Question stringlengths 2 3k | label int64 0 1 |
|---|---|
How American presidents get elected? Who will vote for the candidates in the elections? | 0 |
7. In a circuit shown in figure if the internal resistances of the sources are negligible then at what value of resistance $R$ will the thermal power generated in it will be the maximum. What is the value of maximum power?
 increase
(b) decrease
(c) remain unchanged
(d) either increase or decrease. | 1 |
25. All possible values of $\beta$ are
a. $\left(-\infty, \frac{1}{3}\right)$
b. $\left(-\infty,-\frac{1}{3}\right)$
c. $\left(\frac{1}{3}, \infty\right)$
d. $\left(-\frac{1}{3}, \infty\right)$ | 1 |
Add questions on quora? | 0 |
67. Let $z=1-t+i \sqrt{t^{2}+t+2}$, where $t$ is a real parameter. The locus of $z$ in the Argand plane is
a. a hyperbola
b. an ellipse
c. a straight line
d. none of these | 1 |
How do I start business from nothing? | 0 |
If there is a duplicate question, can I post a duplicate answer? | 0 |
81. If $a, b, c$ are non-zeros, then the system of equations
$(\alpha+a) x+\alpha y+\alpha z=0, \alpha x+(\alpha+b) y+\alpha z=0$,
$\alpha x+\alpha y+(\alpha+c) z=0$ has a non-trivial solution if
a. $\alpha^{-1}=-\left(a^{-1}+b^{-1}+c^{-1}\right)$
b. $\alpha^{-1}=a+b+c$
c. $\alpha+a+b+c=1$
d. none of these | 1 |
In what century did written records of peasant life begin to appear? | 0 |
38. The solubility of $\mathrm{A}_2 X_3$ is $y \mathrm{~mol} / \mathrm{dm}^3$. Its solubility product is
(a) $6 y^4$
(b) $64 y^4$
(c) $36 y^5$
(d) $108 y^5$
(IIT 1997) | 1 |
Is cunnilingus unhealthy? | 0 |
3. Discuss monotonocity of $f(x)=\frac{x}{\sin x}$ and $g(x)=\frac{x}{\tan x}$, where $0<x \leq 1$. | 1 |
17. The IUPAC name of the compound having the formula $\mathrm{CH}_{3}$
$$
\text { }
$$
is :
(a) 3,3,3-Trimethyl-1-propene
(b) 1,1,1-Trimethyl-2-propene
(c) 3, 3-Dimethyl-1-butene
(d) 2, 2-Dimethyl-3-butene
(a) 4 Fill in the Blank | 1 |
What type of religion is Judaism? | 0 |
What makes up the sociological parts of culture according to Simmel? | 0 |
23. If $F(x)$ is continuous at $x=1$, then
(a) $b=a+3$
(b) $b=a-1$
(c) $a=b-2$
(d) None of these | 1 |
Had OJ Simpson not been famous & had the legal defense "dream team" as his attorneys, would he have been convicted in the murder of Nichole Brown Simpson? | 0 |
26. What is the sum of the following four vectors in (a) unitvector notation, and as (b) a magnitude and (c) an angle?
$$
\begin{array}{ll}
\vec{A}=(2.00 \mathrm{~m}) \hat{\mathrm{i}}+(3.00 \mathrm{~m}) \hat{\mathrm{j}} & \vec{B}: 4.00 \mathrm{~m}, \text { at }+65.0^{\circ} \\
\vec{C}=(-4.00 \mathrm{~m}) \hat{\mathrm{... | 1 |
What is the most awkward moment that happened to you due to your clothes? | 0 |
Music is not allowed in Islam than why Arabic singer use music in Arabic songs? | 0 |
19. Two long straight wires, each carrying a current $I$ in opposite directions are separated by a distance $R$. The magnetic induction at a point mid-way between the wires is
(a) zero
(b) $\frac{\mu_{0} I}{\pi R}$
(c) $\frac{2 \mu_{0} I}{\pi R}$
(d) $\frac{\mu_{0} I}{4 \pi R}$ | 1 |
3. A man grows into a giant such that his linear dimensions increase by a factor of 9 . Assuming that his density remains same, the stress in the leg will change by a factor of
(2017 Main)
(a) $\frac{1}{9}$
(b) 81
(c) $\frac{1}{81}$
(d) 9 | 1 |
7. Copper crystallises in fcc with a unit length of $361 \mathrm{pm}$. What is the radius of copper atom?
[Main Online April 25, 2013]
(a) $157 \mathrm{pm}$
(b) $128 \mathrm{pm}$
(c) $108 \mathrm{pm}$
(d) $181 \mathrm{pm}$ | 1 |
20. The number of values of $k$ for which the system of equations $(k+1) x+8 y=4 k$ and $k x+(k+3) y=3 k-1$ has infinitely many solutions, is/are
(2002, 1M)
(a) 0
(b) 1
(c) 2
(d) $\infty$ | 1 |
b) How do rates of evaporation and condensation change initially? | 1 |
Who was the first person to provide education opportunities to females? | 0 |
What's the best way to get a smartphone without data plan? | 0 |
At the end of 2014 what entity would handle the the transaction services for BSE-Mumbai? | 0 |
How do you get a civil engineering degree? | 0 |
28. A particle moves along a horizontal path, such that its velocity is given by $v=\left(3 t^{2}-6 t\right) \mathrm{m} / \mathrm{s}$, where $t$ is the time in seconds. If it is initially located at the origin $O$, determine the distance travelled by the particle in time interval from $t=0$ to $t=3.5$ s and the particl... | 1 |
What is the best webinar platform? | 0 |
A university in USA sent me an acceptance letter without any term of payment regarding my tuition fee will I get a visa? | 0 |
10. The rate coefficient $(k)$ for a particular reactions is $1.3 \times 10^{-4} \mathrm{M}^{-1} \mathrm{~s}^{-1}$ at $100{ }^{\circ} \mathrm{C}$, and $1.3 \times 10^{-3} \mathrm{M}^{-1} \mathrm{~s}^{-1}$ at $150{ }^{\circ} \mathrm{C}$. What is the energy of activation $\left(E_{\mathrm{a}}\right)$ (in $\left.\mathrm{k... | 1 |
7. $A B$ is a wire of uniform resistance. The galvanometer $G$ shows no deflection when the length $A C=20 \mathrm{~cm}$ and $C B=80 \mathrm{~cm}$. The resistance $R$ is equal to
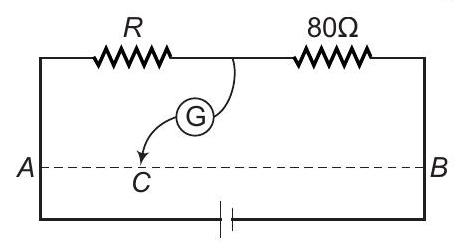
(a) $80 \Om... | 1 |
12. Prove that the locus of a point, which moves so that its distance from a fixed line is equal to the length of the tangent drawn from it to a given circle, is a parabola. | 1 |
Which apps are available to hack wifi password? | 0 |
What is the definitive factor of bandes dessinées? | 0 |
40. Hydrogen bonding is maximum in
(a) Ethanol
(b) Diethyl ether
(c) Ethyl chloride
(d) Triethylamine | 1 |
5. Of the halide ions,______ is the most powerful reducing agent. | 1 |
4. The equation of a tangent to the parabola, $x^{2}=8 y$, which makes an angle $\theta$ with the positive direction of $X$-axis, is
(a) $y=x \tan \theta-2 \cot \theta$
(b) $x=y \cot \theta+2 \tan \theta$
(c) $y=x \tan \theta+2 \cot \theta$
(d) $x=y \cot \theta-2 \tan \theta$ | 1 |
38. For the following electrochemical cell at 298 K
$$
\begin{aligned}
& \mathrm{Pt}(\mathrm{~s})\left|\mathrm{H}_{2}(\mathrm{~g})(1 \mathrm{bar})\right| \mathrm{H}^{+}(\mathrm{aq}) 1 \mathrm{M} \| \mathrm{M}^{4+}(\mathrm{aq}), \mathrm{M}^{2+}(\mathrm{aq}) \mid \mathrm{Pt}(\mathrm{~s}) \\
& E_{\text {cell }}=0.092 \ma... | 1 |
What was the sender in the initial model? | 0 |
7. The line $\frac{x+6}{5}=\frac{y+10}{3}=\frac{z+14}{8}$ is the hypotenuse of an isosceles right angled triangle whose opposite vertex is $(7,2,4)$. Then which of the following is the side of the triangle?
(a) $\frac{x-7}{2}=\frac{y-2}{-3}=\frac{z-4}{6}$
(b) $\frac{x-7}{3}=\frac{y-2}{6}=\frac{z-4}{2}$
(c) $\frac{x-7}{... | 1 |
What is better: IAS or MBA from IIM? | 0 |
What's the best way to eat ice cream? | 0 |
Where did many Indians move to? | 0 |
34.
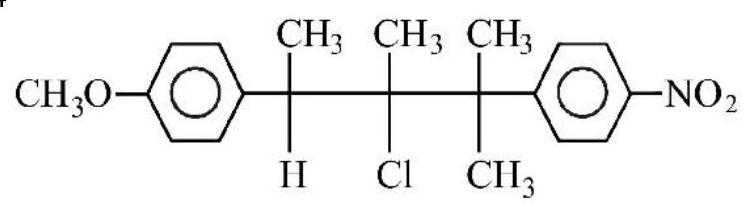
compound on hydrolysis in aqueous acetone will give
(i)
 \cup(\sqrt{7}, 13 / 5)$
c. $(-\infty,-\sqrt{7}) \cup(13 / 5, \sqrt{7})$
d. no such $a$ exists | 1 |
19. Standard electrode potential data are useful for understanding the suitability of an oxidant in a redox titration. Some half cell reactions and their standard potentials are given below :
[2002S]
$$
\begin{array}{r}
\left.\mathrm{MnO}_{4}^{-} \text {(aq.) }+8 \mathrm{H}^{+} \text {(aq.) }\right)+5 \mathrm{e}^{-} ... | 1 |
How can you find out things that you don't know when you don't know what questions to ask? | 0 |
19. The current $i$ in the circuit (see figure) is
(1983, 1M)
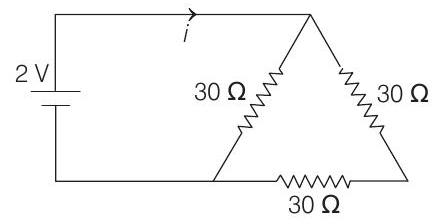
(a) $\frac{1}{45} \mathrm{~A}$
(b) $\frac{1}{15} \mathrm{~A}$
(c) $\frac{1}{10} \mathrm{~A}$
(d) $\frac{1}{5} \mathrm{~A}$
O... | 1 |
The Delhi Metro consists of how many stations? | 0 |
In a study by Masters and Johnson what did the find difficult about KIneys ratings? | 0 |
Whom would you like to walk with? | 0 |
Some people feel that corruption actually reduces costs because it cuts what? | 0 |
Which is the best rom for Lenovo k3 note ? | 0 |
2. If $p+q=1$, then show that $\sum_{r=0}^{n} r^{2}{ }^{n} C_{r} p^{r} q^{n-r}=n p q+n^{2} p^{2}$. | 1 |
What is the difference between "on time" and "in time"? | 0 |
What area of the hippocampus plays a role in storing new memories? | 0 |
60. In an election, the number of candidates is one greater than the persons to be elected. If a voter can vote in 254 ways, the number of candidates is
a. 7
b. 10
c. 8
d. 6 | 1 |
How did the technical committee come up with the idea for the yield operator in JavaScript ES6? How is yield implemented in JavaScript engines? | 0 |
Example 20 After the drop detaches, its surface energy is
(a) $1.4 \times 10^{-6} \mathrm{~J}$
(b) $2.7 \times 10^{-6} \mathrm{~J}$
(c) $5.4 \times 10^{-6} \mathrm{~J}$
(d) $8.1 \times 10^{-9} \mathrm{~J}$ | 1 |
My dog just ate a frog, what can I do? | 0 |
Which is the best laptop below Rs 30000? | 0 |
Why do entrepreneurs need to be resilient? | 0 |
Are there any alternative question and answer websites to Quora? | 0 |
Ex. 36. Calculate the hydrolysis constant of KF. Determine the degree of hydrolysis of the salt in a 0.01 M solution and the pH of the solution. $K_{\mathrm{a}}(\mathrm{HF})=6.6 \times 10^{-4}$. | 1 |
9. The smallest positive integer $n$ for which $\frac{1+i^{n}}{1-i}=1$, is
(a) 8
(b) 16
(c) 12
(d) None of these
(1980, 2M) | 1 |
5. (a) Calculate the molarity of hydrogen chloride in a solution when 0.365 g of it has been dissolved in 100 mL of the solution.
(b) 3 g of a salt of molecular weight 30 is dissolved in 250 g of water. The molality of the solution is ... .
[(a) 0.1 M (b) 0.4 m ] | 1 |
19. In which case is a reaction possible at any temperature?
(a) $\Delta H<0, \Delta S>0$
(b) $\Delta H<0, \Delta S<0$
(c) $\Delta H>0, \Delta S>0$
(d) in none of the cases | 1 |
100. $\mathrm{KOH}+\mathrm{KMnO}_{4}=\mathrm{K}_{2} \mathrm{MnO}_{4}+\mathrm{O}_{2}+\mathrm{H}_{2} \mathrm{O}$ | 1 |
17. A cubical block of side $L$ rests on a rough horizontal surface with coefficient of friction $\mu$. A horizontal force $F$ is applied on the block as shown. If the coefficient of friction is sufficiently high, so that the block does not slide before toppling, the minimum force required to topple the block is
 $\mathrm{A} \rightarrow \mathrm{A}^{+}+\mathrm{e} ; \quad E^{0}=+1.2 \mathrm{~V}$
(ii) $\mathrm{B}^{-} \rightarrow \mathrm{B}+\mathrm{e} ; \quad E^{0}=-2.1 \mathrm{~V}$
(iii) $\mathrm{C} \rightarrow \mathrm{C}^{2+}+2 \mathrm{e} ; \quad E^{0}=-0.38 \mathrm{~V}$
(iv... | 1 |
41. $\mathrm{CO}_{2}$ is isostructural with :
(a) $\mathrm{HgCl}_{2}$
(b) $\mathrm{SnCl}_{2}$
(c) $\mathrm{C}_{2} \mathrm{H}_{2}$
(d) $\mathrm{NO}_{2}$ | 1 |
33. In a crystal $A B$, which of the following crystal systems will have parameters, $a \neq b \neq c$ and $\alpha=\beta=\gamma=90^{\circ}$ ?
(a) Cubic
(b) Orthorhombic
(c) Monoclinic
(d) Triclinic | 1 |
Is demonetization a failed move? | 0 |
How was the Congress split in 1907? | 0 |
10. For a positively charged particle moving in a $x-y$ plane initially along the $x$-axis, there is a sudden change in its path due to the presence of electric and/or magnetic fields beyond $P$. The curved path is shown in the $x-y$ plane and is found to be non-circular.
 find the $y$-coordinates of the 3rd minima assuming the origin at the central maxima . $(\lambda=$ wavelength of monochromatic light used).
(a) $\pm \... | 1 |
16. Which of the following can't show Tautomerism?
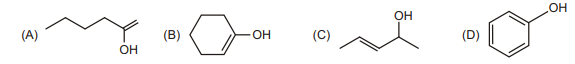 | 1 |
40. When an electric current is passed through acidified water, 112 mL of hydrogen gas at NTP was collected at the cathode in 965 seconds. The current passed, in ampere, is
(a) 0.5
(b) 0.1
(c) 1.0
(d) 2.0
(IIT 2018 Main) | 1 |
Which of the two broad classes of cells: neurons and glial cells send signals to other cells? | 0 |
What is external evidence? | 0 |
Is hair regrowth possible naturally? | 0 |
What is the best drama/ romance anime in English? | 0 |
Example 5 Imagine a light planet revolving around a very massive star in a circular orbit of radius $R$ with a period of revolution T. If the gravitational force of attraction between the planet and the star is proportional to $R^{-5 / 2}$, then
(a) $T^{2}$ is proportional to $R^{2}$
(b) $T^{2}$ is proportional to $R... | 1 |
4. Calculate the packing fraction for the K unit cell. K crystallizes in a body-centred cubic unit cell. | 1 |
Illustration 2.64
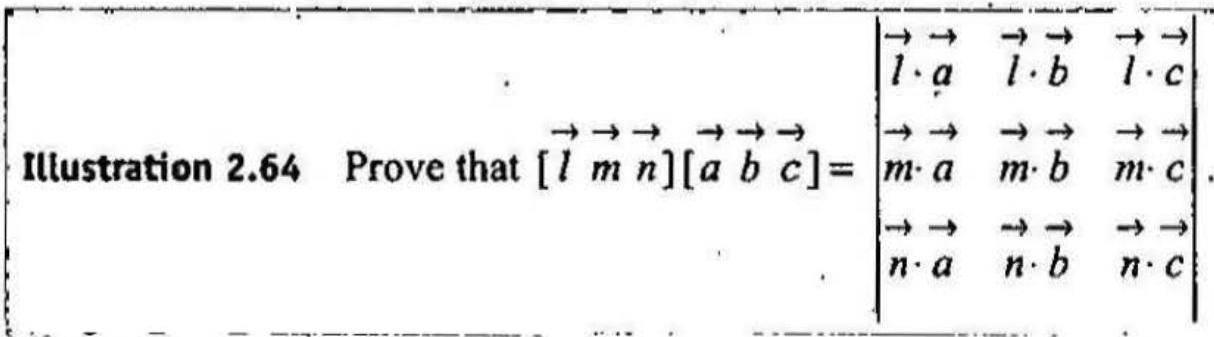 | 1 |
If I'm trapped in a falling elevator, what is my best survival strategy? | 0 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.