Question stringlengths 2 3k | label int64 0 1 |
|---|---|
13. The equation of the curve $C$ is
a. $x^{2}=4 y$
b. $x^{2}=16 y$
c. $x^{2}=12 y$
d. $y^{2}=8 x$ | 1 |
5. If excess metallic iron is added to an $\mathrm{N}-\mathrm{CuSO}_{4}$ solution, calculate the approximate concentration of $\mathrm{Cu}^{2+}$ when equilibrium is established.
$$
\begin{equation*}
\left(3 \times 10^{-27} \mathrm{M}\right) \tag{Yes}
\end{equation*}
$$ | 1 |
56 The electric field in a particular space is $\vec{E}=(x+2) \hat{\mathrm{i}} \mathrm{N} / \mathrm{C}$, with $x$ in meters. Consider a cylindrical Gaussian surface of radius 20 cm that is coaxial with the $x$ axis. One end of the cylinder is at $x=0$. (a) What is the magnitude of the electric flux through the other en... | 1 |
What is the mean yearly temperature in Hyderabad in Celsius? | 0 |
7. A charge $q$ is placed at the centre of the open end of a cylindrical vessel (figure 30-Q3). The flux of the electric field through the surface of the vessel is
(a) zero
(b) $q / \varepsilon_{0}$
(c) $q / 2 \varepsilon_{0}$
(d) $2 q / \varepsilon_{0}$.
 of the height of these six students is
(a) 16
(b) 22
(c) 20
(d) 18 | 1 |
What is it like to live in Dublin? | 0 |
How does sex feel the first time for men? | 0 |
Have Mythical Creatures been scientifically tested? | 0 |
22. The electronic transition from $n=2$ to $n=1$ will produce shortest wavelength in
(a) $\mathrm{Li}^{2+}$
(b) $\mathrm{He}^{+}$
(c) H
(d) $\mathrm{H}^{+}$ | 1 |
25. Vector $\overrightarrow{A B}$ is
a. $\frac{1}{3} \vec{a}+\frac{\vec{a} \times(\vec{b}-\bar{c})}{|\vec{a}|^{2}}$
b. $\frac{1}{3} \vec{a}+\frac{\vec{a} \times(\vec{b}-\vec{c})}{|\vec{a}|^{2}}+\frac{3(\vec{b} \times \vec{a})}{|\vec{a}|^{2}}$
c. $\frac{1}{3} \vec{a}+\frac{\bar{a} \times(\vec{b}-\vec{c})}{|\vec{a}|^{2}}... | 1 |
Why is Saltwater taffy candy imported in China? | 0 |
Ex. 21. 1 mole of $\mathrm{H}_{2}, 2$ moles of $\mathrm{I}_{2}$ and 3 moles of HI were taken in a 1-litre flask. If the value of $\mathrm{K}_{c}$ for the equation $\mathrm{H}_{2}(\mathrm{~g})+\mathrm{I}_{2}(\mathrm{~g}) \rightleftharpoons 2 \mathrm{HI}(\mathrm{g})$ is 50 at $440^{\circ} \mathrm{C}$, what will be the co... | 1 |
What would be the best laptop under Rs. 50k? | 0 |
I want to improve my English? | 0 |
7. What would be the best name for the following compound?
(A) 3-methylhydroxybenzene
(B) 3-methylcyclohexa-13,5-trien-1-ol
(C) 3-methylphenol
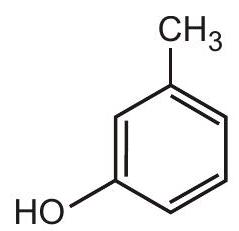
(D) 2-hydroxytoluene | 1 |
What advantages do men have that women don't have? | 0 |
What do some cultures have formal ceremonies for? | 0 |
What are the ways in which I can improve my handwriting? | 0 |
How can you lose weight really quick? | 0 |
67. When a soccer ball is kicked toward a player and the player deflects the ball by "heading" it, the acceleration of the head during the collision can be significant. Figure 2-38 gives the meas-
$
(a) $4 \mathrm{~N}$
(b) $8 \mathrm{~N}$
(c) $12 \mathrm{~N}$
(d)... | 1 |
2. The major product of the following reaction is:

(a) 
(b) 
(c)  in India after many sites have been banned? | 0 |
Why it is said that Joker makes a lot of sense in the movie Dark Knight? | 0 |
What's the latency between shutter button press and the start of the video taking in a GoPro Hero 4? | 0 |
50. Sodium metal crystallises in a body centred cubic lattice with a unit cell edge of $4.29 \AA$. The radius of Na atom is approximately
(a) $1.86 \AA$
(b) $3.22 \AA$
(c) $5.72 \AA$
(d) $0.93 \AA$ | 1 |
50. $2 n$ boys are randomly divided into two subgroups containing $n$ boys each. The probability that the two tallest boys are in different groups is
a. $n /(2 n-1)$
b. $(n-1) /(2 n-1)$
c. $(n-1) / 4 n^{2}$
d. none of these | 1 |
2. Approximate distance travelled by the block when it comes to rest for a second time (not including the initial one) will be (Take $\sqrt{45}=6.70$ )
(a) $30 \mathrm{~cm}$
(b) $25 \mathrm{~cm}$
(c) $40 \mathrm{~cm}$
(d) $20 \mathrm{~cm}$ | 1 |
What is the hollow sphere that a zygote initially develops into called? | 0 |
What are the top 5 resources for learning Javascript? | 0 |
1. The degree of the differential equation satisfying
$$
10 \sqrt{1-x^{2}}+\sqrt{1-y^{2}}=a(x-y) \text { is }
$$
a. 1 .
b. 2
c. 3
d. None of these | 1 |
21. The wave number of the first emission line in the Balmer series of H-Spectrum
$(\mathrm{R}=$ Rydberg constant $):$
(a) $\frac{5}{36} R$
(b) $\frac{9}{400} R$
(c) $\frac{7}{6} R$
(d) $\frac{3}{4} R$ | 1 |
I love a girl. I look decent earn good but m chubby have little self confidence. Should I tell that girl I love you and how? | 0 |
Why haven't companies like Seiko, Fossil, etc. made smart watches? | 0 |
7. The lines $2 x-3 y=5$ and $3 x-4 y=7$ are diameters of a circle of area 154 sq. units. Then the equation of this circle is
a. $x^{2}+y^{2}+2 x-2 y=62$
b. $x^{2}+y^{2}+2 x-2 y=47$
c. $x^{2}+y^{2}-2 x+2 y=47$
d. $x^{2}+y^{2}-2 x+2 y=62$ | 1 |
What are some basic tips to learn French quickly? | 0 |
11. A solution at $20^{\circ} \mathrm{C}$ is composed of $1.5 \mathrm{~mol}$ of benzene and $3.5 \mathrm{~mol}$ of toluene. If the vapour pressure of pure benzene and pure toluene at this temperature are 74.7 torr and 22.3 torr, respectively, then the total vapour pressure of the solution and the benzene mole fraction ... | 1 |
30. When compounds of barium are heated in a flame, green light of wavelength 554 nm is emitted. How much energy is lost when one mole of barium atoms each emit one photon of this wavelength?
(216 kJ) | 1 |
2. A $100 \mathrm{~m}$ long antenna is mounted on a $500 \mathrm{~m}$ tall building. The complex can become a transmission tower for waves with $\lambda$
(a) $\sim 400 \mathrm{~m}$
(b) $\sim 25 \mathrm{~m}$
(c) $\sim 150 \mathrm{~m}$
(d) $\sim 2400 \mathrm{~m}$ | 1 |
Are you going to push to improve the ACA? | 0 |
Why did the United States form independent states? | 0 |
People of what ethnicity settled in the Balkan Peninsula? | 0 |
What is the judicial branch of a government? | 0 |
3. Let $g(x)=\int_{0}^{x} f(t) d t$, where $f$ is such that $\frac{1}{2} \leq f(t) \leq 1$ for $t \in[0,1]$ and $0 \leq f(t) \leq \frac{1}{2}$ for $t \in[1,2]$. Then, $g(2)$ satisfies the inequality
(a) $-\frac{3}{2} \leq g(2)<\frac{1}{2}$
(b) $0 \leq g(2)<2$
(c) $\frac{3}{2}<g(2) \leq 5 / 2$
(d) $2<g(2)<4$ | 1 |
44. Identify the intensive quantities from the following:
(a) Enthalpy
(b) Temperature
(c) Volume
(d) Refractive Index | 1 |
When was England's first permanent settlement in the Americas founded? | 0 |
2. Most people interpret a $9.0 \mathrm{~dB}$ increase in sound intensity level as a doubling in loudness. By what factor must the sound intensity be increased to double the loudness? | 1 |
6. The ratio of mass percent of $\mathrm{C}$ and $\mathrm{H}$ of an organic compound $\left(\mathrm{C}_{\mathrm{X}} \mathrm{H}_{\mathrm{Y}} \mathrm{O}_{\mathrm{Z}}\right)$ is $6: 1$. If one molecule of the above compound $\left(\mathrm{C}_{\mathrm{X}} \mathrm{H}_{\mathrm{Y}} \mathrm{O}_{\mathrm{Z}}\right)$ contains hal... | 1 |
7. The sum of the coefficient in the expansion of $\left(1+a x-2 x^{2}\right)^{n}$ is
a. positive, when $a<1$ and $n=2 k, k \in N$
b. negative, when $a<1$ and $n=2 k+1, k \in N$
c. positive, when $a>1$ and $n \in N$
d. zero, when $a=1$ | 1 |
3. The value of $\mathrm{Kc}$ is 64 at $800 \mathrm{~K}$ for the reaction $\mathrm{N}_{2}(\mathrm{~g})+3 \mathrm{H}_{2}(\mathrm{~g}) \rightleftharpoons 2 \mathrm{NH}_{3}(\mathrm{~g})$. The value of $K_{\mathrm{c}}$ for the following reaction is :
$$
\mathrm{NH}_{3}(\mathrm{~g}) \rightleftharpoons \frac{1}{2} \mathrm... | 1 |
Who was the better Bane: Tom Hardy or Jeep Swenson? | 0 |
How can we learn computer? | 0 |
50. Which of the following relates to light both as wave motion as well as particle?
(a) Diffraction and interference
(b) Photoelectric effect
(c) $E=m c^2$
(d) $E=h \nu$ | 1 |
What is quantitative political science? | 0 |
How do I install windows 95 in pantium4 pc? | 0 |
16. The correct decreasing order for acid strength is:
(a) $\mathrm{NO}_{2} \mathrm{CH}_{2} \mathrm{COOH}>\mathrm{FCH}_{2} \mathrm{COOH}>$ $\mathrm{CNCH}_{2} \mathrm{COOH}>\mathrm{ClCH}_{2} \mathrm{COOH}$
(b) $\mathrm{FCH}_{2} \mathrm{COOH}>\mathrm{CNCH}_{2} \mathrm{COOH}>$ $\mathrm{NO}_{2} \mathrm{CH}_{2} \mathrm{... | 1 |
5. Match the following two columns.
| Column I | Column II |
| :--- | :--- |
| (a) $y=A \sin (\omega t-k x)$ | (p) travelling in positive $x$-direction |
| (b) $y=A \sin (k x-\omega t)$ | (q) travelling in negative $x$-direction |
| (c) $y=-A \cos (\omega t+k x)$ | (r)at $t=0$, velocity of particle is positive at <br>... | 1 |
What does an archaeologist do? | 0 |
What three points do most theists agree on? | 0 |
46. Five identical capacitor plates, each of area $A$, are arranged such that adjacent plates are at a distance $d$ apart, the plates are connected to a source of emf $V$ as shown in the figure.

(a) Equilibrium of plank is stable in vertical direction
(b) For small oscillations of plank in vertic... | 1 |
What instrument can you use to examine the microstructure of the brain? | 0 |
7. At $90^{\circ} \mathrm{C}$, pure water has $\left[\mathrm{H}_3 \mathrm{O}^{+}\right]=10^{-6}$ mole/litre. The value of $K_w$ at $90^{\circ} \mathrm{C}$ is
(a) $10^{-6}$
(b) $10^{-8}$
(c) $10^{-12}$
(d) $10^{-14}$
(IIT 1984) | 1 |
Who discovered dengue fever virus? | 0 |
Are fears rational? | 0 |
Along with France and Spain, the kings of what country consolidated power in the High Middle Ages? | 0 |
16. $10 \mathrm{~mL}$ of $2(\mathrm{M}) \mathrm{NaOH}$ solution is added to $200 \mathrm{~mL}$ of 0.5 (M) of $\mathrm{NaOH}$ solution. What is the final concentration?
(a) $0.57(\mathrm{M})$
(b) $5.7(\mathrm{M})$
(c) 11.4 (M)
(d) 1.14 (M) | 1 |
Example 18 Figure shows a man standing stationary with respect to a horizontal conveyor belt that is accelerating with $1 \mathrm{~ms}^{-2}$. What is the net force on the man? If the coefficient of static friction between the man's shoes and the belt is 0.2 , upto
 $\mathrm{MnO}_{4}^{-}$
(b) $\operatorname{Cr}(\mathrm{CN})_{6}^{3-}$
(c) $\mathrm{NiF}_{6}^{2-}$
(d) $\mathrm{CrO}_{2} \mathrm{Cl}_{2}$ | 1 |
6. The eq. wt. of $\mathrm{K}_{2} \mathrm{CrO}_{4}$ as an oxidising agent in acid medium is
(a) mol. wt./2
(b) $\frac{2 \times \mathrm{mol} . \mathrm{wt}}{3}$
(c) $\frac{\mathrm{mol} . \mathrm{wt}}{3}$
(d) $\frac{\mathrm{mol} . \mathrm{wt}}{6}$ | 1 |
53. According to kinetic theory of gases
(a) collisions are always elastic.
(b) heavier molecules transfer more momentum to the walls of container.
(c) only a small number of molecules have very high velocity.
(d) between collisions, the molecules move in straight lines with constant velocity.
(IIT 2011) | 1 |
How can I start preparing for ias exam? I have 4 yrs. | 0 |
12. Given both $\theta$ and $\phi$ are acute angles and $\sin \theta=1 / 2, \cos \phi=1 / 3$, then the value of $\theta+\phi$ belongs to
a. $\left(\frac{\pi}{3}, \frac{\pi}{2}\right]$
$\mathbf{b}\left(\frac{\pi}{2}, \frac{2 \pi}{3}\right]$
c. $\left(\frac{2 \pi}{3}, \frac{5 \pi}{6}\right]$
d. $\left(\frac{5 \pi}{6}, \p... | 1 |
Is Thor stronger than Hulk? | 0 |
What do you think about the ban on 500 and 1000 denomination notes in India? | 0 |
Where can I download Agents of S.H.I.E.L.D. season 2 complete 1080p (From Torrent in between 12-15 GB)? | 0 |
How would an electric airplane work? | 0 |
What is the difference between : and | 0 |
Which is a better bank HDFC or ICICI? | 0 |
12. In allene $\left(\mathrm{C}_{3} \mathrm{H}_{4}\right)$, the type(s) of hybridization of the carbon atoms is (are):
(a) $s p$ and $s p^{3}$
(b) $s p^{2}$ and $s p$
(c) only $s p^{2}$
(d) $s p^{2}$ and $s p$ | 1 |
Along with Cropanzano, who developed a communication-based theory of emotional experience? | 0 |
19. The correct statement(s) for orthoboric acid is/are
(a) It behaves as a weak acid in water due to self ionization.
(b) Acidity of its aqueous solution increases upon addition of ethylene glycol
(c) It has a three dimensional structure due to hydrogen bonding
(d) It is a weak electrolyte in water
( $\gamma$ Ma... | 1 |
Which iOS game engine does the game "Hay Day" use? | 0 |
What is the Lewis structure of SiF4, and how does it compare to that of nitrogen? | 0 |
56. Let $\alpha$ and $\beta$ be any two positive values of $x$ for which $2 \cos x,|\cos x|$ and $1-3 \cos ^{2} x$ are in GP. The minimum value of $|\alpha-\beta|$ is
a. $\frac{\pi}{3}$
b. $\frac{\pi}{4}$
c. $\frac{\pi}{2}$
d none of these | 1 |
14. $600 \mathrm{~J}$ of heat is added to a monoatomic gas in a process in which the gas performs a work of $150 \mathrm{~J}$. The molar heat capacity for the process is
(a) $3 R$
(b) $4 R$
(c) $2 R$
(d) $6 R$ | 1 |
16. 3-Methyl-pent-2-ene on reaction with $\mathrm{HBr}$ in presence of peroxide forms an addition product. The number of possible stereoisomers for the product is :
[Main 2017]
(a) Six
(b) Zero
(c) Two
(d) Four | 1 |
1. The displacement of a damped harmonic oscillator is given by $x(t)=e^{-0.1 t} \cos (10 \pi t+\varphi)$.
Here, $t$ is in seconds.
The time taken for its amplitude of vibration to drop to half of its initial value is close to
(2019 Main, 10 April I)
(a) $27 \mathrm{~s}$
(b) $13 \mathrm{~s}$
(c) $4 \mathrm{~s}$
(d) ... | 1 |
Where can I get cannabis in Bangalore? | 0 |
What is the name commonly used to refer to the entire metropolis of Delhi? | 0 |
5. Thermal decomposition of a Mn compound ( $\mathrm{X}$ ) at $513 \mathrm{~K}$ results in compound $\mathrm{Y}, \mathrm{MnO}_{2}$ and a gaseous product. $\mathrm{MnO}_{2}$ reacts with $\mathrm{NaCl}$ and concentrated $\mathrm{H}_{2} \mathrm{SO}_{4}$ to give a pungent gas $\mathrm{Z}$. $\mathrm{X}$, Y, and $\mathrm{Z}$... | 1 |
Who was Britain's last serious rival after Napoleon? | 0 |
How do you improve your drawing? | 0 |
82. If $\vec{a}$ and $\vec{b}$ are non-zero non-collinear vectors, then $[\vec{a} \vec{b} \hat{i}] \hat{i}+[\vec{a} \vec{b} \hat{j}] \hat{j}+[\vec{a} \vec{b} \hat{k}] k$ is equal to
a. $\vec{a}+\vec{b}$
b. $\vec{a} \times \vec{b}$
c. $\vec{a}-\vec{b}$
d. $\vec{b} \times \vec{a}$ | 1 |
Do universities worldwide recognize CBSE patrachar? | 0 |
What is the only city that had a lower vaccination rate for children than Hyderabad in the 2005 National Family Health Survey? | 0 |
44. The position vectors of the vertices $A, B$ and $C$ of a triangle are three unit vectors $\hat{a}, \hat{b}$ and $\hat{c}$, respectively. A vector $\vec{d}$ is such that $\vec{d} \cdot \hat{a}=\vec{d} \cdot \hat{b}=\vec{d} \cdot \hat{c}$ and $\vec{d}=\lambda(\hat{b}+\hat{c})$. Then triangle $A B C$ is
a. acute angle... | 1 |
15. In van der Waals equation of state for a non-ideal gas, the term that accounts for intermolecular forces is
[1988 - 1 Mark]
(a) $(V-b)$
(b) $R T$
A. 0.217
C. 18.000
D. 24.060
$$
\text { }
$$
(c) $\left(P+\frac{a}{V^{2}}\right)$
(d) $(R T)^{-1}$ | 1 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.