Question stringlengths 2 3k | label int64 0 1 |
|---|---|
How do Indian students find job after graduating with MS in CS from average universities in the US? | 0 |
What Babylonian ruler created formal law tables? | 0 |
8. $6.022 \times 10^{22}$ molecules of $\mathrm{N}_2$ at NTP will occupy a volume of
(a) 22.4 litres
(b) 2.24 litres
(c) 6.02 litres
(d) 6.02 mL | 1 |
6. The figures below depict two situations in which two infinitely long static line charges of constant positive line charge density $\lambda$ are kept parallel to each other
In their resulting electric field, point charges $q$ and $-q$ are kept in equilibrium between them. The point charges are confined to move in th... | 1 |
What are the most interesting products and innovations that Green Bank is coming out with in 2016? | 0 |
Why do we feel fresh after we sleep? What exactly happens in our body during sleep? | 0 |
Where was Hyderabad ranked in India in 2012 in terms of deposits? | 0 |
2. For virtual objects, match the following two columns.
| | Column I | Column II |
| :---: | :---: | :---: |
| (a) | Plane mirror | (p) only real image |
| (b) | Convex mirror | (q) only virtual image |
| (c) | Concave mirror | (r) may be real or virtual image | | 1 |
What does Thomas More try to say via these lines from an excerpt taken from Thomas More's Utopia? | 0 |
What was Ruskin's career? | 0 |
Why is it wrong to keep animals as pets? | 0 |
13. Which of the following is not true about $h_{1}(x)$ ?
a. It is periodic function with period $\pi$
b. Range is $[0,1]$
c. Domain is $R$
d. None of these | 1 |
On the Quora website, which computer programming related question has had the most views, before anyone decided to answer it, and how many views did the question have? | 0 |
What are the 2 titular names of God in the Jewish religion? | 0 |
Why do central banks exist? | 0 |
79. $500 \mathrm{~mL}$ of $0.2 \mathrm{M}$ aqueous solution of acetic acid is mixed with 500 $\mathrm{mL}$ of $0.2 \mathrm{M} \mathrm{HCl}$ at $25^{\circ} \mathrm{C}$.
(i) Calculate the degree of dissociation of acetic acid in the resulting solution and $\mathrm{pH}$ of the solution.
(ii) If $6 \mathrm{~g}$ of $\math... | 1 |
8. Let $f(x)=\int e^{x}(x-1)(x-2) d x$. Then $f$ decreases in the interval
a. $(-\infty,-2)$
b $(-2,-1)$
c. $(1,2)$
d $(2,+\infty)$ | 1 |
- Example 10 The potential difference applied to an $X$-ray tube is $5 \mathrm{kV}$ and the current through it is $3.2 \mathrm{~mA}$. Then, the number of electrons striking the target per second is
(a) $2 \times 10^{16}$
(b) $5 \times 10^{6}$
(c) $1 \times 10^{17}$
(d) $4 \times 10^{15}$ | 1 |
What should I do if I have a very important question to ask but someone doesn't return my email? | 0 |
What is the additive inverse of a number? | 0 |
What is the theoretical storage capacity of the human brain? | 0 |
67 Continuation of Problem 65. Let reference frame $C$ in Fig37-31 move past reference frame $D$ (not shown). (a) Show that
$$
M_{A D}=M_{A B} M_{B C} M_{C D}
$$
(b) Now put this general result to work: Three particles move parallel to a single axis on which an observer is stationed. Let plus and minus signs indicate... | 1 |
5. Assertion : A particle has constant acceleration is $x-y$ plane. But neither of its acceleration components ( $a_{x}$ and $a_{y}$ ) is zero. Under this condition particle cannot have parabolic path.
Reason : In projectile motion, horizontal component of acceleration is zero. | 1 |
Can you go deaf from too much ear wax? | 0 |
Example 13 An open elevator is ascending with constant speed $v=10 \mathrm{~m} / \mathrm{s}$. A ball is thrown vertically up by a boy on the lift when he is at a height $h=10 \mathrm{~m}$ from the ground. The velocity of projection is $v=30 \mathrm{~m} / \mathrm{s}$ with respect to elevator. Find
(a) the maximum heigh... | 1 |
1. The atomic number of the element unnilennium is :
(a) 109
(b) 102
(c) 108
(d) 119 | 1 |
Who developed a theory similar to that of James? | 0 |
- Example 1 Consider the following information regarding the number of men and women workers in three factories I, II and III
| | Men workers | Women workers |
|-----|-------------|---------------|
| I | 30 | 25 |
| II | 25 | 31 |
| III | 27 | 26 ... | 1 |
Are Japanese people impressed by a foreigner who can fluently speak Japanese? | 0 |
What is the easiest way of committing suicide? | 0 |
How can detect the provenance of news articles? | 0 |
\$\cdot\$0016 The box-like Gaussian surface shown in Fig. 23-38 encloses a net charge of $+24.0 \varepsilon_{0} \mathrm{C}$ and lies in an electric field given by $\vec{E}=[(10.0+2.00 x) \hat{\mathrm{i}}-3.00 \hat{\mathrm{j}}+b z \hat{\mathrm{k}}] \mathrm{N} / \mathrm{C}$, with $x$ and $z$ in meters and $b$ a constant.... | 1 |
Ex. 21. What weight of CO is required to form $\mathrm{Re}_{2}(\mathrm{CO})_{10}$ from 2.50 g of $\operatorname{Re}_{2} \mathrm{O}_{7}$ according to the unbalanced reaction:
$$
\begin{aligned}
& \mathrm{Re}_{2} \mathrm{O}_{7}+\mathrm{CO} \rightarrow \mathrm{Re}_{2}(\mathrm{CO})_{10}+\mathrm{CO}_{2} \\
& (\mathrm{Re}=1... | 1 |
How does YouTube calculate its views? | 0 |
What language is spoken in northwest Spain? | 0 |
What does a Donald Trump presidency mean for the US - India relationship? | 0 |
What net-worth do you consider rich? Why? | 0 |
In every Apple advert, the time on the iPhone screen is 9.41. Why? | 0 |
4. The number of neutrons accompanying the formation of ${ }_{54}^{139} \mathrm{Xe}$ and ${ }_{38}^{94} \mathrm{Sr}$ from the absorption of a slow neutron by ${ }_{92}^{235} \mathrm{U}$, followed by nuclear fission is,
(a) 0
(b) 2
(c) 1
(d) 3 | 1 |
59. The standard potential of the following cell is $0.23 \mathrm{~V}$ at $15^{\circ} \mathrm{C}$ and 0.21 $\mathrm{V}$ at $35^{\circ} \mathrm{C}$.
[2001 - 10 Marks]
Pt $\left|\mathrm{H}_{2}(\mathrm{~g})\right| \mathrm{HCl}(\mathrm{aq})|\operatorname{AgCl}(\mathrm{s})| \mathrm{Ag}(\mathrm{s})$
(i) Write the cell rea... | 1 |
Why don't many people posting questions on Quora check Google first? | 0 |
35. Let $M$ be a $3 \times 3$ matrix satisfying $M 1=2$,
$0 \quad 3$
$$
\begin{aligned}
& \begin{array}{llll}
1 & 1 & 1 & 0
\end{array} \\
& M-1=1 \text {, and } M 1=0 \text {, } \\
& \begin{array}{llll}
0 & -1 & 1 & 12
\end{array}
\end{aligned}
$$
Then, the sum of the diagonal entries of $M$ is ...
(2011) | 1 |
52. A compound $\mathrm{H}_{2} \mathrm{X}$ with molar weight 80 g is dissolved in a solvent having density $0.4 \mathrm{~g} \mathrm{~mL}^{-1}$. Assuming no change in volume upon dissolution, the molality of a 3.2 Molar solution is ... .
(8) | 1 |
Why are some white women attracted to only black men? | 0 |
(ii)
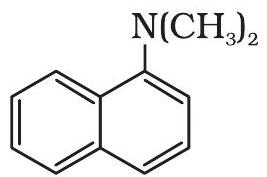 | 1 |
8. A solution containing a group-IV cation gives a precipitate on passing $\mathrm{H}_{2} \mathrm{~S}$. A solution of this precipitate in dil. $\mathrm{HCl}$ produces a white precipitate with $\mathrm{NaOH}$ solution and bluish-white precipitate with basic potassium ferrocyanide. The cation is :
(a) $\mathrm{Co}^{2+... | 1 |
What's the best book on linear algebra? | 0 |
Inland problem transporting a 31m large component | 0 |
35. The standard molar enthalpies of formation of cyclohexane(l)and benzene(1) at $25^{\circ} \mathrm{C}$ are -156 and $+49 \mathrm{~kJ} \mathrm{~mol}^{-1}$ respectively. The
standard enthalpy of hydrogenation of cyclohexene(1) at $25^{\circ} \mathrm{C}$ is -119 $\mathrm{kJ} \mathrm{mol}^{-1}$. Use these data to estima... | 1 |
What's the best way to overcome a fear of spiders? | 0 |
16. Straight lines $3 x+4 y=5$ and $4 x-3 y=15$ intersect at the point $A$. Points $B$ and $C$ are chosen on these two lines such that $A B=A C$. Determine the possible equations of the line $B C$ passing through the point $(1,2)$. | 1 |
29. Draw the current-voltage characteristics for the device shown in figure (45-E7) between the terminals $A$ and $B$.
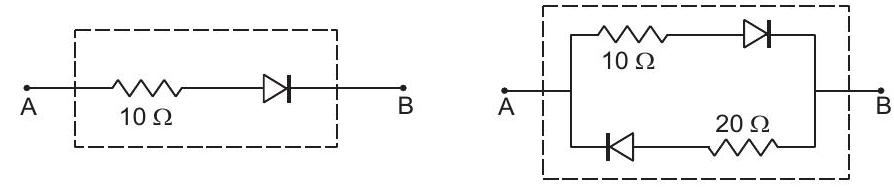
Figure $45-\mathrm{E} 7$ | 1 |
Why is Quora biased towards Trump? | 0 |
10.
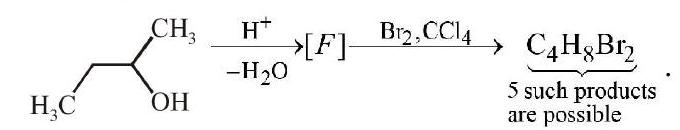
How many structures for $\mathrm{F}$ are possible?
(a) 2
(b) 5
(c) 6
(d) 3 | 1 |
1. The function $f: N \rightarrow N$ ( $N$ is the set of natural numbers), defined by $f(n)=2 n+3$ is
a. surjective only
b. injective only
c. bijective
d. None of these | 1 |
(ii) $g(x)=\log x$ | 1 |
Why is money not the most important thing in life? | 0 |
What is another way to refer to the PhulMali? | 0 |
27. The value of the determinant $\left|\begin{array}{ccc}1 & 1 & 1 \\ { }^{m} C_{1} & { }^{m+1} C_{1} & { }^{m+2} C_{1} \\ { }^{m} C_{2} & { }^{m+1} C_{2} & { }^{m+2} C_{2}\end{array}\right|$ is equal to
a. 1
b. -1
c. 0
d. none of these | 1 |
What do I say to my boss when I'm sick? | 0 |
1. 0.3 g of a compound on combustion gave 0.54 g of water and 0.88 g of carbon dioxide. Find the percentages of carbon and hydrogen in the compound.
(80\%, 20\%) | 1 |
1. Which of the following is not an essential amino acid?
[Main Sep. 05, $2020(\mathbf{I})]$
(a) Tyrosine
(b) Leucine
(c) Valine
(d) Lysine | 1 |
3. Let $A=\left[\begin{array}{ccc}1 & 0 & 0 \\ 0 & 1 & 1 \\ 0 & -2 & 4\end{array}\right], I=\left[\begin{array}{lll}1 & 0 & 0 \\ 0 & 1 & 0 \\ 0 & 0 & 1\end{array}\right]$ and $A^{-1}=\left[\frac{1}{6}\left(A^{2}+c A+d I\right)\right]$. The values of $c$ and $d$ are
a. $(-6,-11)$
b. $(6,11)$
c. $(-6,11)$
d. $(6,-11)$ | 1 |
What type of corruption is hard to get rid of even in developed countries? | 0 |
92. The range of $f(x)=\cos ^{-1}\left(\frac{1+x^{2}}{2 x}\right)+\sqrt{2-x^{2}}$ is
a. $\left\{0,1+\frac{\pi}{2}\right\}$
b. $\{0,1+\pi\}$
c. $\left\{1,1+\frac{\pi}{2}\right\}$
d. $\{1,1+\pi\}$ | 1 |
37. A solution of a non-volatile solute in water freezes at $-0.30^{\circ} \mathrm{C}$. The vapour pressure of pure water at $298 \mathrm{~K}$ is $23.51 \mathrm{~mm} \mathrm{Hg}$ and $K_{f}$ for water is $1.86 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1}$. Calculate the vapour pressure of this solution at $298 \mathrm{~... | 1 |
4. The relative strength of interionic/ intermolecular forces in decreasing order is:
[Main Jan. 07, 2020 (I)]
(a) dipole-dipole $>$ ion-dipole $>$ ion-ion
(b) ion-dipole $>$ ion-ion $>$ dipole-dipole
(c) ion-dipole $>$ dipole-dipole $>$ ion-ion
(d) ion-ion $>$ ion-dipole $>$ dipole-dipole | 1 |
75 SSM A certain stable nuclide, after absorbing a neutron, emits an electron, and the new nuclide splits spontaneously into two alpha particles. Identify the nuclide. | 1 |
Repression of political opponants and police brutality are forms of political what? | 0 |
How do I make $1000 in one day? | 0 |
What architectural writing came from Sri Lanka? | 0 |
What two words is "architecture" a combination of? | 0 |
2.
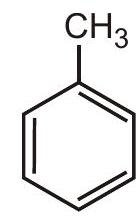
(I)
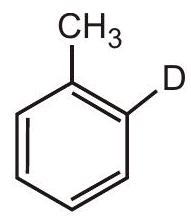
(II)
 0.5
(b) 0.25
(c) 1.0
(d) No sound is heard | 1 |
- Example 21.11 p-V plots for two gases during adiabatic processes are shown in the figure. Plots 1 and 2 should correspond respectively to
(a) $\mathrm{He}$ and $\mathrm{O}_{2}$
(b) $\mathrm{O}_{2}$ and $\mathrm{He}$
(c) He and Ar
(d) $\mathrm{O}_{2}$ and $\mathrm{N}_{2}$
 $\frac{1}{\sqrt{5}}$
(b) $\frac{1}{\sqrt{2}}$
(c) $\frac{1}{\sqrt{3}}$
(d) $\frac{1}{\sqrt{6}}$ | 1 |
18. The electric field $E$ is measured at a point $P(0,0, d)$ generated due to various charge distributions and the dependence of $E$ on $d$ is found to be different for different charge distributions. List-I contains different relations between $E$ and $d$. List-II describes different electric charge distributions, al... | 1 |
Speed of mobile internet (accessed through the SIM card provided by ISP) is lower when used in phone browser but higher when tethered to laptop? Why? | 0 |
7. The radius of the circumcircle of the $\triangle P R S$ is
(a) 5
(b) $3 \sqrt{3}$
(c) $3 \sqrt{2}$
(d) $2 \sqrt{3}$ | 1 |
Will I face any trouble for hosting Tor node in India? | 0 |
What are some tips to clear both groups of IPCC at one shot? | 0 |
How hard is it to practice water polo compared to canoeing? | 0 |
(i) $(\mathrm{A}+\mathrm{B})^{\prime}=\mathrm{A}^{\prime}+\mathrm{B}^{\prime}$, | 1 |
What is the best book for teaching C++? | 0 |
5. The differential equation of all parabolas whose axis are parallel to the $y$-axis is
a. $\frac{d^{3} y}{d x^{3}}=0$
b. $\frac{d^{2} x}{d y^{2}}=C$
c. $\frac{d^{3} y}{d x^{3}}+\frac{d^{2} x}{d y^{2}}=0$
d. $\frac{d^{2} y}{d x^{2}}+2 \frac{d y}{d x}=C$
U. | 1 |
How do I get certified in Salesforce? | 0 |
How do invest in stocks? | 0 |
Did the ancient Romans eat hummus? If so, what ingredients were mixed into their hummus? | 0 |
59. The only CORRECT combination that gives two different carboxylic acids is
(a) (II) (iv) (R)
(b) (IV) (iii) (Q)
(c) (III) (iii) (P)
(d) (I) (i) (S) | 1 |
37. Read the following Assertion and Reason and answer as per the options given below :
Assertion : The electronic structure of $\mathrm{O}_{3}$ is 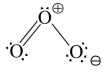
Reason :  according to the equation:
$2 \mathrm{NH}_{2} \mathrm{OH}+4 \mathrm{Fe}^{3+} \rightarrow \mathrm{N}_{2} \mathrm{O}(\mathrm{g}) \uparrow+\mathrm{H}_{2} \mathrm{O}+4 \mathrm{Fe}^{2+}+4 \mathrm{H}^{+}$
Iron (II) thus produced is estimated by titration with a standard permanganate sol... | 1 |
I am a 13 year old girl, and I am overweight. I am 5 feet 5 inches tall, and weigh 120 pounds. How do I lose weight? | 0 |
What can I do to get better grades next quarter? | 0 |
47. Let $\left|z_{r}-r\right| \leq r, \forall r=1,2,3, \ldots, n$. Then $\left|\sum_{r=1}^{n} z_{r}\right|$ is less than
a. $n$
b. $2 n$
c. $n(n+1)$
d. $\frac{n(n+1)}{2}$ | 1 |
4. The magnitude of induced emf on the closed surface of ring will be
(a) $\pi a^{2} B_{0}$
(b) $2 a^{2} B_{0}$
(c) zero
(d) $\frac{1}{2} \pi a^{2} B_{0}$ | 1 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.