Question stringlengths 2 3k | label int64 0 1 |
|---|---|
(ii) have wavelength of $0.50 \AA \AA$. | 1 |
6. The increasing order of $\mathrm{pKa}$ of the following amino acids in aqueous solution is:
Gly Asp Lys Arg (a) Asp $<$ Gly $<$ Arg $<$ Lys
(b) Gly $<$ Asp $<$ Arg $<$ Lys
(c) Asp $<$ Gly $<$ Lys $<$ Arg
(d) Arg $<$ Lys $<$ Gly $<$ Asp | 1 |
How can I improve my communication skills? | 0 |
If a = 149 , then find the value of x? | 0 |
12. Let $E=\{1,2,3,4\}$ and $F=\{1,2\}$. Then, the number of onto functions from $E$ to $F$ is
(2001, 1M)
(a) 14
(b) 16
(c) 12
(d) 8 | 1 |
4. If the man wants to move the block up the incline, what minimum force is required to start the motion?
(a) $\frac{2}{3} m g$
(b) $\frac{m g}{2}$
(c) $\frac{7 m g}{6}$
(d) $\frac{5 m g}{6}$ | 1 |
What city has the highest number of registered cars of any city in India? | 0 |
Why are people addicted to alcohol and or other drugs? | 0 |
What was New Delhi's Per Capita GDP in dollars for 2009-10? | 0 |
32. A list of species having the formula $\mathrm{XZ}_{4}$ is given below.
$\mathrm{XeF}_{4}, \mathrm{SF}_{4}, \mathrm{SiF}_{4}, \mathrm{BF}_{4}^{-}, \mathrm{BrF}_{4}^{-},\left[\mathrm{Cu}\left(\mathrm{NH}_{3}\right)_{4}\right]^{2+},\left[\mathrm{FeCl}_{4}\right]^{2-},\left[\mathrm{CoCl}_{4}\right]^{2-}$ and $\l... | 1 |
1. Consider the following reactions
$\mathrm{A} \rightarrow \mathrm{P} 1 ; \mathrm{B} \rightarrow \mathrm{P} 2 ; \mathrm{C} \rightarrow \mathrm{P} 3 ; \mathrm{D} \rightarrow \mathrm{P} 4$,
The order of the above reactions are (i), (ii), (iii), and (iv), respectively. The following graph is obtained when $\log [$ rate... | 1 |
19. Give reasons for the following:
(i)

Explain.
(ii) 7-Bromo-1, 3, 5-cycloheptatriene exists as ionic compound, while 5 bromo-1, 3-cyclopentadiene does not ionise even in presence of $\mathrm{Ag}^{+}$ ion. Explain. | 1 |
4. The oxoacid of sulphur that does not contain bond between Sulphur atoms is :
(a) $\mathrm{H}_{2} \mathrm{~S}_{4} \mathrm{O}_{6}$
(b) $\mathrm{H}_{2} \mathrm{~S}_{2} \mathrm{O}_{3}$
(c) $\mathrm{H}_{2} \mathrm{~S}_{2} \mathrm{O}_{7}$
(d) $\mathrm{H}_{2} \mathrm{~S}_{2} \mathrm{O}_{4}$ | 1 |
25. Let $S=\{1,2,3, \ldots, 100\}$. The number of non-empty subsets $A$ of $S$ such that the product of elements in $A$ is even, is
(a) $2^{50}\left(2^{50}-1\right)$
(b) $2^{50}-1$
(c) $2^{50}+1$
(d) $2^{100}-1$ | 1 |
Is it required to take rest/sleep after consuming medicines? | 0 |
1. Let $\vec{A}$ be a unit vector along the axis of rotation of a purely rotating body and $\vec{B}$ be a unit vector along the velocity of a particle $P$ of the body away from the axis. The value of $\vec{A} \cdot \vec{B}$ is
(a) 1
(b) -1
(c) 0
(d) None of these. | 1 |
45. State with balanced equations what happens when : acetic anhydride reacts with phenol in presence of a base. | 1 |
What reflects the prevalence rates varying? | 0 |
How can I get rid of tiny cockroaches? | 0 |
15. $A$ is a set containing $n$ elements. $A$ subset $P$ of $A$ is chosen at random. The set $A$ is reconstructed by replacing the elements of $P$. A subset $Q$ of $A$ is again chosen at random. Find the prob ability that $P$ and $Q$ have no common elements. | 1 |
Why do so many people post questions on Quora that could be easily and thoroughly answered by simply typing the question into any search engine? | 0 |
Who has the most beautiful natural boobs you've ever seen? | 0 |
How can I clear bank exams after my graduation? Which books to refer? | 0 |
I recently had a serious misunderstanding with my boyfriend, my only love. After 2 years of a relationship, he gave me all my things back and said he might need to think about what we mean to each other. Is he trying to break up with me? | 0 |
What type of parties allow leaders to form frontbench teams? | 0 |
20. The piston is taken completely out of the cylinder. The hole at the top is sealed. A water tank is brought below the cylinder and put in a position so that the water surface in the tank is at the same level as the top of the cylinder as shown in the figure. The density of the water is $\rho$. In
 increases
(b) decreases
(c) remains same
(d) Nothing can be said | 1 |
A girl that I like, who shall remain nameless, has a boyfriend that she met on Kik. I'm really sad now, what should I do? | 0 |
How can you survive a punctured lung? | 0 |
What is the reason for the UNO's division into regions? | 0 |
Would ISPs block website builders if there wasn't net neutrality? | 0 |
4. Two tangents are drawn from the point $(-2,-1)$ to the parabola $y^{2}=4 x$. If $\theta$ is the angle between these tangents then $\tan \theta=$ | 1 |
9. Figure (3-Q5) shows the position of a particle moving on the $X$-axis as a function of time.
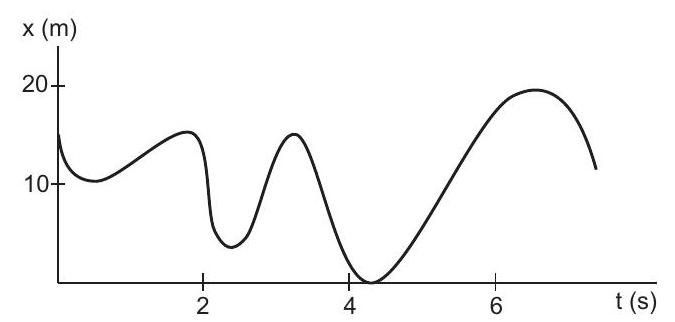
(a) The particle has come to rest 6 times.
(b) The maximum speed is at $t=6 \mathrm{~s}$.
(c... | 1 |
Daniel Ek: Why hasn't Spotify come to India yet? When is it launching in India? | 0 |
What does it mean if God can't predict the future? | 0 |
21. A small bar $A$ resting on a smooth horizontal plane is attached by threads to a point $P$ as shown in the figure and by means of a weightless pulley, to a weight $B$ possessing the same mass as the bar itself. Besides, the bar is also attached to a point $O$ by means of a light non-deformed spring of length $\ell_... | 1 |
4. For the reaction $2 \mathrm{~A}+3 \mathrm{~B}+\frac{3}{2} \mathrm{C} \rightarrow 3 \mathrm{P}$, which statement is correct ?
(a) $\frac{\mathrm{dn}_{\mathrm{A}}}{\mathrm{dt}}=\frac{3}{2} \frac{\mathrm{dn}_{\mathrm{B}}}{\mathrm{dt}}=\frac{3}{4} \frac{\mathrm{dn}_{\mathrm{C}}}{\mathrm{dt}}$
(b) $\frac{\mathrm{dn}_... | 1 |
Which state in Bangladesh speaks pure Hindi? | 0 |
Which country was Britain's most valuable and populous possession? | 0 |
3. Three ammeters $A, B$ and $C$ of resistances $R_{A}, R_{B}$ and $R_{C}$ respectively are joined as shown. When some potential difference is applied across the terminals $T_{1}$ and $T_{2}$, their readings are $I_{A}, I_{B}$ and $I_{C}$ respectively. Then,
=\left\{\begin{array}{ll}\sin 2 x, & 0, x \leq \pi / 6 \\ a x+b, & \pi / 6<x<1\end{array}\right.$. If $f(x)$ and $f^{\prime}(x)$ are
- 1 continuous, then
a. $a=1, b=\frac{1}{\sqrt{2}}+\frac{\pi}{6}$
b. $a=\frac{1}{\sqrt{2}}, b=\frac{1}{\sqrt{2}}$
c. $a=1, b=\frac{\sqrt{3}}{2}-\frac{\pi}{6}$
d. None of the... | 1 |
17. A particle is moving in a circular path in the vertical plane. It is attached at one end of a string of length $l$ whose other end is fixed. The velocity at lowest point is $u$. The tension in the string is $\mathbf{T}$ and acceleration of the particle is a at any position. Then $\mathbf{T} \cdot \mathbf{a}$ is zer... | 1 |
6. A curve is given by the equations $x=\sec ^{2} \theta, y=\cot \theta$. If the tangent at $P$ where $\theta=\pi / 4$ meets the curve again at $Q$, then $[P Q]$ is, where $[\cdot]$ represents the greatest integer function, | 1 |
What is the 'Early Modern period" known as in Japan? | 0 |
Example 14 If A and B are two independent events, then the probability of occurrence of at least one of A and B is given by $1-\mathrm{P}\left(\mathrm{A}^{\prime}\right) \mathrm{P}\left(\mathrm{B}^{\prime}\right)$ | 1 |
Is it possible to account for the Hebrew Bible without believing that it is the revealed word of God? | 0 |
21. $2.5 \mathrm{~mL}$ of $(2 / 5) \mathrm{M}$ weak monoacidic base $\left(K_{b}=1 \times 10^{-12}\right.$ at $\left.25^{\circ}\right)$ is titrated $(2 / 15) \mathrm{M} \mathrm{HCl}$ in water at $25^{\circ} \mathrm{C}$. The concentration of $\mathrm{H}^{+}$at equivalence point is ( $K_{w}=1 \times 10^{-14}$ at $25^{\ci... | 1 |
18. A thin uniform circular tube is kept in a vertical plane. Equal volumes of two immiscible liquids whose densities are $\rho_{1}$ and $\rho_{2}$ fill half of the tube as shown. In equilibrium the radius passing through the interface makes an angle of $30^{\circ}$ with vertical. The ratio of densities $\left(\rho_{1}... | 1 |
How can I get Indian-made detergent powder in South India? Is there any online solution for it? | 0 |
Where can I watch Inferno movie online? | 0 |
1. If $\alpha, \beta(\alpha<\beta)$ are the roots of the equation $6 x^{2}+11 x+3=0$, then which of the following are real?
a. $\cos ^{-1} \alpha$
h. $\sin ^{-1} \beta$
c. $\operatorname{cosec}^{-1} \alpha$
d Both $\cot ^{-1} \alpha$ and $\cot ^{-1} \beta$ | 1 |
12. The rms velocity of hydrogen is $\sqrt{7}$ times the rms velocity of nitrogen. If $T$ is the temperature of the gas, then
(a) $T\left(\mathrm{H}_{2}\right)=T\left(\mathrm{~N}_{2}\right)$
(b) $T\left(\mathrm{H}_{2}\right)>T\left(\mathrm{~N}_{2}\right)$
(c) $T\left(\mathrm{H}_{2}\right)<T\left(\mathrm{~N}_{2}\righ... | 1 |
(x) Schiff's base | 1 |
4. 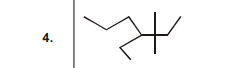 | 1 |
Which Presidency would mean better for Indo-US relationship? Donald Trump's or Hillary Clinton's? | 0 |
69 Figure $23-59$ shows, in cross section, three infinitely large nonconducting sheets on which charge is uniformly spread. The surface charge densities are $\sigma_{1}=+2.00$ $\mu \mathrm{C} / \mathrm{m}^{2}, \sigma_{2}=+4.00 \mu \mathrm{C} / \mathrm{m}^{2}$, and $\sigma_{3}=-5.00 \mu \mathrm{C} / \mathrm{m}^{2}$, and... | 1 |
Which part of the arousal system controls the body's biological clock? | 0 |
14. The increasing order of the atomic radii of the following elements is:
(i) $\mathrm{C}$
(ii) $\mathrm{O}$
(iii) $\mathrm{F}$
(iv) $\mathrm{Cl}$
(v) $\mathrm{Br}$
(a) (ii) $<$ (iii) $<$ (iv) $<$ (i) $<$ (v)
(b) (iv) $<$ (iii) $<$ (ii) $<$ (i) $<$ (v)
(c) (iii) $<$ (ii) $<$ (i) $<$ (iv) $<$ (v)
(d) (i) $<$ (ii) $<$ ... | 1 |
What tools are used to carve pencil lead? | 0 |
Best places to eat in Chennai? | 0 |
What is Greece ranked in the EU in the production of marble? | 0 |
What is beauty? | 0 |
37. Galvanisation is applying a coating of
(a) Cr
(b) Cu
(c) Zn
(d) Pb (IIT 2016 Main) | 1 |
What are civil laws and what are some examples? | 0 |
Example 45.1
The mean free path of conduction electrons in copper is about $4 \times 10^{-8} \mathrm{~m}$. For a copper block, find the electric field which can give, on an average, $1 \mathrm{eV}$ energy to a conduction electron. | 1 |
3. A value of $b$ for which the equations $x^2+b x-1=0$, $x^2+x+b=0$ have one root in common is
(2011)
(a) $-\sqrt{2}$
(b) $-i \sqrt{3}$
(c) $i \sqrt{5}$
(d) $\sqrt{2}$ | 1 |
Was Joseph Goebbels a drug addict? I know Hitler was. | 0 |
Can we see a car moving at the speed of light? | 0 |
77. Match the conics in Column I with the statements/expressions in Column II.
| | Column I | Column II |
| :--- | :--- | :--- |
| A. | Circle | p. The locus of the point $(\mathrm{h}, \mathrm{k})$ for which the line $h x+k y=1$ touches the circle $x^{2}+y^{2}=4$ |
| B. | Parabola | q. Points $\mathrm{z}$ is the c... | 1 |
How can a students start learning c programming? | 0 |
What are some good ps4 games? | 0 |
(iii) interaction between dispersed phase and dispersion medium? | 1 |
Is Quora only for smart people? | 0 |
Is Alcoholics Anonymous a cult? | 0 |
What is the best way to save/ invest money? | 0 |
3.39. Demonstrate that the potential of the field generated by a dipole with the electric moment $p$ (Fig. 3.4) may be represented as $\varphi=\operatorname{pr} / 4 \pi \varepsilon_{0} r^{3}$, where $\mathbf{r}$ is the radius vector. Using this expression, find the magnitude of the electric field strength vector as a f... | 1 |
26. The geometries of the ammonia complexes of $\mathrm{Ni}^{2+}, \mathrm{Pt}^{2+}$ and $\mathrm{Zn}^{2+}$ respectively, are
(a) octahedral, square planar and tetrahedral
(b) square planar, octahedral and tetrahedral
(c) tetrahedral, square planar and octahedral
(d) octahedral, tetrahedral and square planar | 1 |
Where are cases usually argued? | 0 |
What can invalidate the rule of law? | 0 |
3. One of the possible argument of complex number $i\left(z_{1} / z_{2}\right)$
a. $\frac{\pi}{2}$
b. $-\frac{\pi}{2}$
c. 0
d. none of these | 1 |
(b) $x$-coordinate of particle at $t=3 \mathrm{~s}$ | 1 |
25. On the interval $[0,1]$, the function $x^{25}(1-x)^{75}$ takes its maximum value at the point
(a) 0
(b) $1 / 4$
(c) $1 / 2$
(d) $1 / 3$ | 1 |
What is Laird's job title? | 0 |
Why the countries fail to move together fight against terrorism? | 0 |
28. For the chemical reaction $3 X(\mathrm{~g})+Y(\mathrm{~g}) \rightleftharpoons X_{3} Y(\mathrm{~g})$, the amount of $X_{3} Y$ at equilibrium is affected by
(a) temperature and pressure
(b) temperature only
(c) pressure only
(d) temperature, pressure and catalyst | 1 |
Can I get Delhi as a job location after clearing the GATE exam with a high rank? | 0 |
4. Which of the following quantities related to an electron has a finite upper limit?
(a) Mass
(b) Momentum
(c) Speed
(d) Kinetic energy | 1 |
What thing mostly influences your choices about your career? | 0 |
How is oscillation defined? Does it have something to do with a stable equilibrium point. | 0 |
What is the best advice you received in college? | 0 |
How does it benefit you to answer questions on Quora? | 0 |
Which is the best phone to buy under Rs.6000? | 0 |
(c) Calculate the stored energy in the electric field before and after the process. | 1 |
My dating life made me depressed and feel hopeless I took Prozac and started to feel better. It's been 4.5 weeks and now feel a bit hopeless. Why? | 0 |
33. What is the decreasing order of strength of the bases $\mathrm{OH}^{-}, \mathrm{NH}_{2}^{-}, \mathrm{HC} \equiv \mathrm{C}^{-}$and $\mathrm{CH}_{3} \mathrm{CH}_{2}^{-}$?
(a) $\mathrm{CH}_{3}-\mathrm{CH}_{2}^{-}>\mathrm{NH}_{2}^{-}>\mathrm{H}-\mathrm{C} \equiv \mathrm{C}^{-}>\mathrm{OH}^{-}$
(b) $\mathrm{H}-\mathr... | 1 |
13. A block rests on a rough inclined plane making an angle of $30^{\circ}$ with horizontal. The coefficient of static friction between the block and inclined plane is 0.8 . If the frictional force on the block is $10 \mathrm{~N}$, the mass of the block in $\mathrm{kg}$ is ( $g=10 \mathrm{~m} / \mathrm{s}^{2}$ )
(a) 2.... | 1 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.