Question stringlengths 2 3k | label int64 0 1 |
|---|---|
5. $\left[\begin{array}{cc}2 & -2 \\ 4 & 3\end{array}\right]$ | 1 |
Career Advice: What are the career options after BCA? | 0 |
How many beds are available in all of Hyderabad's hospitals and nursing homes combined? | 0 |
5. $\lim _{x \rightarrow 0} \frac{e^{x}+e^{-x}-2}{x^{2}}$ | 1 |
3. The solution set of inequation $g(x)\left(\cos ^{-1} x-\sin ^{-1} x\right) \leq 0$
a. $\left[-1, \frac{1}{\sqrt{2}}\right]$
b. $\left[\frac{1}{\sqrt{2}}, 1\right]$
c. $\left[0, \frac{1}{\sqrt{2}}\right]$
d. $\left(0, \frac{1}{\sqrt{2}}\right]$ | 1 |
Is whey protein good? | 0 |
What is callback function in AngularJs? | 0 |
47.$ The temperature of 2.00 mol of an ideal monatomic gas is raised 15.0 K at constant volume. What are (a) the work $W$ done by the gas, (b) the energy transferred as heat $Q$, (c) the change $\Delta E_{\text {int }}$ in the internal energy of the gas, and (d) the change $\Delta K$ in the average kinetic energy per a... | 1 |
What is the name of the government structure created for New Delhi that is comprised of ten representatives? | 0 |
What are some ways to make a soap cap? | 0 |
64. Let $\vec{r}, \vec{a}, \vec{b}$ and $\vec{c}$ be four non-zero vectors such that $\vec{r} \cdot \vec{a}=0,|\vec{r} \times \vec{b}|=|\vec{r}||\vec{b}|$ and $|\vec{r} \times \vec{c}|=|\vec{r}||\vec{c}|$. Then $[a b c$ ] is equal to
a. $|a||b||c|$
b. $-|a||b||c|$
c. 0
d. none of these | 1 |
17. If $a, b, c, d \in R^{+}$and $a, b, c, d$ are in H.P., then
a. $a+d>b+c$
b. $a+b>c+d$
c. $a+c>b+d$
d. none of these | 1 |
(d) $\mathrm{N}_2 \mathrm{H}_4(\mathrm{l})+2 \mathrm{H}_2 \mathrm{O}_2(\mathrm{l}) \rightarrow \mathrm{N}_2(\mathrm{~g})+4 \mathrm{H}_2 \mathrm{O}$ (l) | 1 |
When was the 'education through recreation" applied to childhood? | 0 |
Did the Irish Parliamentary Party ever adopt the Parnellite model? | 0 |
What should I do if the crime rate in my city has increased drastically and the police and government are not taking any action? Is there any way a common man can connect to the government officials? | 0 |
27. When a salt of a weak acid and a weak base is dissolved in water at $25^{\circ} \mathrm{C}$, the pH of the resulting solution will always
(a) be 7
(b) be greater than 7
(c) be less than 7
(d) depend upon $K_{\mathrm{a}}$ and $K_{\mathrm{b}}$ values | 1 |
Ex. 21. A bottle of commercial sulphuric acid (density $1.787 \mathrm{~g} / \mathrm{mL}$ ) is labelled as $86 \%$ by weight. What is the molarity of the acid? What volume of the acid has to be used to make 1 litre of $0 \cdot 2 \mathrm{M} \mathrm{H}_{2} \mathrm{SO}_{4}$ ? | 1 |
How can I lose weight in a span of 2-3 months? | 0 |
24. The radius of which of the following orbit is same as that of the first Bohr's orbit of hydrogen atom?
(a) $\mathrm{He}^{+}(n=2)$
(b) $\mathrm{Li}^{2+}(n=2)$
(c) $\operatorname{Li}^{2+}(n=3)$
(d) $\operatorname{Be}^{3+}(n=2)$ | 1 |
21. Which one of the following molecules is expected to exhibit diamagnetic behaviour ?
(a) $\mathrm{C}_{2}$
(b) $\mathrm{N}_{2}$
(c) $\mathrm{O}_{2}$
(d) $\mathrm{S}_{2}$ | 1 |
What is a notable piece of surviving Late Middle Age royal plate? | 0 |
9.26 A mopte peraon has been taing apectackes of power -1.0 dboptre for distant vistoen. During old age he also nerds to use seprarate reading glass of power +2.0 dsoptres. Explain what may have happened. | 1 |
43. Which of the following properties of liquids increases with the increase in temperature?
(a) Vapour pressure
(b) Surface tension
(c) Viscosity
(d) None of these | 1 |
Which is better as a company, Apple or Google? | 0 |
How many people were there in the 13 colonies? | 0 |
16. Consider the following reaction and statements:
[Main 2018]
$\left[\mathrm{Co}\left(\mathrm{NH}_{3}\right)_{4} \mathrm{Br}_{2}\right]^{+}+\mathrm{Br}^{-} \rightarrow\left[\mathrm{Co}\left(\mathrm{NH}_{3}\right)_{3} \mathrm{Br}_{3}\right]+\mathrm{NH}_{3}$
(I) Two isomers are produced if the reactant complex ion i... | 1 |
Where is the best place to get Cabbage Patch doll clothes in the US? | 0 |
What do you think about ban on Rs. 500 and Rs. 1000 currency notes? | 0 |
9. Among the following, the essential amino acid is :
[Main Online April 8, 2017]
(a) Alanine
(b) Valine
(c) Aspartic acid
(d) Serine | 1 |
What did defeating Napoleon leave Britain without? | 0 |
(a) the acceleration and | 1 |
What are some of the best soft rock songs ever? | 0 |
30. The carboxyl functional group $(-\mathrm{COOH})$ is present in
(a) picric acid
(b) barbituric acid
(c) ascorbic acid
(d) aspirin | 1 |
Cronyism is also when people are selected from a closed and exclusive social network, which is called what? | 0 |
Example 6.9. Find the coefficient of $x^{k}$ in $1+(1+x)$ $+(1+x)^{2}+\cdots+(1+x)^{n}(0 \leq k \leq n)$. | 1 |
Do you love your job as a programmer? Is every challenging thing enjoyable ? I'm a graphic designer and I love it. Will I love programming ? | 0 |
What does kleptocracy mean? | 0 |
Can you pay users to click in-app ads? | 0 |
- Example 1.72 Find the distance of the point $(1,3)$ from the line $2 x-3 y+9=0$ measured along a line $x-y+1=0$. | 1 |
What are the most common traffic convictions in Arkansas, and how does the severity of the convictions differ in Ohio? | 0 |
23. Which one among the following pairs of ions cannot be separated by $\mathrm{H}_{2} \mathrm{~S}$ in dilute hydrochloric acid?
(a) $\mathrm{Bi}^{3+}, \mathrm{Sn}^{4+}$
(b) $\mathrm{Al}^{3+}, \mathrm{Hg}^{2+}$
(c) $\mathrm{Zn}^{2+}, \mathrm{Cu}^{2+}$
(d) $\mathrm{Ni}^{2+}, \mathrm{Cu}^{2+}$ | 1 |
22. For the gas phase reaction $\mathrm{C}_2 \mathrm{H}_4+\mathrm{H}_2 \rightleftharpoons \mathrm{C}_2 \mathrm{H}_6(\Delta H=-32.7 \mathrm{kcal})$ carried out in a vessel, the equilibrium concentration of $\mathrm{C}_2 \mathrm{H}_4$ can be increased by
(a) increasing the temperature
(b) decreasing the pressure
(c) remo... | 1 |
Do you think that Lebron James takes PEDs? | 0 |
29. When equal volumes of two substances are mixed, the specific gravity of the mixture is 4 . When equal weights of the same substances are mixed, the specific gravity of the mixture is 3 . The specific gravities of the two substances could be
(a) 6 and 2
(b) 3 and 4
(c) 2.5 and 3.5
(d) 5 and 3 | 1 |
What are the key benefits of Cloud computing? | 0 |
1. A student plots a graph from his readings on the determination of Young modulus of a metal wire but forgets to put the labels (figure 14-Q4). The quantities on $X$ and $Y$-axes may be respectively
(a) weight hung and length increased
(b) stress applied and length increased
(c) stress applied and strain developed
... | 1 |
What are some good ways to insult an atheist? | 0 |
31. With respect to hypochlorite, chlorate and perchlorate ions, choose the correct statement(s).
(a) The hypochlorite ion is the strongest conjugate base.
(b) The molecular shape of only chlorate ion is influenced by the lone pair of electrons of $\mathrm{Cl}$.
(c) The hypochlorite and chlorate ions disproportionat... | 1 |
12. In the below circuit, the current in each resistance is
(2017 Main)
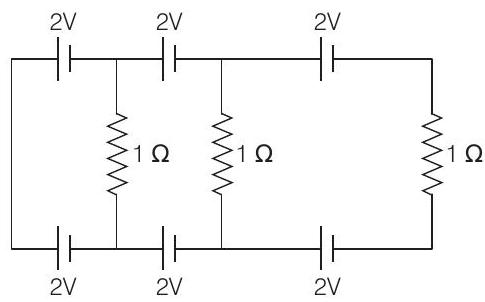
(a) $0.25 \mathrm{~A}$
(b) $0.5 \mathrm{~A}$
(c) $0 \mathrm{~A}$
(d) $1 \mathrm{~A}$ | 1 |
19. A boat of length $10 \mathrm{~m}$ and mass $450 \mathrm{~kg}$ is floating without motion in still water. A man of mass $50 \mathrm{~kg}$ standing at one end of it walks to the other end of it and stops. The magnitude of the displacement of the boat in metres relative to ground is
(a) zero
(b) $1 \mathrm{~m}$
(c) $2... | 1 |
(ii) 1 mole of carbon is burnt in 16 g of dioxygen. | 1 |
What can I do to reduce the effects of smoking? | 0 |
Would Quora be greatly improved without so many stupid polarizing political questions? | 0 |
7.20 The oxygen molecule has a mass of $5.30 \times 10^{-26} \mathrm{~kg}$ and a moment of inertia of $1.94 \times 10^{-46} \mathrm{~kg} \mathrm{~m}^{2}$ about an axis through its centre perpendicular to the lines joining the two atoms. Suppose the mean speed of such a molecule in a gas is $500 \mathrm{~m} / \mathrm{s}... | 1 |
What emotion do many cultures discourage in women? | 0 |
What are the practical aspects of strategic management? | 0 |
2. Current in an $\mathrm{AC}$ circuit is given by $i=3 \sin \omega t+4 \cos \omega t$, then
(a) rms value of current is $5 \mathrm{~A}$
(b) mean value of this current in positive one-half period will be $\frac{6}{\pi}$
(c) if voltage applied is $V=V_{m} \sin \omega t$, then the circuit may contain resistance and ca... | 1 |
9. The rate of a reaction quadruples when the temperature changes from 300 to $310 \mathrm{~K}$. The activation energy of this reaction is : (Assume activation energy and pre-exponential factor are independent of temperature; $\ln 2=0.693 ; \mathrm{R}=8.314 \mathrm{~J} \mathrm{~mol}^{-1} \mathrm{~K}^{-1}$
(a) $107.2... | 1 |
25. A uniform chain of length $\pi r$ lies inside a smooth semicircular tube $A B$ of radius $r$. Assuming a slight disturbance to start the chain in motion, the velocity with which it will emerge from the end $B$ of the tube will be
 +1.5
(b) -1.5
(c) +6.67
(d) -6.67 | 1 |
Where can I found efficient painters in Sydney? | 0 |
How can I become a Top Writer on Quora? | 0 |
If an editor can't achieve an exact reproduction of an authors work, what should be the goal? | 0 |
3. An illuminated object is placed on the principal axis of a converging lens so that a real image is formed on the other side of the lens. If the object is shifted a little, (a) the image will be shifted simultaneously with the object (b) the image will be shifted a little later than the object (c) the image will be s... | 1 |
5. If a long cylindrical conductor carries a steady current parallel to its length, then
(a) the electric field along the axis is zero
(b) the magnetic field along the axis is zero
(c) the magnetic field outside the conductor is zero
(d) the electric field outside the conductor is zero | 1 |
28. How many compounds are aromatic :
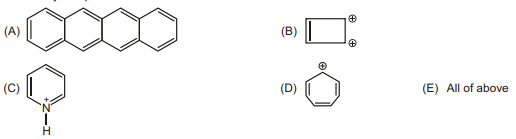 | 1 |
3. The increasing order of reactivity of the following compounds towards aromatic electrophilic substitution reaction is:
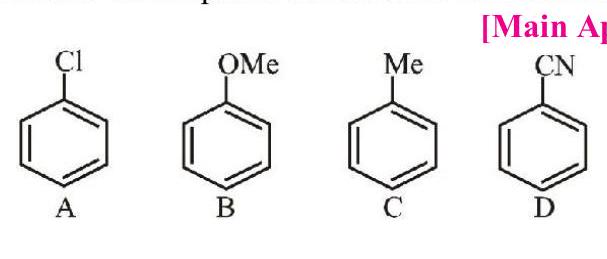
[Main April 9, 2019 (I)]
(a) D $<$ A $<$ C $<$ B
(b) B $<$ C $<$... | 1 |
What did Christopher Columbus discover in 1492? | 0 |
What would be an example of mild shame? | 0 |
I am far away from my girlfriend. What should I have to gift her on her birthday? | 0 |
102. The Pyrex glass mirror in a telescope has a diameter of 170 in. The temperature ranges from $-16^{\circ} \mathrm{C}$ to $32^{\circ} \mathrm{C}$ on the location of the telescope. What is the maximum change in the diameter of the mirror, assuming that the glass can freely expand and contract? | 1 |
9. Which of the following are not aromatic :
(A) Benzene
(B) Cyclo-octatetrarenyl dianion
(C) Tropyllium cation
(D) Cyclopentadienyl cation | 1 |
13. Marsh gas mainly contains
(a) $\mathrm{C}_{2} \mathrm{H}_{2}$
(b) $\mathrm{CH}_{4}$
(c) $\mathrm{H}_{2} \mathrm{~S}$
(d) $\mathrm{CO}$ | 1 |
What is the best book for image processing? | 0 |
4. The locus of the midpoint of a chord of the circle $x^{2}+y^{2}$ $=4$ which subtends a right angle at the origin is
a. $x+y=2$
b. $x^{2}+y^{2}=1$
c. $x^{2}+y^{2}=2$
d. $x+y=1$
(IIT-JEE, 1984) | 1 |
4. For what values of $x$ : $[\left.\begin{array}{lll}1 & 2 & 1\end{array}\right]\left[\begin{array}{lll}1 & 2 & 0 \\ 2 & 0 & 1 \\ 1 & 0 & 2\end{array}\right]\left[\begin{array}{l}0 \\ 2 \\ x\end{array}\right]=\mathrm{O}$ ? | 1 |
What is the difference between boeing and airbus? | 0 |
19. In a face-centred cubic cell, an atom at the face centre is shared by
(a) 6 unit cells
(b) 1 unit cell
(c) 4 unit cells
(d) 2 unit cells | 1 |
What is the difference between ";" and ","? | 0 |
What is diamond grading report? | 0 |
(ii) $\left[\mathrm{Co}(\mathrm{en})_3\right] \mathrm{Cl}_3$ | 1 |
Which tool is better in automation testing: QTP or Selenium IDE, for the future? | 0 |
45. If $2 \sin ^{-1} x=\sin ^{-1}\left(2 x \sqrt{1-x^{2}}\right)$, then
a. $[-1,1]$
b $\left[-\frac{1}{\sqrt{2}}, 1\right]$
c. $\left[-\frac{1}{\sqrt{2}}, \frac{1}{\sqrt{2}}\right]$
d. none of these | 1 |
Describe a situation when you faced a difficult problem and you were able to solve it? | 0 |
What treaty was signed in 1936? | 0 |
What type of deposits are found at Sambhar? | 0 |
In the 1840s, Töpffer wrote theories about whose work? | 0 |
20. The value of $\log _{10} K$ for a reaction $A \rightleftharpoons B$ is
(Given : $\Delta_{r} H_{298 \mathrm{~K}}^{\circ}=-54.07 \mathrm{~kJ} \mathrm{~mol}^{-1}, \Delta_{r} S_{298 \mathrm{~K}}^{\circ}$
$=10 \mathrm{JK}^{-1} \mathrm{~mol}^{-1}$ and $R=8.314 \mathrm{JK}^{-1} \mathrm{~mol}^{-1}$;
$2.303 \times 8.314 ... | 1 |
9.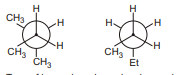
Type of isomerism shown by given pairs is :
(A) conformatioinal
(B) Positional
(C) Nuclear
(D) Metamerism | 1 |
The Spring Festival which is held each year in Delhi is also known by what name? | 0 |
19. The species present in solution when $\mathrm{CO}_2$ is dissolved in water are
(a) $\mathrm{CO}_2, \mathrm{H}_2 \mathrm{CO}_3, \mathrm{HCO}_3^{-}, \mathrm{CO}_3^{2-}$
(b) $\mathrm{H}_2 \mathrm{CO}_3, \mathrm{CO}_3^{2-}$
(c) $\mathrm{CO}_3^{2-}, \mathrm{HCO}_3^{-}$
(d) $\mathrm{CO}_2, \mathrm{H}_2 \mathrm{CO}_3... | 1 |
Should I start my start-up in Boulder or Denver? | 0 |
What's the difference between WiFi and Ethernet? | 0 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.