Question stringlengths 2 3k | label int64 0 1 |
|---|---|
5. In the figure shown, $Z_{1}$ and $Z_{2}$ are two screens. Line $P O$ is the bisector line of $S_{1} S_{2}$ and $S_{3} S_{4}$. When $Z_{1}$ is removed, resultant intensity at $O$ due to slits $S_{1}$ and $S_{2}$ is $I$. Now, $Z_{1}$ is placed. For different values of $y$ given in Column I, match the resultant intensi... | 1 |
42. The coefficient of $x^{99}$ in the polynomial $(x-1)(x-2) \ldots(x-100)$ is....
$(1982,2 \mathrm{M})$ | 1 |
Why does “Cold War thinking” still not obsolete? | 0 |
3. A double star system consists of two stars A and B which have time periods $T_{A}$ and $T_{B}$. Radius $R_{A}$ and $R_{B}$ and mass $M_{A}$ and $M_{B}$. Choose the correct option.
(a) If $T_{A}>T_{B}$ then $R_{A}>R_{B}$ (b) If $T_{A}>T_{B}$ then $M_{A}>M_{B}$
(c) $\frac{T_{A}}{T_{B}}=\frac{R_{A}}{R_{B}}$
(d) $T_... | 1 |
What is the Sahara, and how do the average temperatures there compare to the ones in the Mojave Desert? | 0 |
What are some human primal instincts? | 0 |
What are the best books on Joseph Goebbels? | 0 |
Which of the following Configuration Management tool is easy to learn and implement? What are the advantages of choosing one over the other? Chef/Puppet | 0 |
What is everyone's favorite movie? | 0 |
When did the Suez Canal open? | 0 |
Is a second cold war with Russia a real possibility? | 0 |
In what ways is ego creative? | 0 |
1. One kilogram of water at $20^{\circ} \mathrm{C}$ is heated in an electric kettle whose heating element has a mean (temperature averaged) resistance of $20 \Omega$. The rms voltage in the mains is $200 \mathrm{~V}$. Ignoring heat loss from the kettle, time taken for water to evaporate fully, is close to
[Specific he... | 1 |
11. To position $\vec{r}$ of a particle moving in an $x y$ plane is given by $\vec{r}=\left(2.00 t^{3}-5.00 t\right) \hat{\mathrm{i}}+\left(6.00-7.00 t^{4}\right) \hat{\mathrm{j}}$, with $\vec{r}$ in meters and $t$ in seconds. In unit-vector notation, calculate (a) $\vec{r}$, (b) $\vec{v}$, and (c) $\vec{a}$ for $t=2.... | 1 |
What are the best horror movie scenes that shocked you? | 0 |
4. The strength of 11.2 volume solution of $\mathrm{H}_{2} \mathrm{O}_{2}$ is : [Given that molar mass of $\mathrm{H}=1 \mathrm{~g} \mathrm{~mol}^{-1}$ and $\left.\mathrm{O}=16 \mathrm{~g} \mathrm{~mol}^{-1}\right]$
(c) $\mathrm{SO}_{4}^{--}$ion
(a) $13.6 \%$
(b) $3.4 \%$
(c) $34 \%$
(d) $1.7 \%$ | 1 |
14. Which of the following pairs of molecules are NOT structural isomers?
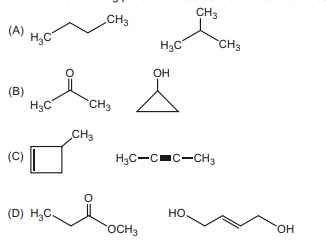 | 1 |
6.87. Find the possible values of energy of a particle of mass $m$ located in a spherically symmetrical potential well $U(r)=0$ for $r<r_{0}$ and $U(r)=\infty$ for $r=r_{0}$, in the case when the motion of the particle is described by a wave function $\psi(r)$ depending only on $r$. | 1 |
Why do more tourists go to India despite Turkey being more developed, more beautiful, & having the same culture? | 0 |
How do New York, Illinois and California maintain civil procedure laws? | 0 |
11. A small, conducting circular loop is placed inside a long solenoid carrying a current. The plane of the loop contains the axis of the solenoid. If the current in the solenoid is varied, the current induced in the loop is
(a) clockwise
(b) anticlockwise
(c) zero
(d) clockwise or anticlockwise depending on whether t... | 1 |
How affordable is the cost of living in Bangalore? | 0 |
16. A fixed mortar fires a bomb at an angle of $53^{\circ}$ above the horizontal with a muzzle velocity of $80 \mathrm{~ms}^{-1}$. A tank is advancing directly towards the mortar on level ground at a constant speed of $5 \mathrm{~m} / \mathrm{s}$. The initial separation (at the instant mortar is fired) between the mort... | 1 |
10. The arrangement of $\mathrm{Cl}^{-}$ions in CsCl structure is
(a) h.c.p.
(b) simple cubic
(c) f.c.c.
(d) b.c.c. | 1 |
Am I sociopath for not feeling empathy? | 0 |
How is the alcohol in beer/wine/others different from other types of alcohol? | 0 |
In the future: Can humans become immortals? | 0 |
17. The centre of circle inscribed in square formed by the lines $x^{2}-8 x+12=0$ and $y^{2}-14 y+45=0$, is
a. $(4,7)$
b. $(7,4)$
c. $(9,4)$
d. $(4,9)$
(IIT-JEE, 2003) | 1 |
b) 10 mL of $0.01 \mathrm{M} \mathrm{H}_{2} \mathrm{SO}_{4}+10 \mathrm{~mL}$ of $0.01 \mathrm{M} \mathrm{Ca}(\mathrm{OH})_{2}$ | 1 |
24. For which change $\Delta H \neq \Delta E$ :
(a) $\mathrm{H}_{2}(\mathrm{~g})+\mathrm{I}_{2}(\mathrm{~g}) \rightarrow 2 \mathrm{HI}(\mathrm{g})$
(b) $\mathrm{HCl}+\mathrm{NaOH} \rightarrow \mathrm{NaCl}$
(c) $\mathrm{C}(\mathrm{s})+\mathrm{O}_{2}(\mathrm{~g}) \rightarrow \mathrm{CO}_{2}(\mathrm{~g})$
$$
\text { ... | 1 |
What are some things that make Indians sad? | 0 |
7. An electric heater is used in a room of total wall area $137 \mathrm{~m}^{2}$ to maintain a temperature of $20^{\circ} \mathrm{C}$ inside it, when the outside temperature is $-10^{\circ} \mathrm{C}$. The walls have three different layers of materials. The innermost layer is of wood of thickness $2.5 \mathrm{~cm}$, t... | 1 |
What did social order mandate be strictly enforced? | 0 |
43. $3 \mathrm{~g}$ of a salt of molecular weight 30 is dissolved in $250 \mathrm{~g}$ of water. The molality of the solution is | 1 |
12. The solutions of equations $x^{2}+y^{2}-4 x-4 y=60$ and $x y+2 x+2 y=20$ satisfy the following equation(s).
(a) $x+y=10$
(b) $x+y=20$
(c) $x-y=10$
(d) $x+y=-10$ | 1 |
2. Consider the circle $x^{2}+y^{2}-10 x-6 y+30=0$. Let $O$ be the centre of the circle and tangent at $A(7,3)$ and $B(5,1)$ meet at $C$. Let $S=0$ represents family of circles passing through $A$ and $B$, then
a. area of quadrilateral $O A C B=4$
b. the radical axis for the family of circles $S=0$ is $x+y=10$
c. th... | 1 |
13. $\mathrm{KF}$ combines with $\mathrm{HF}$ to form $\mathrm{KHF}_{2}$. The compound contains the species.
(a) $\mathrm{K}^{+}, \mathrm{F}^{-}$and $\mathrm{H}^{+}$
(b) $\mathrm{K}^{+}, \mathrm{F}^{-}$and $\mathrm{HF}$
(c) $\mathrm{K}^{+}$and $\left[\mathrm{HF}_{2}\right]^{-}$
(d) $[\mathrm{KHF}]^{+}$and $\mathr... | 1 |
2. An ideal spring with spring constant $k$ is hung from the ceiling and a block of mass $M$ is attached to its lower end. The mass is released with the spring initially unstretched.
Then the maximum extension in the spring is $\quad
(a) $\frac{4 M g}{k}$
(b) $\frac{2 M g}{k}$
(c) $\frac{M g}{k}$
(d) $\frac{M g}{2 k}$ | 1 |
Example 5.65 A box contains 5 different red and 6 different white balls. In how many ways can 6 balls be selected so that there are at least two balls of each colour? | 1 |
5. The number of arrangements of the letters of the word BANANA in which the two N's do not appear adjacently, is
(2002, 1M)
(a) 40
(b) 60
(c) 80
(d) 100 | 1 |
What is the database used for Quora? | 0 |
What is the best way to cook a ham? | 0 |
5. Electric charges are distributed in a small volume. The flux of the electric field through a spherical surface of radius $10 \mathrm{~cm}$ surrounding the total charge is $25 \mathrm{Vm}$. The flux over a concentric sphere of radius $20 \mathrm{~cm}$ will be
(a) $25 \mathrm{~V} \mathrm{~m}$
(b) $50 \mathrm{~V} \math... | 1 |
How do you counter a C'Thun deck? | 0 |
Which belief is that God is the universe? | 0 |
19. If $A$ and $B$ are two independent events such that $P(A)>0$, and $P(B) \neq 1$, then $P(\bar{A} / \bar{B})$ is equal to
(a) $1-P(A / B)$
(b) $1-P(A / \bar{B})$
$(1982,2 \mathrm{M})$
(c) $\frac{1-P(A \cup B)}{P(B)}$
(d) $\frac{P(\bar{A})}{P(\bar{B})}$ | 1 |
109. If $\frac{\sin x}{\sin y}=\frac{1}{2}, \frac{\cos x}{\cos y}=\frac{3}{2}$ where $x, y \in\left(0, \frac{\pi}{2}\right)$, then the value of $\tan (x+y)$ is equal to
a. $\sqrt{13}$
h $\sqrt{14}$
c. $\sqrt{17}$
d. $\sqrt{15}$ | 1 |
What is helwett packaging? | 0 |
Why do I have no motivation to do anything? | 0 |
Identify electrophilic centre in the following: $\mathrm{CH}_3 \mathrm{CH}=\mathrm{O}, \mathrm{CH}_3 \mathrm{CN}, \mathrm{CH}_3 \mathrm{I}$. | 1 |
4. The point $P$ is at
(a) the centre of the rod
(b) the edge of the table supporting the end $B$
(c) the edge of the table supporting end $A$
(d) None of the above | 1 |
1. Find the sum of the eighth powers of all roots of the equation $x^{3}-x+1=0$
(a) 12
(b) 10
(c) 8
(d) None of these | 1 |
44. If $\int_{-\pi / 4}^{3 \pi / 4} \frac{e^{\pi / 4} d x}{\left(e^{x}+e^{\pi / 4}\right)(\sin x+\cos x)}=k \int_{-\pi / 2}^{\pi / 2} \sec x d x$, then the value of $k$ is
a. $\frac{1}{2}$
b. $\frac{1}{\sqrt{2}}$
c. $\frac{1}{2 \sqrt{2}}$
d. $-\frac{1}{\sqrt{2}}$ | 1 |
Why should I not invest in stocks? | 0 |
10. When a lens is inserted between an object and a screen which are a fixed distance apart the size of the image is either $6 \mathrm{~cm}$ or $\frac{2}{3} \mathrm{~cm}$. Find size of the object.
 If both Assertion and Reason are true and the Reas... | 1 |
Russia: How did Vladimir Putin become so popular in Russia? | 0 |
74. If $y=x \log \left(\frac{x}{a+b x}\right)$, then $x^{3} \frac{d^{2} y}{d x^{2}}$ equals to
a. $x \frac{d y}{d x}-y$
b. $\left(x \frac{d y}{d x}-y\right)^{2}$
c. $y \frac{d y}{d x}-x$
d. $\left(y \frac{d y}{d x}-x\right)^{2}$ | 1 |
Who were the 3 people who bought CBS Songs? | 0 |
What type of crisis resulted from the budget deficit and rising debt levels of Greece? | 0 |
10. The least value of $g(x)$ is
a. $-\frac{1}{4}$
b. -1
c. $-\frac{1}{3}$
d. $-\frac{1}{2}$ | 1 |
Why dogs bark more at nights? | 0 |
21. The total number of electrons that take part in forming the bond in $\mathrm{N}_{2}$ is
(a) 2
(b) 4
(c) 6
(d) 10 | 1 |
Why do I have different hair colors? | 0 |
11. If three distinct numbers are chosen randomly from the first 100 natural numbers, then the probability that all three of them are divisible by both 2 and 3 , is $(2004,1 \mathrm{M})$
(a) $\frac{4}{55}$
(b) $\frac{4}{35}$
(c) $\frac{4}{33}$
(d) $\frac{4}{1155}$ | 1 |
Are all conspiracy theories false? | 0 |
How to activate Commonwealth Bank debit card? | 0 |
What is the best framework to start build progressive web app? | 0 |
- Example 1.11 If the coordinates of two points $A$ and $B$ are $(3,4)$ and $(5,-2)$, respectively. Find the coordinates of any point $P$ if $P A=P B$ and area of $\triangle P A B=10$ sq. units. | 1 |
[SPOILER] How big of a shock was it that Darth Vader was Luke's father when it was first revealed? | 0 |
29. If metal ions of group III are precipitated by $\mathrm{NH}_{4} \mathrm{Cl}$ and $\mathrm{NH}_{4} \mathrm{OH}$ without prior oxidation by conc. $\mathrm{HNO}_{3} \ldots \ldots \ldots \ldots \ldots .$. is not completely precipitated. | 1 |
Why is Quora showing me the same questions again and again when I visit the website or app? | 0 |
What are some examples of "cultural perspective"? | 0 |
35. This question contains Statement-1 (Assertion) and Statement-2 (Reason) and has 4 choices (a), (b), (c) and (d) out of which ONLY ONE is correct
Statement-1 : Glucose gives a reddish-brown precipitate with Fehling's solution.
because
Statement-2 : Reaction of glucose with Fehling's solution give $\mathrm{CuO}$ a... | 1 |
13. Which one of the following alkenes when treated with $\mathrm{HCl}$ yields majorly an anti Markovnikov product?
[Main April 8, 2019 (II)]
(a) $\mathrm{CH}_{3} \mathrm{O}-\mathrm{CH}=\mathrm{CH}_{2}$
(b) $\mathrm{Cl}-\mathrm{CH}=\mathrm{CH}_{2}$
(c) $\mathrm{H}_{2} \mathrm{~N}-\mathrm{CH}=\mathrm{CH}_{2}$
(d) $... | 1 |
16. The number of nine-non-zero digits such that all the digits in the first four places are less than the digit in the middle and all the digits in the last four places are greater than that in the middle is
a. $2(4$ !)
b. $3(7!) / 2$
c. $2(7!)$
d. ${ }^{4} P_{+} \times{ }^{4} P_{4}$ | 1 |
10. The pair of the compounds in which both the metals are in the highest possible oxidation state is
(a) $\left[\mathrm{Fe}(\mathrm{CN})_6\right]^{3-},\left[\mathrm{Co}(\mathrm{CN})_6^{\mathrm{}}\right]^{3-}$
(b) $\mathrm{CrO}_2 \mathrm{Cl}_2, \mathrm{MnO}_4^{-}$
(c) $\mathrm{TiO}_3, \mathrm{MnO}_2$
(d) $\left[\... | 1 |
15. The compound which has one isopropyl group is :
(a) 2, 2, 3,3-tetramethylpentane
(b) 2, 2-dimethylpentane
(c) 2,2,3-trimethylpentane
(d) 2-methylpentane | 1 |
How are you doing today? | 0 |
How many articles does the Constitution have? | 0 |
What is it like to visit North Korea? | 0 |
2.13 The partial pressure of ethane over a solution containing $6.56 \times 10^{-3} \mathrm{~g}$ of ethane is 1 bar . If the solution contains $5.00 \times 10^{-2} \mathrm{~g}$ of ethane, then what shall be the partial pressure of the gas? | 1 |
2. A coil of resistance $100 \Omega$ is connected across a battery of emf $6 \cdot 0 \mathrm{~V}$. Assume that the heat developed in the coil is used to raise its temperature. If the heat capacity of the coil is $4.0 \mathrm{~J} \mathrm{~K}^{-1}$, how long will it take to raise the temperature of the coil by $15^{\circ... | 1 |
Where are more moist areas found in southern Europe? | 0 |
10. A Vernier callipers has 1 mm marks on the main scale. It's 20 equal divisions on the Vernier scale which match with 16 main scale divisions, For this Vernier callipers, the least count is
(a) 0.02 mm
(b) 0.05 mm
(c ) 0.1 mm
(d) 0.2 mm | 1 |
1. Light passes through a closed cylindrical tube containing a gas. If the gas is gradually pumped out, the speed of light inside the tube will
(a) increase
(b) decrease
(c) remain constant
(d) first increase and then decrease. | 1 |
What is the best publication for primary school? | 0 |
What needs to stay away from loopholes? | 0 |
- Example 5.4
If the base angles of a triangle are $22 \frac{1}{2}^{\circ}$ and $112 \frac{1}{2}^{\circ}$, then prove that the altitude of the triangle is equal to $\frac{1}{2}$ of its base. | 1 |
What are the pros and cons in the various sectors of the Indian economy by banning Indian Rupees of 500 and 1000 notes? What lead to this short notice by PM Narendra Modi? | 0 |
38. From the figure shown, establish a relation between $\mu_{1}, \mu_{2}$ and $\mu_{3}$
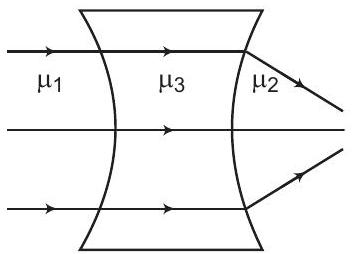
(a) $\mu_{1}<\mu_{2}<\mu_{3}$
(b) $\mu_{3}<\mu_{2} ; \mu_{3}=\mu_{1}$
(c) $\mu_{3}>\mu_{2} ; \mu_{3}... | 1 |
26. Which of the following colligative properties is associated with the concentration term 'molarity'?
(a) Lowering of vap. pressure
(b) Osmotic pressure
(c) Depression in f.p.
(d) Elevation in b.p. | 1 |
113. If $A, B, C$ are acute positive angles such that $A+B+C=\pi$ and $\cot A \cot B \cot C=k$, then
a. $K \leq \frac{1}{3 \sqrt{3}}$
h $K \geq \frac{1}{3 \sqrt{3}}$
c. $K<\frac{1}{9}$
d. $K>\frac{1}{3}$ | 1 |
17. The freezing point (in ${ }^{\circ} \mathrm{C}$ ) of a solution containing 0.1 gof $\mathrm{K}_{3}\left[\mathrm{Fe}(\mathrm{CN})_{6}\right]\left(\right.$ Mol. wt. 329) in $100 \mathrm{~g}$ of water $\left(K_{f}=1.86 \mathrm{Kkg} \mathrm{mol}^{-1}\right)$ is
(a) $-2.3 \times 10^{-2}$
(b) $-5.7 \times 10^{-2}$
(... | 1 |
In what country was the trading base for the Dutch? | 0 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.