Question stringlengths 2 3k | label int64 0 1 |
|---|---|
28. In an experiment to determine the specific heat of a metal, a $0.20 \mathrm{~kg}$ block of the metal at $150^{\circ} \mathrm{C}$ is dropped in a copper calorimeter (of water equivalent $0.025 \mathrm{~kg}$ ) containing $150 \mathrm{~cm}^{3}$ of water at $27^{\circ} \mathrm{C}$. The final temperature is $40^{\circ} ... | 1 |
What is the total area of New Delhi in square miles? | 0 |
10. Assertion : Light converges on a virtual object.
Reason : Virtual object is always behind a mirror. | 1 |
7. The equation of the transverse axis of the hyperbola $(x-3)^{2}+(y+1)^{2}=(4 x+3 y)^{2}$ is
a. $x+3 y=0$
b. $4 x+3 y=9$
c. $3 x-4 y=13$
d. $4 x+3 y=0$ | 1 |
5.11 What are lyophilic and lyophobic sols? Give one example of each type. Why are hydrophobic sols easily coagulated ? | 1 |
113. Two parabola have the focus $(3,-2)$. Their directrices are the $x$-axis, and the $y$-axis, respectively. Then the slope of their common chord is
a. -1
b. $-\frac{1}{2}$
c. $-\frac{\sqrt{3}}{2}$
d. none of these | 1 |
Which would be the best book to prepare for UGC-NET Paper 1? | 0 |
Who is a jobber? | 0 |
What is the best transcription app for the iPhone? | 0 |
What resources do developed countries have better access to? | 0 |
18. If $a, b$ and $c$ are non-zero real numbers, then $\Delta=\left|\begin{array}{lll}b^{2} c^{2} & b c & b+c \\ c^{2} a^{2} & c a & c+a \\ a^{2} b^{2} & a b & a+b\end{array}\right|$ is equal to
a. $a b c$
b. $a^{2} b^{2} c^{2}$
c. $b c+c a+a b$
d. none of these | 1 |
What are some tips to get over writer's block? | 0 |
21. The compound 1, 2-butadiene has
(a) only $s p$ hybridized carbon atoms
(b) only $s p^{2}$ hybridized carbon atoms
(c) both $s p$ and $s p^{2}$ hybridized carbon atoms
(d) $s p, s p^{2}$ and $s p^{3}$ hybridized carbon atoms | 1 |
What is your favourite quote? | 0 |
26. For all $x, x^{2}+2 a x+10-3 a>0$, then the interval in which $a$ lies is
a. $a<-5$
b. $-5<a<2$
c. $a>5$
d. $2<a<5$
(IIT-JEE, 2004) | 1 |
36. The rate constant for the first order decomposition of $\mathrm{N}_{2} \mathrm{O}_{5}$ dissolved in chloroform at $45^{\circ} \mathrm{C}$ is $3.1 \times 10^{-4} \mathrm{~min}^{-1}$.
$$ 2 \mathrm{~N}_{2} \mathrm{O}_{5} \rightarrow 4 \mathrm{NO}_{2}+\mathrm{O}_{2}
$$
(a) What is the rate of decomposition of $\mathr... | 1 |
How can you arrange the atoms in solids, liquids and gases? | 0 |
Ex. 24. Zinc sulphide crystallizes with zinc ions occupying one-half of the tetrahedral holes in a closest-packed array of sulphide ions. What is the formula of zinc sulphide? | 1 |
6. An AC voltage source of variable angular frequency $\boldsymbol{\omega}$ and fixed amplitude $V_{0}$ is connected in series with a capacitance $C$ and an electric bulb of resistance $R$ (inductance zero). When $\omega$ is increased,
(a) the bulb glows dimmer
(b) the bulb glows brighter
(c) total impedance of th... | 1 |
103. An unknown compound of carbon, hydrogen and oxygen contains $69.77 \%$ carbon and $11.63 \%$ hydrogen and has a molecular weight of 86. It does not reduce Fehling solution, but forms a bisulphite addition compound and gives a positive iodoform test. What are the possible structures for the unknown compound? | 1 |
11. Let $z$ and $\omega$ be two complex numbers such that $|z| \leq 1,|\omega|$ $\leq 1$ and $|z-i \omega|=|z-i \bar{\omega}|=2$ then $z$ equals
a. 1 or $i$
b. $i$ or $-i$
c. 1 or -1
d. $i$ or -1 | 1 |
When a comic panel has events that are not at the same time the panel is considered to be what? | 0 |
Can our body absorb systemic enzymes through our skin? | 0 |
What are some of the best Indian deodorants available in the market? | 0 |
(iii) $\{x: x$ is a student of Class XI of your school $\} \ldots\{x: x$ student of your school $\}$ | 1 |
Do SOME Afghans dislike the US for starting a war in their country? | 0 |
How is the procedure for making a passport? | 0 |
What is Newton's third law of motion with examples? | 0 |
29. Given the function $f(x)=\frac{a^{x}+a^{-x}}{2}$ (where $a>2$ ). Then
$f(x+y)+f(x-y)=$
a. $2 f(x) \cdot f(y)$
b. $f(x) . f(y)$
c. $\frac{f(x)}{f(y)}$
d. None of these | 1 |
16. The rusting of iron takes place as follows
$$
\begin{equation*} 2 \mathrm{H}^{+}+2 \mathrm{e}^{-}+1 / 2 \mathrm{O}_{2} \longrightarrow \mathrm{H}_{2} \mathrm{O}(1) ; E^{\circ}=+1.23 \mathrm{~V} \tag{2005S}
\end{equation*}
$$
$\mathrm{Fe}^{2+}+2 \mathrm{e}^{-} \longrightarrow \mathrm{Fe}(\mathrm{s}) ; E^{\circ}=-0... | 1 |
What are the two most powerful political parties in the United States? | 0 |
37. Line $L$ has intercepts $a$ and $b$ on the coordinate axes. When, the axes are rotated through a given angle, keeping the origin fixed, the same line $L$ has intercepts $p$ and $q$, then
(a) $a^{2}+b^{2}=p^{2}+q^{2}$
(b) $\frac{1}{a^{2}}+\frac{1}{b^{2}}=\frac{1}{p^{2}}+\frac{1}{q^{2}}$
(c) $a^{2}+p^{2}=b^{2}+q^{2... | 1 |
$\bullet 07$ A light detector (your eye) has an area of $2.00 \times 10^{-6} \mathrm{~m}^{2}$ and absorbs $80 \%$ of the incident light, which is at wavelength 500 nm . The detector faces an isotropic source, 3.00 m from the source. If the detector absorbs photons at the rate of exactly $4.000 \mathrm{~s}^{-1}$, at wha... | 1 |
Who can be credited with assisting the word humanism with finding a home in the English language? | 0 |
4. The statement that is not true about ozone is :
(a) in the stratosphere, CFCs release chlorine free radicals $(\mathrm{Cl})$ which reacts with $\mathrm{O}_{3}$ to give chlorine dioxide radicals.
(b) in the atmosphere, it is depleted by CFCs.
(c) in the stratosphere, it forms a protective shield against UV radia... | 1 |
Example 5.43 Find the number of seven letter words that can be formed by using the letters of the word SUC. CESS so that the two $C$ are together but no two $S$ are together. | 1 |
Is disqus comment in wordpress bad for SEO? | 0 |
35. The number of values of $x \in[0,2]$ at which $f(x)=\left|x-\frac{1}{2}\right|+$
Lo $|x-1|+\tan x$ is not differentiable at
a. 0
b. 1
c. 3
d. None of these :- | 1 |
Is it possible to detect if a computer connected to a network is hosting its own ad-hoc network? | 0 |
4. If all matter were made of electrically neutral particles such as neutrons, (a) there would be no force of friction (b) there would be no tension in the string (c) it would not be possible to sit on a chair (d) the earth could not move around the sun. | 1 |
2. The vertex of a parabola is the point $(a, b)$ and latus rectum is of length $l$. If the axis of the parabola is along the positive direction of $y$-axis, then its equation is
a. $(x+a)^{2}=\frac{l}{2}(2 y-2 b)$
b. $(x-a)^{2}=\frac{l}{2}(2 y-2 b)$
c. $(x+a)^{2}=\frac{l}{4}(2 y-2 b)$
d. $(x-a)^{2}=\frac{l}{8}(2 y-2 b... | 1 |
Song meaning: where does this Yes song derive its lyrics from? | 0 |
7. The correct order of hydration enthalpies of alkali metal ions is :
(a) $\mathrm{Li}^{+}>\mathrm{Na}^{+}>\mathrm{K}^{+}>\mathrm{Cs}^{+}>\mathrm{Rb}^{+}$
(b) $\mathrm{Na}^{+}>\mathrm{Li}^{+}>\mathrm{K}^{+}>\mathrm{Rb}^{+}>\mathrm{Cs}^{+}$
(c) $\mathrm{Na}^{+}>\mathrm{Li}^{+}>\mathrm{K}^{+}>\mathrm{Cs}^{+}>\mathrm{... | 1 |
53. The ratio of the number of atoms of two radioactive elements $A$ and $B$, in equilibrium with each other, is $3.1 \times 10^9: 1$. If $t_{\frac{1}{2}}$ of element $B$ is 6.45 yrs , calculate that of element $A$.
$$
\left(2 \times 10^{10}\right)
$$ | 1 |
(c) A hydrogen molecule rotating about its centre of mass. | 1 |
19. The pair of compounds which cannot exist together in solution is:
(a) $\mathrm{NaHCO}_{3}$ and $\mathrm{NaOH}$
(b) $\mathrm{Na}_{2} \mathrm{CO}_{3}$ and $\mathrm{NaHCO}_{3}$
(c) $\mathrm{Na}_{2} \mathrm{CO}_{3}$ and $\mathrm{NaOH}$
(d) $\mathrm{NaHCO}_{3}$ and $\mathrm{NaCl}$ | 1 |
How do you train German Shepherd/Pitbull mix? | 0 |
18. In the method using the transformers, assume that the ratio of the number of turns in the primary to that in the secondary in the step-up transformer is $1: 10$. If the power to the consumers has to be supplied at $200 \mathrm{~V}$, the ratio of the number of turns in the primary to that in the secondary in the ste... | 1 |
19. An ideal gas undergoes a quasi static, reversible process in which its molar heat capacity $C$ remains constant. If during this process the relation of pressure $p$ and volume $V$ is given by $p V^{n}=$ constant, then $n$ is given by (Here, $C_{p}$ and $C_{V}$ are molar specific heat at constant pressure and consta... | 1 |
12. A series $R-C$ circuit is connected to $\mathrm{AC}$ voltage source. Consider two cases; $(A)$ when $C$ is without a dielectric medium and $(B)$ when $C$ is filled with dielectric of constant 4 . The current $I_{R}$ through the resistor and voltage $V_{C}$ across the capacitor are compared in the two cases. Which o... | 1 |
12. The locus of point of intersection of perpendicular tangents is
a. $x^{2}+y^{2}=\frac{5}{4}$
b. $x^{2}+y^{2}=\frac{3}{2}$
c. $x^{2}+y^{2}=2$
d. none of these | 1 |
In what century did Darwin write? | 0 |
What is utility programs? | 0 |
What is Abyssinia? | 0 |
7. Two numbers are randomly selected and multiplied. Consider two events $E_{1}$ and $E_{2}$ defined as
$E_{1}$ : Their product is divisible by 5
$E_{2}:$ Unit's places in their product is 5
Which of the following statement is/are correct?
a. $E_{1}$ is twice as likely to occur as $E_{2}$
b. $E_{1}$ and $E_{2}$ are ... | 1 |
What is Richard Dawkins belief about the existence of God? | 0 |
How do I prepare for bank SO exam without coaching? | 0 |
What if God comes and gives you his powers what will you do? | 0 |
What hotel in Yercaud Hill-station would be safe for unmarried couples, without the harassment of police, hotel staff, and moral police? | 0 |
How much is the estimated cost of a tourist trip to USA in Indian rupees? | 0 |
1. In a gaseous reaction of the type, $a \mathrm{~A}+b \mathrm{~B} \rightarrow c \mathrm{C}+d \mathrm{D}$, which statement is wrong?
(a) $a$ litres of A combine with $b$ litres of B to give C and D
(b) $a$ moles of A combine with $b$ moles of B to give C and D
(c) $a \mathrm{~g}$ of A combine with $b \mathrm{~g}$ of B ... | 1 |
The word comic comes from the humorous pieces found where? | 0 |
Has political correctness in U.S. universities gone too far? | 0 |
What message did Voeller deliver using this myth? | 0 |
Where did John Hawkins and Francis Drake attack Spanish and Portuguese slave ships? | 0 |
(c) what are the roots of the $f(x)=10$ ? | 1 |
2. Show that $1!+2!+3!+\cdots+n!$ cannot be a perfect square for any $n \in N, n \geq 4$. | 1 |
People who study the anatomy of the central nervous system are known as what? | 0 |
Who did benevolent believers in humanism turn to for ideas about the philosophy? | 0 |
6. The co-ordinates $(2,3)$ and $(1,5)$ are the foci of an ellipse which passes through the origin, then the equation of
a. tangent at the origin is $(3 \sqrt{2}-5) x+(1-2 \sqrt{2}) y=0$
b. tangent at the origin is $(3 \sqrt{2}+5) x+(1+2 \sqrt{2} y)=0$
c. normal at the origin is $(3 \sqrt{2}+5) x-(2 \sqrt{2}+1) y=0$... | 1 |
13. If $\int \sin ^{-1} x \cos ^{-1} x d x=f^{-1}(x)$
$\left[A x-x f^{-1}(x)-2 \sqrt{1-x^{2}}\right]+2 x+C$, then
a. $f(x)=\sin x$
c. $A=\frac{\pi}{4}$
d. $A=\frac{\pi}{2}$
h. $f(x)=\cos x$ | 1 |
10.18 Two towers on top of two hills are 40 km apart. The line joining them passes 50 m above a hill halfway between the towers. What is the longest wavelength of radio waves, which can be sent between the towers without appreciable diffraction effects? | 1 |
What should I do to so that my five-year-old son can learn to control his anger? | 0 |
What is it like to take ketamine and opioids together? | 0 |
Example 1.24 Find the number of solutions of the equation $\sin x=x^{2}+x+1$. | 1 |
12. For the reaction $\mathrm{C}(\mathrm{s})+\mathrm{CO}_2(\mathrm{~g}) \rightleftharpoons 2 \mathrm{CO}(\mathrm{g})$, the partial pressures of $\mathrm{CO}_2$ and CO are respectively 4 and 8 atm . $K_p$ for the reaction is
(a) 16
(b) 2
(c) 0.5
(d) 4 | 1 |
10.24 Explain the significance of sodium, potassium, magnesium and calcium in biological fluids. | 1 |
9. The area (in sq units) of the quadrilateral formed by the tangents at the end points of the latusrectum to the ellipse $\frac{x^{2}}{9}+\frac{y^{2}}{5}=1$ is
(a) $\frac{27}{4}$
(b) 18
(c) $\frac{27}{2}$
(d) 27 | 1 |
What is the possibility of time travel becoming a reality? | 0 |
Why do people on Quora ask silly questions about the facts which can be googled very easily? | 0 |
10. When the temperature is increased, surface tension of water
(a) increases
(b) decreases
(c) remains constant
(d) shows irregular behaviour | 1 |
What two instruments were invented during The European Renaissance? | 0 |
18. The rays of different colours fail to converge at a point after going through a converging lens. This defect is called
(a) spherical aberration
(b) distortion
(c) coma
(d) chromatic aberratio | 1 |
What is the best street food in Delhi? | 0 |
Problem 10.4
Why does the solubility of alkaline earth metal hydroxides in water increase down the group? | 1 |
(ii) exactly one of them solves the problem. | 1 |
6. Which of the following compounds will show retention in configuration on nucleophic substitution by $\mathrm{OH}^{-}$ion ?
(a)
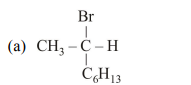
(b)
 elastically per second over an area $1 \mathrm{~m}^{2}$ with a speed $10^{4} \mathrm{~m} / \mathrm{s}$, the pressure exerted by the gas molecules will be of the order of
(a) $10^{4} \mathrm{~N} / \mathrm{m}^{2... | 1 |
(b) Find the time at which it reaches $x=0.25 \mathrm{~m}$. | 1 |
What are the Ctenophora and the Cnidaria animals sometimes called? | 0 |
What does Twitter Impression mean? "Your Tweets earned 3,247 impressions over the last week" . Is this good or bad? | 0 |
5. Are the following pairs of structures resonance contributors or different compounds?
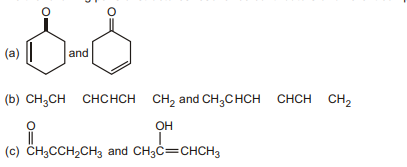
(d)
 in $0.01 \mathrm{~s}$ and (b) in $0.02 \mathrm{~s}$
Find the magnitudes of average values of $e$, $i$ and $\Delta q$ in both the cases. | 1 |
What author had a great impact in Rome? | 0 |
11.2 Identify allylic alcohols in the above examples. | 1 |
6.278. How much, in per cent, does the threshold energy of gamma quantum exceed the binding energy of a deuteron $\left(E_{b}=2.2 \mathrm{MeV}\right)$ in the reaction $\gamma+\mathrm{H}^{2} \rightarrow n+p$ ? | 1 |
Ex. 31. Lithium forms a b.c.c. lattice. If the lattice constant is $3.50 \times 10^{-10} \mathrm{~m}$ and the experimental density is $5.30 \times 10^{2} \mathrm{~kg} \mathrm{~m}^{-3}$, calculate the percentage occupancy of Li metal. $(\mathrm{Li}=7)$ | 1 |
2. A metal wire of length $L$, area of cross-section $A$ and Young's modulus $Y$ is stretched by a variable force $F$ such that $F$ is always slightly greater than the elastic forces of resistance in the wire. When the elongation of the wire is $l$
(a) the work done by $F$ is $\frac{Y A l^{2}}{2 L}$
(b) the work done... | 1 |
Can you "train" your brain? | 0 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.