Question stringlengths 2 3k | label int64 0 1 |
|---|---|
How do I evaluate the integral [math]\displaystyle\int \frac{\sin x}{\sin 4x}\,dx[/math]? | 0 |
Is Indian English more formal than other English dialects? | 0 |
13. Sodium thiosulphate is prepared by
(a) reducing $\mathrm{Na}_{2} \mathrm{SO}_{4}$ solution with $\mathrm{H}_{2} \mathrm{~S}$
(b) boiling $\mathrm{Na}_{2} \mathrm{SO}_{3}$ solution with $\mathrm{S}$ in alkaline medium
(c) neutralising $\mathrm{H}_{2} \mathrm{~S}_{2} \mathrm{O}_{3}$ solution with $\mathrm{NaOH}$
... | 1 |
11. Sodium Carbonate cannot be used in place of $\left(\mathrm{NH}_{4}\right)_{2} \mathrm{CO}_{3}$ for the identification of $\mathrm{Ca}^{2+}, \mathrm{Ba}^{2+}$ and $\mathrm{Sr}^{2+}$ ions (in group $\mathrm{V}$ ) during mixture
analysis because :
a) $\mathrm{Mg}^{2+}$ ions will also be precipitated.
(b) Concentrat... | 1 |
18. Higher order $(>3)$ reactions are rare due to :
(a) shifting of equilibrium towards reactants due to elastic collisions
(b) loss of active species on collision
(c) low probability of simultaneous collision of all the reacting species
(d) increase in entropy and activation energy as more molecules are involved | 1 |
What happens to employees after your startup gets acquired? | 0 |
5. Let $f(x)=\left|\begin{array}{ccc}x^{3} & \sin x & \cos x \\ 6 & -1 & 0 \\ p & p^{2} & p^{3}\end{array}\right|$, where $p$ is a constant. Then $\frac{d^{3}}{d x^{3}}(f(x))$ at $x=0$ is
a. $p$
b. $p-p^{3}$
c. $p+p^{3}$
d independent of $p$
(IIT-JEE, 1997) | 1 |
4. Let $n_{p}$ and $n_{e}$ be the numbers of holes and conduction electrons in an extrinsic semiconductor.
(a) $n_{p}>n_{e}$
(b) $n_{p}=n_{e}$
(c) $n_{p}<n_{e}$
(d) $n_{p} \neq n_{e}$ | 1 |
10. $f(x)$ is equal to
a. $4 x^{2 / 3}\left(\frac{1+x}{1-x}\right)^{1 / 3}$
b. $x^{1 / 3}\left(\frac{1-x}{1+x}\right)^{1 / 3}$
c. $x^{2 / 3}\left(\frac{1-x}{1+x}\right)^{1 / 3}$
d $x\left(\frac{1+x}{1-x}\right)^{1 / 3}$ | 1 |
Which is the best commerce college for B.Com in Madhya Pradesh? | 0 |
49. ${ }_{13}^{27} \mathrm{Al}$ is a stable isotope. ${ }_{13}^{29} \mathrm{Al}$ is expected to disintegrate by
(a) $\alpha$-emission
(b) $\beta$-emission
(c) positron emission
(d) proton emission | 1 |
What is the biggest ocean on the earth? | 0 |
Is it true that fat people feel less cold than thin people? | 0 |
6. The number of roots of the equation $g(x)=1$ is
a. 2
b. 1
c. 3
d. 0
$c^{2}$
Let $f: N \rightarrow R$ be a function satisfying the following conditions, $f(1)=1 / 2$ and $f(1)+2, f(2)+3, f(3)+\cdots+n f(n)=n(n+1)$, $f(n)$ for $n \geq 2$. | 1 |
Who authored the Summa Theologica? | 0 |
What is a good book on Greek mythology? | 0 |
Which credit card will give access to the largest number of airport lounges? | 0 |
61. Two players $P_{1}$ and $P_{2}$ play a series of ' $2 n$ ' games. Each game can result in either a win or a loss for $P_{1}$. The total number of ways in which $P_{1}$ can win the series of these games is equal to
a. $\frac{1}{2}\left(2^{2 n}-{ }^{2 n} C_{n}\right)$
b. $\frac{1}{2}\left(2^{2 n}-2 \times{ }^{2 n} C_... | 1 |
12. A disc of radius $R$ is pivoted at its rim. The period for small oscillations about an axis perpendicular to the plane of disc is
(a) $2 \pi \sqrt{\frac{R}{g}}$
(b) $2 \pi \sqrt{\frac{2 R}{g}}$
(c) $2 \pi \sqrt{\frac{2 R}{3 g}}$
(d) $2 \pi \sqrt{\frac{3 R}{2 g}}$ | 1 |
What type of University would Yale fall under? | 0 |
Can a black person have white privilege? | 0 |
7.16 Can $\mathrm{PCl}_5$ act as an oxidising as well as a reducing agent? Justify. | 1 |
How does a question on Quora become a "Best Question"? | 0 |
Example 12.2 In a Geiger-Marsden experiment, what is the distance of closest approach to the nucleus of a $7.7 \mathrm{MeV} \alpha$-particle before it comes momentarily to rest and reverses its direction? | 1 |
15. At $0.3 \mathrm{~V}$ and $0.7 \mathrm{~V}$, the diodes $\mathrm{Ge}$ and $\mathrm{Si}$ become conductor respectively. In given figure, if ends of diode Ge overturned, the change in potential $V_{0}$ will be
 $\mathrm{CHCl}_{3}$
(b) $\mathrm{Cl}_{3} \mathrm{CCHO}$
(c) $\mathrm{CCl}_{4}$
(d) $\mathrm{CICH}_{2} \mathrm{CH}_{2} \mathrm{Cl}$ | 1 |
How many number of customer Eat24 does have? | 0 |
9. For a reaction, $\mathrm{A}(\mathrm{g}) \rightarrow \mathrm{A}(\mathrm{l}) ; \Delta \mathrm{H}=-3 \mathrm{RT}$. The correct statement for the reaction is :
(a) $\Delta \mathrm{H}=\Delta \mathrm{U} \neq \mathrm{O}$
(b) $\Delta \mathrm{H}=\Delta \mathrm{U}=\mathrm{O}$
(c) $|\Delta \mathrm{H}|<|\Delta \mathrm{U}|$
(... | 1 |
12. If the curve $C$ in the $x y$ plane has the equation $x^{2}+x y+$ $y^{2}=1$, then the fourth power of the greatest distance of a point on $C$ from the origin, is | 1 |
How do I cope with extreme anxiety? | 0 |
Is yeshyip.com legit? | 0 |
61 Spatial separation between two events. For the passing reference frames of Fig. 37-25, events $A$ and $B$ occur with the following spacetime coordinates: according to the unprimed frame, $\left(x_{A}, t_{A}\right)$ and $\left(x_{B}, t_{B}\right)$; according to the primed frame, $\left(x_{A}^{\prime}, t_{A}^{\prime}\... | 1 |
Can I quit my job via email effective immediately if I only worked there one week? | 0 |
59. When $3.06 \mathrm{~g}$ of solid $\mathrm{NH}_{4} \mathrm{HS}$ is introduced into a two litre evacuated flask at $27^{\circ} \mathrm{C}, 30 \%$ of the solid decomposes into gaseous ammonia and hydrogen sulphide. (i) Calculate $K_{c}$ and $K_{p}$ for the reaction at $27^{\circ} \mathrm{C}$. (ii) What would happen to... | 1 |
What are the differences in sexual identity and sexual behaviour? | 0 |
31. The complex ion which has no ' $d$ ' electron in the central metal atom is
[2001S]
(a) $\left[\mathrm{MnO}_{4}\right]$
(b) $\left[\mathrm{Co}\left(\mathrm{NH}_{3}\right)_{6}\right]^{3+}$
(c) $\left[\mathrm{Fe}(\mathrm{CN})_{6}\right]^{3-}$
(d) $\left[\mathrm{Cr}\left(\mathrm{H}_{2} \mathrm{O}\right)_{6}\right]... | 1 |
84. The magnetic field at a point inside a $2.0 \mathrm{mH}$ inductorcoil becomes 0.80 of its maximum value in $20 \mu$ s when the inductor is joined to a battery. Find the resistance of the circuit. | 1 |
Do you strain your eyes while working at an IT job? | 0 |
| 6 |  | | | | | | 1 |
Is the US the most racist country in The Americas? | 0 |
8. Two plane mirrors are inclined to each other at an angle $\theta$. A ray of light is reflected first at one mirror and then at the other. Find the total deviation of the ray. | 1 |
What are some things new employees should know going into their first day at Sealed Air? | 0 |
13. In the figure shown, water drains out through a small hole of a large tank.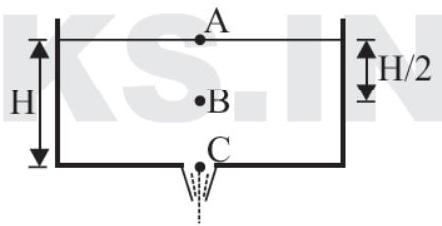
(a) pressure at the point C is greater than atmospheric
(b) pressure at the point B is $\rho \mathrm{g} \fra... | 1 |
What kind of temples were built for Hindu Gods? | 0 |
If energy cannot be created nor destroyed, only transferred, where did it come from? | 0 |
7. The correct sequence of amino acids present in the tripeptide given below is:
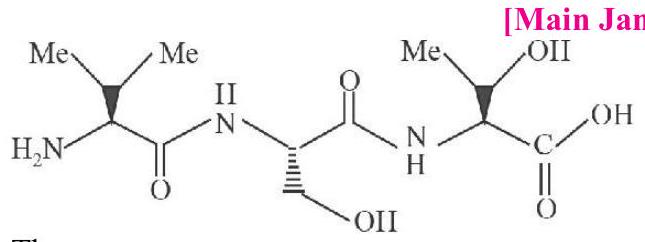
(a) Val - Ser - Thr
(b) $\mathrm{Thr}-\mathrm{Ser}-\mathrm{Va}$
(c) Leu - Ser - Val
(d) Thr - Ser - Leu | 1 |
How it feels to be in CYIENT as a structural engineer from IIT? | 0 |
What do Korean/Japanese/Chinese people think of Indians? | 0 |
25. In which of the following aqueous solutions, $\mathrm{H}_{2}$ and $\mathrm{O}_{2}$ are not liberated at cathode and anode respectively on electrolysis using inert electrodes?
(a) $\mathrm{H}_{2} \mathrm{SO}_{4}$ solution
(b) NaOH solution
(c) $\mathrm{Na}_{2} \mathrm{SO}_{4}$ solution
(d) $\mathrm{A... | 1 |
76. If $\alpha, \beta, \gamma$ are the angles of a triangle and the system of equations
$\cos (\alpha-\beta) x+\cos (\beta-\gamma) y+\cos (\gamma-\alpha) z=0$
$\cos (\alpha+\beta) x+\cos (\beta+\gamma) y+\cos (\gamma+\alpha) z=0$
$\sin (\alpha+\beta) x+\sin (\beta+\gamma) y+\sin (\gamma+\alpha) z=0$
has non-trivial... | 1 |
20. Consider the following reversible reaction,
$A(\mathrm{~g})+B(\mathrm{~g}) \rightarrow A B(\mathrm{~g})$
The activation energy of the backward reaction exceeds that of the forward reaction by $2 R T$ (in $\mathrm{J} \mathrm{mol}^{-1}$ ). If the pre-exponential factor of the forward reaction is 4 times that of the... | 1 |
What is the history behind pet therapy? | 0 |
42. Which among the following statement(s) is(are) true for the extraction of aluminium from bauxite?
(a) Hydrated $\mathrm{Al}_{2} \mathrm{O}_{3}$ precipitates, when $\mathrm{CO}_{2}$ is bubbled through a solution of sodium aluminate.
(b) Addition of $\mathrm{Na}_{3} \mathrm{AlF}_{6}$ lowers the melting point of al... | 1 |
6. In which of the following interval $f(x)$ is strictly increasing?
a. $(-\infty, \infty)$
b. $(-\infty, 0)$
c. $(0, \infty)$
d. None of these | 1 |
Around what time did Mass Culture emerge? | 0 |
What are some good psychological thriller movies? | 0 |
48. Amongst $\left[\mathrm{Ni}(\mathrm{CO})_{4}\right],\left[\mathrm{Ni}(\mathrm{CN})_{4}\right]^{2-}$ and $\left[\mathrm{NiCl}_{4}^{2-}\right]$
[1991 - 1 Mark]
(a) $\left[\mathrm{Ni}(\mathrm{CO})_{4}\right]$ and $\left[\mathrm{NiCl}_{4}^{2-}\right]$ are diamagnetic and $\left[\mathrm{Ni}(\mathrm{CN})_{4}\right]^{2-}... | 1 |
56. If $\alpha$ and $\beta$ are the roots of the equation $x^{2}+p x+1=0 ; \gamma, \delta$ are the roots of $x^{2}+q x+1=0$, then $q^{2}-p^{2}=(\alpha-\gamma)(\beta-\gamma)(\alpha+\delta)(\beta+\delta)$
(1978, 2M) | 1 |
Where was a notable hoard found near the former lands of the Byzantine Empire? | 0 |
What is an example of a language that is not restricted to properties of human language? | 0 |
6. If ' K ' is the total number of integers n between 1 and 10000 (both inclusive) such that n is divisible by $[\sqrt{\mathrm{n}}]$, then Here [] is G.I.F
(a) K is more than 198
(b) K is an even no.
(c) K is less than 300
(d) K is more than 200 | 1 |
109. Thirty-two players ranked 1 to 32 are playing in a knockout tournament. Assume that in every match between any two players, the better-ranked player wins, the probability that ranked 1 and ranked 2 players are winner and runner up, respectively, is
a. $16 / 31$
b. $1 / 2$
c. $17 / 31$
d. none of these | 1 |
What is a notable architectural feature of the basilica? | 0 |
83 Figure 12-84 shows a stationary arrangement of two crayon boxes and three cords. Box $A$ has a mass of 11.0 kg and is on a ramp at angle $\theta=30.0^{\circ}$; box $B$ has a mass of 7.00 kg and hangs on a cord. The cord connected to box $A$ is parallel to the ramp, which is frictionless. (a) What is the tension in t... | 1 |
39. Let the angle between two non-zero vectors $\mathbf{A}$ and $\mathbf{B}$ be $120^{\circ}$ and its resultant be $\mathbf{C}$.
(a) $C$ must be equal to $|A-B|$
(b) $C$ must be less than $|A-B|$
(c) $C$ must be greater than $|A-B|$
(d) $C$ may be equal to $|A-B|$ | 1 |
7. Find the condition that the line $A x+B y=1$ may be normal to the curve $a^{n-1} y=x^{n}$. | 1 |
Are animals killed in the process of farming vegetarian foods? | 0 |
11. The mass of a copper calorimeter is $40 \mathrm{~g}$ and its specific heat in SI units is $4.2 \times 10^{2} \mathrm{~J} \mathrm{~kg}^{-1}{ }^{\circ} \mathrm{C}^{-1}$. The thermal capacity is
(a) $4 \mathrm{~J}^{\circ} \mathrm{C}^{-1}$
(b) $18.6 \mathrm{~J}$
(c) $16.8 \mathrm{~J} / \mathrm{kg}$
(d) $16.8 \mathrm{~J... | 1 |
What is the best way to get rid of acne? | 0 |
When did Rhodesia become Zimbabwe? | 0 |
Who were the really decent guys from both sides of world war 2, even by today's standard? | 0 |
What is the difference between scheduled caste (SC) and scheduled tribe (ST) ? How government has classified tribes and caste into it? | 0 |
12. The work-function of a substance is $1.6 \mathrm{eV}$. The longest wavelength of light that can produce photoemission from the substance is
(a) $7750 \AA$
(b) $3875 \AA$
(c) $5800 \AA$
(d) $2900 \AA$ | 1 |
(a) velocity of B with respect to A ?, | 1 |
Where was the location origin of the Maurya and Gupta empires? | 0 |
Around what year did the great wave of church-building in stone occur? | 0 |
107. The number of three-digit numbers of the form $x y z$ such that $x<y$ and $z \leq y$ is
a. 276
b. 285
c. 240
d. 244 | 1 |
What is the best way to make non alcoholic ginger beer? | 0 |
Is there any medically mutatable serum that can grant humans incredible strength without side effects (like captain america)? | 0 |
(a) How much potential energy is stored in the electric field of this charged conductor? | 1 |
18. A resistance of $2 \Omega$ is connected across one gap of a meter-bridge (the length of the wire is $100 \mathrm{~cm}$ ) and an unknown resistance, greater than $2 \Omega$, is connected across the other gap. When these resistances are interchanged, the balance point shifts by $20 \mathrm{~cm}$. Neglecting any corre... | 1 |
Why does rabies affects some mammals, but not others? | 0 |
What specific Scala coding skills do I need to master in order to get a job as a Scala dev? | 0 |
What is the best online site to learn C programming by myself? | 0 |
2. The strengths of 5.6 volume hydrogen peroxide (of density $1 \mathrm{~g} / \mathrm{mL}$ ) in terms of mass percentage and molarity (M), respectively, are :
(Take molar mass of hydrogen peroxide as $34 \mathrm{~g} / \mathrm{mol}$ )
(a) 1.7 and 0.5
(b) 0.85 and 0.25
(c) 1.7 and 0.25
(d) 0.85 and 0.5 | 1 |
How will banning of Rs 500 and Rs 1000 notes affect the Indian stock market? | 0 |
7.40 List the uses of neon and argon gases. | 1 |
What mind blowing technologies exist in 2016 that most people don't know about? | 0 |
What is information systems? | 0 |
Why can't India won Olympic gold medals despite the fact that African countries are winning more medal than we are? | 0 |
What comes out of the vagina when girls have orgasm? | 0 |
22. Which of the following is the wrong statement
(a) $\mathrm{ONCl}$ and $\mathrm{ONO}^{-}$ are not isoelectronic.
(b) $\mathrm{O}_{3}$ molecule is bent
(c) Ozone is violet-black in solid state
(d) Ozone is diamagnetic gas. | 1 |
Who would win a fight between a male lion and a silver back gorilla? | 0 |
29. If a salt of a strong acid and a weak base hydrolyses appreciably, which of the following formulae is to be used to calculate degree of hydrolysis ' $X$ '?
(a) $X=\sqrt{\frac{K_{\mathrm{w}}}{K_{\mathrm{a}} \cdot a}}$
(b) $X=\sqrt{\frac{K_{\mathrm{w}}}{K_{\mathrm{b}} \cdot a}}$
(c) $X=\sqrt{\frac{K_{\mathrm{w}}}{K_{... | 1 |
12. In a quadrilateral $A B C D$, it is given that $A B \| C D$ and the diagonals $A C$ and $B D$ are perpendicular to each other. Show that $A D \cdot B C \geq A B \cdot C D$. | 1 |
Many people of which ethnicity moved to Malaysia, Singapore and the Caribbean? | 0 |
27. Compare the stability of carbocation which are formed when react with $\mathrm{AgNO}_{3}$.
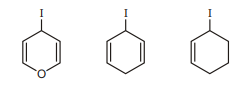
(A) a $>$ b $>$ c
(B) a $>$ c $>$ b
(C) b $>$ c $>$ a
(D) b $>$ a $>$ c | 1 |
Is the rake (fee) too high on fantasy sport sites? | 0 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.