Question stringlengths 2 3k | label int64 0 1 |
|---|---|
10. Let $g(x)=x f(x)$, where $f(x)=\left\{\begin{array}{ll}x \sin \frac{1}{x}, & x \neq 0 \\ 0, & x=0\end{array}\right.$. At $x=0$
a. $g$ is differentiable but $g^{\prime}$ is not continuous
b. $g$ is differentiable while $f$ is not
c. both $f$ and $g$ are differentiable
d. $g$ is differentiable and $g^{\prime}$ is con... | 1 |
2. $\lim _{x \rightarrow \infty} \sqrt{\frac{x+\sin x}{x-\cos x}}$ is equal to
a. 0
b. 1
c. -1
d. None of these | 1 |
I've almost 2 years of an experience as an ETL developer. What are the good consultancies in Bangalore to apply for jobs? | 0 |
Where do the terms "Middle Ages" and "Modern Ages" originate from? | 0 |
8. A particle moves along the parabolic path $x=y^{2}+2 y+2$ in such a way that $Y$-component of velocity vector remains $5 \mathrm{~ms}^{-1}$ during the motion. The magnitude of the acceleration of the particle is
(a) $50 \mathrm{~ms}^{-2}$
(b) $100 \mathrm{~ms}^{-2}$
(c) $10 \sqrt{2} \mathrm{~ms}^{-2}$
(d) $0.1 \math... | 1 |
How many people go to the beach in the US each year? | 0 |
How did Rey become proficient in the Force so quickly? | 0 |
58. State with balanced equations, what happens when propene is bubbled through a hot aqueous solution of potassium permanganate.
[1982 - 1 Mark] | 1 |
I want to build promotional animated video which explains what my Android app is about. What are the best sources which can help me make it? | 0 |
124. The equation of the straight line which passes through the point $(-4,3)$ such that the portion of the line between the axes is divided internally by the point in the ratio $5: 3$ is
a. $9 x-20 y+96=0$
b. $9 x+20 y=24$
c. $20 x+9 y+53=0$
d. none of these | 1 |
How do I become a motivational speaker? | 0 |
21. An open pipe is suddenly closed at one end with the result that the frequency of third harmonic of the closed pipe is found to be higher by $100 \mathrm{~Hz}$ then the fundamental frequency of the open pipe. The fundamental frequency of the open pipe is
(a) $200 \mathrm{~Hz}$
(b) $300 \mathrm{~Hz}$
(c) $240 \math... | 1 |
3. A moving coil galvanometer allows a full scale current of $10^{-4} \mathrm{~A}$. A series resistance of $2 \mathrm{M} \Omega$ is required to convert the above galvanometer into a voltmeter of range $0-5 \mathrm{~V}$. Therefore, the value of shunt resistance required to convert the above galvanometer into an ammeter ... | 1 |
3. Which of the following is not a colligative property?
(a) Vapour pressure
(b) Depression in f.p.
(c) Elevation in b.p.
(d) Osmotic pressure | 1 |
What are the best sites for C Programming language? | 0 |
Ex. 15. Calculate vapour pressure of a 5\% (by weight) solution of water in glycerol (mol. wt. 92\$\cdot\$1) at $100^{\circ} \mathrm{C}$, assuming Raoult's law to be valid and neglecting the vapour pressure of glycerol. | 1 |
2. If the volume of parallelopiped formed by the vectors $\hat{\mathbf{i}}+\lambda \hat{\mathbf{j}}+\hat{\mathbf{k}}, \hat{\mathbf{j}}+\lambda \hat{\mathbf{k}}$ and $\lambda \hat{\mathbf{i}}+\hat{\mathbf{k}}$ is minimum, then $\lambda$ is equal to
(a) $-\frac{1}{\sqrt{3}}$
(b) $\frac{1}{\sqrt{3}}$
(c) $\sqrt{3}$
(d) ... | 1 |
What is an iPad? | 0 |
How do you say 'blender' in Spanish? | 0 |
What is the term that the English use for the comics out of Japan? | 0 |
Who was Harold Wilson's Defense Secretary? | 0 |
13. SSM A particle moves so that its position (in meters) as a function of time (in seconds) is $\vec{r}=\hat{\mathrm{i}}+4 t^{\hat{j}} \hat{\mathrm{j}}+t \hat{\mathrm{k}}$. Write expressions for (a) its velocity and (b) its acceleration as functions of time. | 1 |
The India Gate commemorates how many soldiers of past wars? | 0 |
4. Let $z=\cos \theta+i \sin \theta$. Then, the value of $\sum_{m=1}^{15} \operatorname{Im}\left(z^{2 m-1}\right)$ at $\theta=2^{\circ}$ is
(a) $\frac{1}{\sin 2^{\circ}}$
(b) $\frac{1}{3 \sin 2^{\circ}}$
(c) $\frac{1}{2 \sin 2^{\circ}}$
(d) $\frac{1}{4 \sin 2^{\circ}}$
(2009) | 1 |
5. The correct structure of histidine in a strongly acidic solution $(\mathrm{pH}=$ 2) is :
[Main Jan. 12, 2019 (II)]
(a)
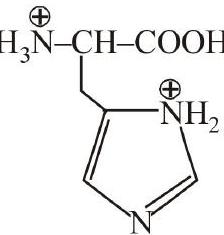
(b)
$ on the parabola $y^{2}=4 x$. | 1 |
23. Pure ammonia is placed in a vessel at a temperature where its dissociation constant is appreciable. At equilibrium,
(a) $K_p$ does not change significantly with pressure
(b) $\alpha$ does not change with pressure
(c) concentration of $\mathrm{NH}_3$ does not change with pressure
(d) concentration of $\mathrm{H}_2$ ... | 1 |
How can I make my laptop a WiFi hotspot if it doesn't have WiFi? | 0 |
There are two types of sleep called what? | 0 |
11. If $\lim _{x \rightarrow 0} \frac{((a-n) n x-\tan x) \sin n x}{x^{2}}=0$, where $n$ is nonzero real number, then $a$ is
a. 0
b $\frac{n+1}{n}$
c. $n$
d. $n+\frac{1}{n}$ | 1 |
10. Which one of the following oxides is neutral?
(a) $\mathrm{CO}$
(b) $\mathrm{SnO}_{2}$
(c) $\mathrm{ZnO}$
(d) $\mathrm{SiO}_{2}$ | 1 |
How can I earn money from Twitter? | 0 |
9. If one of the lines given by the equation $2 x^{2}+p x y+3 y^{2}=0$ coincide with one of those given by $2 x^{2}+q x y-3 y^{2}=0$ and the other lines represented by them be perpendicular, then
(a) $p=5$
(b) $p=-5$
(c) $q=-1$
(d) $q=1$ | 1 |
6. If $S_{n}=1^{2}-2^{2}+3^{2}-4^{2}+5^{2}-6^{2}+\cdots$, then
a. $S_{40}=-820$
b. $S_{2 n}>S_{2 n+2}$
c. $S_{51}=1326$
d. $S_{2 n+1}>S_{2 n-1}$ | 1 |
What changes do you want to see in India? Why? | 0 |
- Example 23 A charged particle carrying charge $q=1 \mu C$ moves in uniform magnetic field with velocity $v_{1}=10^{6} \mathrm{~m} / \mathrm{s}$ at angle $45^{\circ}$ with $x$-axis in the $x y$-plane and experiences a force $F_{1}=5 \sqrt{2} \mathrm{mN}$ along the negative $z$-axis. When the same particle moves with v... | 1 |
12. Glucose on prolonged heating with $\mathrm{HI}$ gives :
[Main 2018]
(a) $n$-Hexane
(b) 1- Hexene
(c) Hexanoic acid
(d) 6-iodohexanal | 1 |
11. The formation of which of the following polymers involves hydrolysis reaction?
[Main 2017]
(a) Nylon 6
(b) Bakelite
(c) Nylon 6,6
(d) Terylene | 1 |
47. Which of the following exists as covalent crystals in solid state?
(a) Iodine
(b) Silicon
(c) Sulphur
(d) Phosphorus | 1 |
(b) Find the transverse displacement of a point at $x=0.25 \mathrm{~m}$ at time $t=0.15 \mathrm{~s}$. | 1 |
Where can I get a private loan? | 0 |
Why do people laugh at other people when they hurt themselves? | 0 |
Which takes precedent when a law passed by Parliament conflicts with a law passed by Delhi's Legislative assembly? | 0 |
21. The locus of $z$ which lies in shaded region (excluding the boundaries) is best represented by
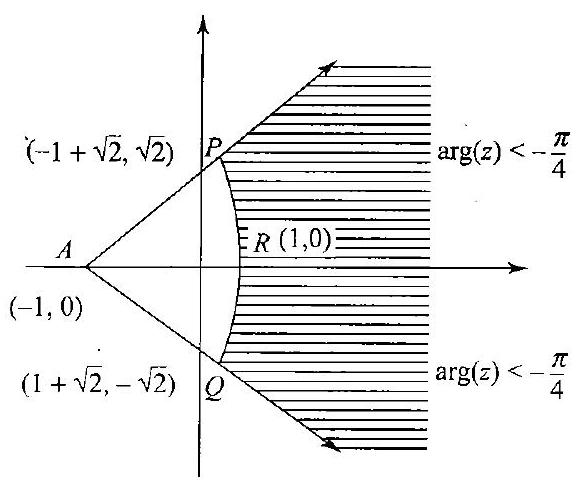
Fig. 2.47
a. $z:|z+1|>2$ and $\operatorname{larg}(z+1) \mid<\pi / 4$
b. $z:|z-1|>2$ and $... | 1 |
What does '^' mean in python? | 0 |
What group opposed the nominalists on the subject of universals? | 0 |
How do you test a product? | 0 |
12. Water of volume $2 \mathrm{~L}$ in a container is heated with a coil of $1 \mathrm{~kW}$ at $27^{\circ} \mathrm{C}$. The lid of the container is open and energy dissipates at rate of $160 \mathrm{~J} / \mathrm{s}$. In how much time temperature will rise from $27^{\circ} \mathrm{C}$ to $77^{\circ} \mathrm{C}$ ?
[... | 1 |
2. If $x^{m}$ occurs in the expansion of $\left(x+1 / x^{2}\right)^{2 n}$, then the coefficient of $x^{m}$ is
a. $\frac{(2 n)!}{(m)!(2 n-m)!}$
b. $\frac{(2 n)!3!3!}{(2 n-m)!}$
c. $\frac{(2 n)!}{\left(\frac{2 n-m}{3}\right)!\left(\frac{4 n+m}{3}\right)!}$
d. none of these | 1 |
What is MBoX mail? | 0 |
In the wake of the October resolution, what happened to the Russian Imperial Army? | 0 |
What's the process to start study of IAS? | 0 |
What is the scope of PhD in data analytics in India? | 0 |
14. If $y=\mathrm{A} e^{m x}+\mathrm{B} e^{n x}$, show that $\frac{d^{2} y}{d x^{2}}-(m+n) \frac{d y}{d x}+m n y=0$ | 1 |
Critics based in what movement find fault in objective realism? | 0 |
In which wars was France defeated? | 0 |
What are examples of various sources of earning for a photographer? | 0 |
How do people deal with boredom? | 0 |
How are animals and plants adaptated to survive in a tropical rainforest? | 0 |
What should I do to be interesting? | 0 |
1. If $\alpha={ }^{m} C_{2}$, then ${ }^{\alpha} C_{2}$ is equal to
a. ${ }^{m+1} C_{4}$
b. ${ }^{m-1} C_{4}$
c. $3^{m+2} C_{4}$
d. $3^{m+1} C_{4}$ | 1 |
What is endoscopy? | 0 |
What other traits are emotional dispositions similar to? | 0 |
Is positive thinking helpful? | 0 |
If dark and vacuum energy are created with expansion is there potential energy that transforms into these types of energy with expansion? | 0 |
17 SSM In Fig. 29-44, point $P_2$ is at perpendicular distance $R=$ 25.1 cm from one end of a straight wire of length $L=13.6 \mathrm{~cm}$ carrying current $i=0.693 \mathrm{~A}$. (Note that the wire is not long.) What is the magnitude of the magnetic field at $P_2$ ? | 1 |
Do people still worship the Greek gods? | 0 |
What is a good food item to get from India for your non-Indian colleagues? | 0 |
Example 7.19 Prove that $\left|\begin{array}{lll}x^{2} & x^{2}-(y-z)^{2} & y z \\ y^{2} & y^{2}-(z-x)^{2} & z x \\ z^{2} & z^{2}-(x-y)^{2} & x y\end{array}\right|$
$=(x-y)(y-z)(z-x)(x+y+z)\left(x^{2}+y^{2}+z^{2}\right)$. | 1 |
4. An urn contains three red balls and $n$ white balls. Mr. A draws two balls together from the urn. The probability that they have the same colour is $1 / 2$. Mr. $B$ draws one balls from the urn, notes its colour and replaces it. He then draws a second ball from the urn and finds that both balls have the same colour ... | 1 |
13. The correct set of four quantum numbers for the valence electrons of rubidium atom $(Z=37)$ is:
(a) $5,0,0,+\frac{1}{2}$
(b) $5,1,0,+\frac{1}{2}$
(c) $5,1,1,+\frac{1}{2}$
(d) $5,0,1,+\frac{1}{2}$ | 1 |
What is your review of Leonard Cohen? | 0 |
48. How will the concentration of $\mathrm{Ag}^{+}$in a saturated solution of AgCl diminish if such an amount of HCl is added to it that the concentration of the $\mathrm{Cl}^{-}$in the solution becomes equal to 0.03 mole/litre?
$$
\begin{aligned}
K_{\mathrm{sp}}(\mathrm{AgCl}) & =1.8 \times 10^{-10} . \\
& \left(\fra... | 1 |
7. If $f(x)=\operatorname{sgn}\left(x^{2}-a x+1\right)$ has maximum number of points of discontinuity, then
$\square \quad$ a. $a \in(2, \infty)$
b. $a \in(-\infty,-2)$
c. $a \in(-2,2)$
d None of these | 1 |
1. The line $2 x+y=1$ is tangent to the hyperbola $\frac{x^{2}}{a^{2}}-\frac{y^{2}}{b^{2}}$ $=1$. If this line passes through the point of intersection of the nearest directrix and the $x$-axis, then the eccentricity of the hyperbola is
(IIT-JEE, 2010) | 1 |
What is special about Ahmedabad city? | 0 |
What do the people of Bhutan think about India? | 0 |
(iii) Which atoms are indicated by the following configurations ?
(a) $[\mathrm{He}] 2 s^{1}$
(b) $[\mathrm{Ne}] 3 s^{2} 3 p^{3}$
(c) $[\mathrm{Ar}] 4 s^{2} 3 d^{1}$. | 1 |
What are the subjects for the UPSC exam? | 0 |
If there was a world war III around this time period what would most likely be the start of it? | 0 |
Ex. 23. Two liquids $A$ and $B$ form an ideal solution. At 300 K , the vapour pressure of a solution containing 1 mole of $A$ and 3 moles of $B$ is 550 mmHg . At the same temperature, if one mole of $B$ is added to this solution, the vapour pressure of the solution increases by 10 mmHg . Determine the vapour pressure o... | 1 |
9. Let $P Q$ and $R S$ be tangents at the extremities of the diameter $P R$ of a circle of radius $r$. If $P S$ and $R Q$ intersect at a point $X$ on the circumference of the circle, then $2 r$ equals
(a) $\sqrt{P Q \cdot R S}$
(b) $\frac{P Q+R S}{2}$
(c) $\frac{2 P Q \cdot R S}{P Q+R S}$
(d) $\sqrt{\frac{P Q^{2}+R S... | 1 |
Who inaugurated the city of New Delhi? | 0 |
2. A power transmission line feeds input power at $2300 \mathrm{~V}$ to a step-down transformer with its primary windings having 4000 turns. The output power is delivered at $230 \mathrm{~V}$ by the transformer. If the current in the primary of the transformer is $5 \mathrm{~A}$ and its efficiency is $90 \%$, the outpu... | 1 |
Ranthamb National Park is known by photographers as what? | 0 |
Can we create a time machine? | 0 |
1. Two fair dice are tossed. Let $x$ be the event that the first die shows an even number and $y$ be the event that the second die shows an odd number. The two events $x$ and $y$ are
a. mutually exclusive
b. independent and mutually exclusive
c. dependent
d. none of these | 1 |
76. Statement-1 : Acetate ion is more basic than the methoxide ion.
Statement-2 : The acetate ion is resonance stabilized | 1 |
Name the nineteenth century English atheist? | 0 |
10. Which one of the following statements about water is FALSE?
(a) There is extensive intramolecular hydrogen bonding in the condensed phase.
(b) Ice formed by heavy water sinks in normal water.
(c) Water is oxidized to oxygen during photosynthesis.
(d) Water can act both as an acid and as a base. | 1 |
Question 7:
Find the sum up to 20 terms in the geometric progression $0.15,0.015,0.0015 \ldots$ | 1 |
8. When stopping potential is applied in an experiment on photoelectric effect, no photocurrent is observed. This means that
(a) the emission of photoelectrons is stopped
(b) the photoelectrons are emitted but are re-absorbed by the emitter metal
(c) the photoelectrons are accumulated near the collector plate
(d) t... | 1 |
1. Calculate the fringe width.
(a) $0.63 \mathrm{~mm}$
(b) $1.26 \mathrm{~mm}$
(c) $1.67 \mathrm{~mm}$
(d) $2.2 \mathrm{~mm}$ | 1 |
What is ISO 9001:2000? | 0 |
13. The molecule that has minimum/no role in the formation of photochemical smog, is:
(a) $\mathrm{N}_{2}$
(b) $\mathrm{CH}_{2}=\mathrm{O}$
(c) $\mathrm{O}_{3}$
(d) $\mathrm{NO}$ | 1 |
The second wave of metrics focuses on what level of corruption? | 0 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.