find the shortest distance between lines $\vec{r}=(\hat{i}+2 \hat{j}+\hat{k})+\lambda(2 \hat{i}+\hat{j}+2 \hat{k})$ and $\vec{r}=2 \hat{i}-\hat{j}-\hat{k}+\mu(2 \hat{i}+\hat{j}+2 \hat{k})$ | 1 |
Are there people in the U.S. Navy that hide their rank from their families in order to perform special operations missions? | 0 |
What are the advantages of a city using underground cables? | 0 |
(d) its time period. Mass of the earth $=6 \times 10^{24} \mathrm{~kg}$. | 1 |
Along with 860, in what year did Swedish raiders attempt to conquer Constantinople? | 0 |
39. SSM ILW (a) Find the speed of waves on a violin string of mass 800 mg and length 22.0 cm if the fundamental frequency is 920 Hz . (b) What is the tension in the string? For the fundamental, what is the wavelength of (c) the waves on the string and (d) the sound waves emitted by the string? | 1 |
What defines Philosophy education? | 0 |
How do I solve 5x5x5 cube? | 0 |
Why does people here in Quora still ask some questions that has been already answered somewhere else, and can be found easily using Google search? | 0 |
17. Given $\left(1-2 x+5 x^{2}-10 x^{3}\right)(1+x)^{n}=1+a_{1} x+a_{2} x^{2}+\cdots$ and that $a_{1}^{2}=2 a_{2}$ then the value of $n$ is. | 1 |
20. The $K_{\alpha} \mathrm{X}$-ray emission line of tungsten occurs at $\lambda=0.021 \mathrm{~nm}$. The energy difference between $K$ and $L$ levels in this atoms is about
(a) $0.51 \mathrm{MeV}$
(b) $1.2 \mathrm{MeV}$
(c) $59 \mathrm{keV}$
(d) $13.6 \mathrm{eV}$ | 1 |
96. The set of points on the axis of the parabola $(x-1)^{2}$ $=8(y+2)$, from where three distinct normals can be drawn to the parabola is the set $(h, k)$ of points satisfying
a. $h>2$
b. $h>1$
c. $k>2$
d. none of these | 1 |
What event did the American revolution cause? | 0 |
Can the pain in my lower left abdomen be the muscle pain? | 0 |
26. Which one of the following statements is correct?
(a) The balls will stick to the top plate and remain there
(b) The balls will bounce back to the bottom plate carrying the same charge they went up with
(c) The balls will bounce back to the bottom plate carrying the opposite charge they went up with
(d) The bal... | 1 |
How can I find best budget hotels in Haridwar for accommodation in this new year eve? | 0 |
How can I treat a hard pea sized lump under my skin? | 0 |
8. Which one of the following is an example of thermosetting polymers?
[Main Online April 19, 2014]
(a) Neoprene
(b) Buna-N
(c) Nylon 6,6
(d) Bakelite | 1 |
Federal law overrides what laws? | 0 |
What is the Omnipotence paradox? | 0 |
5. A stone is dropped into a quiet lake and waves move in circles at the speed of $5 \mathrm{~cm} / \mathrm{s}$. At the instant when the radius of the circular wave is 8 cm , how fast is the enclosed area increasing? | 1 |
- Identify what suffix you would use in naming each of the following compounds.
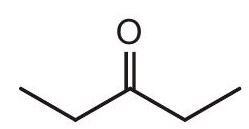
suffix :
$ and $\Delta P(x, t)$ equation in case of longitudinal wave. | 1 |
What are the career options for an electrical and electronics engineer in the IOT field? | 0 |
11. A 2.0 kg particle moves along an $x$ axis, being propelled by a variable force directed along that axis. Its position is given by $x=$ $3.0 \mathrm{~m}+(4.0 \mathrm{~m} / \mathrm{s}) t+c t^{2}-\left(2.0 \mathrm{~m} / \mathrm{s}^{3}\right) t^{3}$, with $x$ in meters and $t$ in seconds. The factor $c$ is a constant.... | 1 |
Who is Katy Perry? | 0 |
Creatures that have a diffuse nerve net are called what? | 0 |
How should I get a girl who rejected me to love me? | 0 |
4.7 What is the effect of temperature on the rate constant of a reaction? How can this effect of temperature on rate constant be represented quantitatively? | 1 |
How do you add a link on Quora? | 0 |
4. The diameter and height of a cylinder are measured by a meter scale to be $12.6 \pm 0.1 \mathrm{~cm}$ and $34.2 \pm 0.1 \mathrm{~cm}$, respectively. What will be the value of its volume in appropriate significant figures?
(a) $4300 \pm 80 \mathrm{~cm}^{3}$
(b) $4260 \pm 80 \mathrm{~cm}^{3}$
(c) $4264.4 \pm 81.0 ... | 1 |
How do I edit my question on Quora when I realize that I misspelled a word? | 0 |
22. The correct match between Item I and Item II is:
\section*{Item I}
(A) Benzaldehyde
(B) Alumina
(C) Acetonitrile
\section*{Item II}
(P) Mobile phase
(Q) Adsorbent
(R) Adsorbate (a) $(\mathrm{A}) \rightarrow(\mathrm{Q}) ;(\mathrm{B}) \rightarrow(\mathrm{P}) ;(\mathrm{C}) \rightarrow(\mathrm{R})$
(b) $(\ma... | 1 |
59. The value of $\sum_{r=0}^{10} r^{10} C_{r} 3^{\prime}(-2)^{10-r}$ is
a. 20
b. 10
c. 300
d. 30 | 1 |
8. If $\vec{a}, \vec{b}, \vec{c}$ are unit vectors such that $\vec{a} \cdot \vec{b}=0=\vec{a} \cdot \vec{c}$ and the angle between $\vec{b}$ and $\vec{c}$ is $\pi / 3$, then the value of $|\vec{a} \times \vec{b}-\vec{a} \times \vec{c}|$ is
a. $1 / 2$
b. 1
c. 2
d. none of these | 1 |
Why has the central government cancelled the Pongal holiday as a government holiday in India? | 0 |
23. (a) An atom initially in an energy level with $E=-6.52 \mathrm{eV}$ absorbs a photon that has wavelength $860 \mathrm{~nm}$. What is the internal energy of the atom after it absorbs the photon? (b) An atom initially in an energy level with $E=-2.68 \mathrm{eV}$ emits a photon that has wavelength $420 \mathrm{~nm}$.... | 1 |
In what region of Hyderabad is Mecca Masjid? | 0 |
16. The value of $\int_{-\pi}^{\pi} \frac{\cos ^{2} x}{1+a^{x}} d x$, where $a>0$, is
a. $\pi$
c. $\pi / 2$
d. $2 \pi$
h. $a \pi$ | 1 |
8. The angle formed by the positive $y$-axis and the tangent to $y=x^{2}+4 x-17$ at $(5 / 2,-3 / 4)$ is
a. $\tan ^{-1}(9)$
b. $\frac{\pi}{2}-\tan ^{-1}(9)$
c. $\frac{\pi}{2}+\tan ^{-1}(9)$
d. None of these | 1 |
41. An object is moving towards a converging lens on its axis. The image is also found to be moving towards the lens. Then, the object distance $u$ must satisfy
(a) $2 f<u<4 f$
(b) $f<u<2 f$
(c) $u>4 f$
(d) $u<f$ | 1 |
26. Which of the following is the correct resonance hybrid of buta-1,3-diene?
(A) $\mathrm{CH}_{2}=\mathrm{CH}=\mathrm{CH}-\mathrm{CH}_{2}$
(B) $\mathrm{CH}_{2}=\mathrm{CH}=\mathrm{CH}=\mathrm{CH}_{2}$
(C) $\mathrm{CH}_{2}=\mathrm{CH}-\mathrm{CH}=\mathrm{CH}_{2}$
(D) None of these | 1 |
What is meant by flow length scale in fluid dynamics? | 0 |
Example 2.30 Consider two complex numbers $\alpha$ and $\beta$ as $\alpha=[(a+b i) /(a-b i)]^{2}+[(a-b i) /(a+b i)]^{2}$, where $a, b \in R$ and $\beta=(z-1) /(z+1)$, where $|z|=1$, then find the correct statement:
a. both $\alpha$ and $\beta$ are purely real
b. both $\alpha$ and $\beta$ are purely imaginary
c. $\al... | 1 |
22. The number of points $f(x)=\left\{\begin{array}{cc}{[\cos \pi x],} & 0 \leq x \leq 1 \\ |2 x-3|[x-2], & 1<x \leq 2\end{array}\right.$ is discontinuous at ([.] denotes the greatest integral function)
a. two points
b. three points
c. four points
d. no points | 1 |
What is the difference between human shampoo and dog shampoo? | 0 |
41. Which one of the following is the smallest in size?
(a) $\mathrm{N}^{3-}$
(b) $\mathrm{O}^{2-}$
(c) $\mathrm{F}^{-}$
(d) $\mathrm{Na}^{+}$ | 1 |
13. The area between the curve $y=2 x^{4}-x^{2}$, the $x$-axis and the ordinates of the two minima of the curve is $\mathrm{p} / \mathrm{q}$, then
(a) $p+q=127$
(b) $p-q=113$
(c) $p<10$
(d) $q>10$ | 1 |
16. The value of $\lim _{x \rightarrow 2} \frac{\sqrt{1+\sqrt{2+x}}-\sqrt{3}}{x-2}$ is
a. $\frac{1}{8 \sqrt{3}}$
b. $\frac{1}{4 \sqrt{3}}$
c. 0
d. None of these | 1 |
What is sure test for electric charge? | 0 |
Example 6.71 Prove that $\frac{C_{1}}{1}-\frac{C_{2}}{2}+\frac{C_{3}}{3}-\frac{C_{4}}{4}+\cdots$
$+\frac{(-1)^{n-1}}{n} C_{n}=1+\frac{1}{2}+\frac{1}{3}+\cdots+\frac{1}{n}$ | 1 |
 | 1 |
7. The INCORRECT statement is
(a) the gemstone, ruby, has $\mathrm{Cr}^{3+}$ ions occupying the octahedral sites of beryl.
(b) the spin-only magnetic moment of $\left[\mathrm{Ni}\left(\mathrm{NH}_{3}\right)_{4}\left(\mathrm{H}_{2} \mathrm{O}\right)_{2}\right]^{2+}$ is $2.83 \mathrm{BM}$
(c) the color of $\left[\ma... | 1 |
How do allergy shots work? | 0 |
How can I fix up "JVM Error" 517 in BlackBerry Bold 9900 ? | 0 |
Should I buy Xiaomi Redmi Note 3? Why? | 0 |
3. Among the statements (A)-(D), the correct ones are:
(A) Lithium has the highest hydration enthalpy among the alkali metals.
(B) Lithium chloride is insoluble in pyridine.
(C) Lithium cannot form ethynide upon its reaction with ethyne.
(D) Both lithium and magnesium react slowly with $\mathrm{H}_{2} \mathrm{O}$
... | 1 |
6. 12 g of Mg will react completely with an acid to give
(a) 1 mole of $\mathrm{O}_{2}$
(b) $\frac{1}{2}$ mole of $\mathrm{H}_{2}$
(c) 1 mole of $\mathrm{H}_{2}$
(d) 2 moles of $\mathrm{H}_{2}$ | 1 |
Example 7.47 Find $\int \sin ^{3} x \cos ^{5} x d x$. | 1 |
Example 3.101 Find the sum of the products of the ten numbers $\pm 1, \pm 2, \pm 3, \pm 4$, and $\pm 5$ taking two at a time. | 1 |
43. 1 mole of an ideal gas at 300 K in thermal contact with surroundings expands isothermally from 1.0 L to 2.0 L against a constant pressure of 3.0 atm . In this process, the change in entropy of surroundings $\left(\Delta S_{s u r}\right)$ in $\mathrm{JK}^{-1}$ is $(1 \mathrm{~L} \mathrm{~atm}=101.3 \mathrm{~J})$
(a)... | 1 |
3. Can $\mathrm{F}^{-}$be oxidised to $\mathrm{F}_{2}$ by any substance listed in the electrochemical series?
(No, but it can be oxidised electrolytically) | 1 |
Chrome does not work in window version 8.1. What is the problem with windows 8? | 0 |
How much money do you make from your Android application? | 0 |
What Indian city is the headquarters of the Northern Railway? | 0 |
4. The value of $x y$ is
a. $\frac{2 a b}{a^{2}-b^{2}}$
b.$\frac{c^{2}-b^{2}}{a^{2}-b^{2}}$
c. $\frac{c^{2}-b^{2}}{a^{2}+b^{2}}$
d none of these | 1 |
Ex 2.52 find the condition if circles whose equations are $x^{2}+y^{2}+c^{2}=2 a x$ and $x^{2}+y^{2}+c^{2}-2 b y=0$ will touch one another externally. | 1 |
Are these guys related? Please help? | 0 |
How many awards did Indira Gandhi International Airport win at the Skytrax World Airport Awards 2015? | 0 |
Who believed that gods are just projections of a person's father? | 0 |
Why do people kill people? | 0 |
2. During an experiment, an ideal gas is found to obey a condition $V p^{2}=$ constant. The gas is initially at a temperature $T$, pressure $p$ and volume $V$. The gas expands to volume $4 V$.
(a) The pressure of gas changes to $\frac{p}{2}$
(b) The temperature of gas changes to $4 T$
(c) The graph of the above proc... | 1 |
What are the properties of pure metals? | 0 |
Which phone is best to buy under 15k? | 0 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.