Question stringlengths 2 3k | label int64 0 1 |
|---|---|
Are there other websites like Quora? | 0 |
32. The $(x, y)$ coordinates of the corners of a square plate are $(0,0),(L, 0),,(L, L)$ and $(0, L)$. The edges of the plate are clamped and transverse standing waves are set-up in it. If $u(x, y)$ denotes the displacement of the plate at the point $(x, y)$ at some instant of time, the possible expression (s) for $u$ ... | 1 |
Why do you think you're special? | 0 |
In what territory is New Delhi located? | 0 |
What are some of the best hangout places in Pune? | 0 |
61. At $518^{\circ} \mathrm{C}$ the rate of decomposition of a sample of gaseous acetaldehyde at a pressure of 363 torr, was 1.00 torr $\mathrm{s}^{-1}$ when $5 \%$ had reacted and 0.5 torr s $^{-1}$ when $33 \%$ had reacted. The order of the reaction is
(a) 0
(b) 2
(c) 3
(d) 1
[Hint: Rate $=k\left(\text { pressure of ... | 1 |
What kind of song is referred to as a bhajan? | 0 |
14. Let a circle be given by $2 x(x-a)+y(2 y-b)=0,(a \neq 0$, $b \neq 0$ ). Find the condition on $a$ and $b$ if two chords, each bisected by the $x$-axis, can be drawn to the circle from $\left(a, \frac{b}{2}\right)$.
(IIT-JEE, 1992) | 1 |
How has Kpop ruined your life? | 0 |
How does technology change culture? | 0 |
1. The mass of an electron is $9.11 \times 10^{-31} \mathrm{~kg}$, that of a proton is $1.67 \times 10^{-27} \mathrm{~kg}$. Find the ratio $F_{e} / F_{g}$ of the electric force and the gravitational force exerted by the proton on the electron. | 1 |
What are some diet regimes I can do to lose 29 pounds in 2 months? | 0 |
What should I do if I lost my money? | 0 |
What is the meaning of Hindi/Urdu word 'Swarg' and 'Jannat'? | 0 |
What characterized the Napoleonic Era? | 0 |
What did Bower's say about Greg's method? | 0 |
1. A solution of two components containing $n_{1}$ moles of the $1^{\text {st }}$ component and $n_{2}$ moles of the $2^{\text {nd }}$ component is prepared. $\mathrm{M}_{1}$ and $\mathrm{M}_{2}$ are the molecular weights of component 1 and 2 respectively. If $d$ is the density of the solution in $g \mathrm{~mL}^{-1}, ... | 1 |
71. A new carbon-carbon bond formation is possible in
(a) Cannizzaro reaction
(b) Friedel-Craft alkylation
(c) Clemmensen reduction
(d) Reimer-Tiemann reaction | 1 |
24. Global warming is due to increase of:
(a) methane and nitrous oxide in atmosphere
(b) methane and $\mathrm{CO}_{2}$ in atmosphere
(c) methane and $\mathrm{O}_{3}$ in atmosphere
(d) methane and $\mathrm{CO}$ in atmosphere | 1 |
4. Which of the following sequences regarding ease of abstraction of hydrogen atom is correct?
(A) $3^{\circ}>2^{\circ}>1^{\circ}$
(B) $3^{\circ}<2^{\circ}<1^{\circ}$
(C) $3^{\circ}<2^{\circ}>1^{\circ}$
(D) $3^{\circ}>2^{\circ}<1^{\circ}$ | 1 |
(b) behind the car? Speed of sound in air $=340 \mathrm{~m} / \mathrm{s}$. | 1 |
43. On hybridization of one $s$ and one $p$ orbitals we get :
(a) two mutually perpendicular orbitals
(b) two orbitals at $180^{\circ}$
(c) four orbitals directed tetrahedrally
(d) three orbitals in a plane | 1 |
51. If $\int_{1}^{2} e^{x^{2}} d x=a$, then $\int_{e}^{e^{4}} \sqrt{\ln x} d x$ is equal to
a. $2 e^{4}-2 e-a$
b. $2 e^{4}-e-a$
c. $2 e^{4}-e-2 a$
d. $e^{4}-e-a$ | 1 |
How do I increase organic search engine ranking? | 0 |
## Determing the Products of Oxidative Cleavage
What products would you would expect to obtain when the following compounds react with ozone and then with dimethylsulfide?
(a)
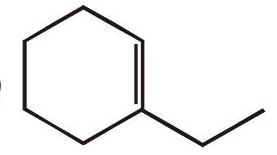
(b)
 $\mathrm{N}^{3-}<\mathrm{O}^{2-}<\mathrm{F}^{-}<\mathrm{Na}^{+}<\mathrm{Mg}^{2+}<\mathrm{Al}^{3+}$
(b) $\mathrm{Al}^{3+}<\mathrm{Na}^{+}<\mathrm{Mg}^{2+}<\math... | 1 |
5. System shown in figure is in equilibrium and at rest. The spring and string are massless, now the string is cut. The acceleration of mass $2 m$ and $m$ just after the string is cut will be
 \hat{\mathbf{i}}$, where $E$ is in $\mathrm{NC}^{-1}$ and $x$ is in metres. The values of constants are $A=20$ SI unit and $B=10$ SI unit. If the potential at $x=1$ is $V_{1}$ and that at $x=-5$ is $V_{2}$, then $V_{1}-V_{2}$ is
(a) $-48 \mathrm{~V}$
(b)... | 1 |
7. A 1-litre container contains 2 moles of $\mathrm{PCl}_5$ initially. If at equilibrium, $K_c$ is found to be 1 , the degree of dissociation of $\mathrm{PCl}_5$ is
(a) 1
(b) -1
(c) $\frac{1}{2}$
(d) 50 | 1 |
What are some of your New Year's resolutions for 2017? | 0 |
Do you care what others think of you? | 0 |
Example 2.14 Evaluate $\lim _{x \rightarrow 1} \frac{x^{2}+x \log _{e} x-\log _{e} x-1}{\left(x^{2}-1\right)}$. | 1 |
1. If the normal to the ellipse $3 x^{2}+4 y^{2}=12$ at a point $P$ on it is parallel to the line, $2 x+y=4$ and the tangent to the ellipse at $P$ passes through $Q(4,4)$ then $P Q$ is equal to
(a) $\frac{5 \sqrt{5}}{2}$
(b) $\frac{\sqrt{61}}{2}$
(c) $\frac{\sqrt{221}}{2}$
(d) $\frac{\sqrt{157}}{2}$ | 1 |
1. Define a positive integer $n$ to be squarish if either $n$ is itself a perfect square or the distance from $n$ to the nearest perfect square is a perfect square. For example, 2016 is squarish, because the nearest perfect square to 2016 is $45^{2}=2025$ and $2025-2016=9$ is a perfect square. (Of the positive integers... | 1 |
111. A car is parked among $N$ cars standing in a row, but not at either end. On his return, the owner finds that exactly ' $r$ ' of the $N$ places are still occupied. The probability that the places neighouring his car are empty is
a. $\frac{(r-1)!}{(N-1)!}$
b. $\frac{(r-1)!(N-r)!}{(N-1)!}$
c. $\frac{(N-r)(N-r-1)}{(N+... | 1 |
13. A capacitor of capacity $C$ is charged to a potential difference $V$ and another capacitor of capacity $2 C$ is charged to a potential difference $4 V$. The charging batteries are disconnected and the two capacitors are connected with reverse polarity (i.e. positive plate of first capacitor is connected to negative... | 1 |
How do you remove yourself from the Internet? | 0 |
What is a cyborg? | 0 |
How does a person find a technical co-founder if he/she is very interested in developing a site he/she is convinced will solve some problem? | 0 |
What was the Golden Age of Hollywood? | 0 |
6. An object of mass $m$ has a speed $v_{0}$ as it passes through the origin. It is subjected to a retarding force given by $F_{x}=-A x$. Here, $A$ is a positive constant. Find its $x$-coordinate when it stops. | 1 |
The company I work for is changing partners in the U.S. Even the name changing. Would it be considered as a new employer in my CV? | 0 |
60. IUPAC name of the given compound is :
$$
\left(\mathrm{CH}_{3}\right)_{2} \mathrm{C}=\mathrm{CHC}\left(\mathrm{C}_{2} \mathrm{H}_{5}\right)=\mathrm{CH}_{2}
$$
(A) 2-methyl-4-methylidenehex-2-ene
(B) 4-ethyl-2-methylpenta-1,3-diene
(C) 2-ethyl-4-methylpenta-1,3-diene
(D) 4-methyl-2-methylidenehex-2-ene | 1 |
19. 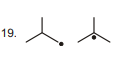 | 1 |
3. The excitation energy of a hydrogen like ion to its first excited state is $40.8 \mathrm{eV}$. The energy needed to remove the electron from the ion in the ground state is
(a) $54.4 \mathrm{eV}$
(b) $62.6 \mathrm{eV}$
(c) $72.6 \mathrm{eV}$
(d) $58.6 \mathrm{eV}$ | 1 |
What event led to African goods no longer being traded in Western Europe by the end of the 7th century? | 0 |
What is the best way to do intraday trading in the NSE/BSE equity? | 0 |
In which city is the Gandhi Smriti located? | 0 |
3. Assertion : If work done by conservative forces is positive, kinetic energy will increase.
Reason : Because potential energy will decrease. | 1 |
What type of architecture did Pugin consider truly Christian? | 0 |
What are the funniest jokes / stories you ever heard? | 0 |
13. The expression $\left(\tan ^{4} x+2 \tan ^{2} x+1\right) \cos ^{2} x$ when $x=\pi / 12$ can be equal to
a. $4(2-\sqrt{3})$
b $4(\sqrt{2}+1)$
c. $16 \cos ^{2} \pi / 12$
d $16 \sin ^{2} \pi / 12$ | 1 |
(xvii) Chloroethane to butane | 1 |
19. A capacitance of $2 \mu \mathbf{F}$ is required in an electrical circuit across a potential difference of $1 \mathrm{kV}$. A large number of $1 \mu \mathbf{F}$ capacitors are available which can withstand a potential difference of not more than $300 \mathrm{~V}$. The minimum number of capacitors required to achieve... | 1 |
Sample Problem 18.04 Heat to change temperature and state
(a) How much heat must be absorbed by ice of mass $m=$ 720 g at $-10^{\circ} \mathrm{C}$ to take it to the liquid state at $15^{\circ} \mathrm{C}$ ? | 1 |
What was the estimated net State Domestic Product of New Delhi for 2010? | 0 |
10. An aqueous solution of $6.3 \mathrm{~g}$ oxalic acid dihydrate is made up to 250 $\mathrm{mL}$. The volume of $0.1 \mathrm{~N} \mathrm{NaOH}$ required to completely neutralize 10 $\mathrm{mL}$ of this solution is
(a) $40 \mathrm{~mL}$
(b) $20 \mathrm{~mL}$
(c) $10 \mathrm{~mL}$
(d) $4 \mathrm{~mL}$ | 1 |
What is Bengel's rule? | 0 |
1. Normality of a solution is defined as
(a) number of eq./litre of solution
(b) number of eq./litre of solvent
(c) number of mole $/ \mathrm{kg}$ of solvent
(d) number of mole $/ \mathrm{kg}$ of solution | 1 |
80. Potassium manganate $\left(\mathrm{K}_{2} \mathrm{MnO}_{4}\right)$ is formed when
(a) chlorine is passed into aqueous $\mathrm{KMnO}_{4}$ solution
(b) manganese dioxide is fused with potassium hydroxide in air
(c) formaldehyde reacts with potassium permanganate in presence of a strong alkali
(d) potassium per... | 1 |
(iv) $\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{CH}_{2} \mathrm{OCH}_{3}$ | 1 |
10. Statement 1: $f(x)=|x| \sin x$ is non-differentiable at $x=0$. Statement 2: If $f(x)$ is not differentiable and $g(x)$ is differentiable at $x=a$, then $f(x) g(x)$ can still be differentiable at $x=a$. | 1 |
29. A uniform slender rod of mass $m$ and length $L$ is released from rest, with its lower end touching a frictionless horizontal floor. At the initial moment, the rod is inclined at an angle $\theta=30^{\circ}$ with the vertical. Then the value of normal reaction from the floor just after release will be
(a) $4 \mathr... | 1 |
Where were the Portuguese capacities centralized? | 0 |
4. Assertion : In circular motion work done by all the forces acting on the body is zero.
Reason : Centripetal force and velocity are mutually perpendicular. | 1 |
What are the pros and cons Ubuntu and Elementary OS? | 0 |
What dimensions are OK for an HTC One wallpaper that won't be cropped? | 0 |
7. In an $n-p-n$ transistor circuit, the collector current is $10 \mathrm{~mA}$. If 95 per cent of the electrons emitted reach the collector, which of the following statements are true?
(a) The emitter current will be $8 \mathrm{~mA}$.
(b) The emitter current will be $10.53 \mathrm{~mA}$.
(c) The base current will b... | 1 |
35. The length of the perpendicular drawn from $(1,2,3)$ to the line $\frac{x-6}{3}=\frac{y-7}{2}=\frac{z-7}{-2}$ is
a. 4
b. 5
c. 6
d. 7 | 1 |
Which two wrote Cultural studies [were] grounded in a pragmatic, liberal-pluralist tradition? | 0 |
Is Lionel Messi a good leader? | 0 |
3. Which one of the following equations represented parametrically equation to a parabolic curve?
a. $x=3 \cos t ; y=4 \sin t$
b. $x^{2}-2=2 \cos t ; y=4 \cos ^{2} \frac{t}{2}$
c. $\sqrt{x}=\tan t ; \sqrt{y}=\sec t$
d. $x=\sqrt{1-\sin t} ; y=\sin \frac{t}{2}+\cos \frac{t}{2}$ | 1 |
What do you think is different in your experience of life as a digital nomad compared to that of men? | 0 |
Along with Cooley, from whose work did Scheff derive social bond theory? | 0 |
17. Which species has the maximum number of lone pair of electrons on the central atom?
(a) $\left[\mathrm{ClO}_{3}\right]^{-}$
(b) $\mathrm{XeF}_{4}$
(c) $\mathrm{SF}_{4}$
(d) $\left[\mathrm{I}_{3}\right]^{-}$ | 1 |
11. Identify the incorrect statement regarding heavy water :
(a) It reacts with $\mathrm{SO}_{3}$ to form deuterated sulphuric acid $\left(\mathrm{D}_{2} \mathrm{SO}_{4}\right)$.
(b) It is used as a coolant in nuclear reactors
(c) It reacts with $\mathrm{CaC}_{2}$ to produce $\mathrm{C}_{2} \mathrm{D}_{2}$ and $\m... | 1 |
10. If for any reaction, the rate constant is equal to the rate of the reaction at all concentrations, the order is
(a) 0
(b) 2
(c) 1
(d) 3 | 1 |
What is 4 2/3-1 1/3÷2? | 0 |
6. A kettle with 2 litre water at $27^{\circ} \mathrm{C}$ is heated by operating coil heater of power $1 \mathrm{~kW}$. The heat is lost to the atmosphere at constant rate $160 \mathrm{~J} / \mathrm{s}$, when its lid is open. In how much time will water heated to $77^{\circ} \mathrm{C}$ with the lid open? (specific hea... | 1 |
Is it iOS is better than Android? | 0 |
19. In the electrolysis of $\mathrm{H}_{2} \mathrm{SO}_{4}, 9.72$ litres and 2.35 litres of $\mathrm{H}_{2}$ and $\mathrm{O}_{2}$ were liberated. Number of equivalent of persulphuric acid $\left(\mathrm{H}_{2} \mathrm{~S}_{2} \mathrm{O}_{8}\right)$ produced is
(a) $0 \cdot 448$
(b) 0.224
(c) 0.868
(d) 0... | 1 |
How would demonetizing 500 and 1000 rupee notes and introducing new 2000 rupee notes help curb black money and corruption? | 0 |
Is there any b.com 1st year student from IGNOU? | 0 |
4. $\frac{d y}{d x}=\frac{y}{2 y \ln y+y-x}$ | 1 |
52. The ionization energy of a hydrogen like Bohr atom is 4 rydberg. | 1 |
7. The temperature at which oxygen molecules have the same root mean square speed as helium atoms have at $300 \mathrm{~K}$ is: Atomic masses: $\mathrm{He}$ $=4 \mathrm{u}, \mathrm{O}=16 \mathrm{u})$
(a) $300 \mathrm{~K}$
(b) $600 \mathrm{~K}$
(c) $1200 \mathrm{~K}$
(d) $2400 \mathrm{~K}$ | 1 |
1 A uniform cylindrical rod of length $L$ and radius $r$, is made from a material whose Young's modulus of elasticity equals $Y$. When this rod is heated by temperature $T$ and simultaneously subjected to a net longitudinal compressional force $F$, its length remains unchanged. The coefficient of volume expansion of th... | 1 |
24. The total number of roots of the equation $f(x)=g(x)$ is / are
(a) $\infty$
(b) 1
(c) 2
(d) 0 | 1 |
30. Which one of the following will react fastest with $\mathrm{H}_{2}$ under catalytic hydrogenation condition
(a)
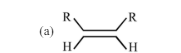
(b)
 length, mass and density
(b) length, time and velocity
(c) mass, time and velocity
(d) length, time and mass | 1 |
3. Number of hydrogen replaced by Deuterium in the given reaction are :
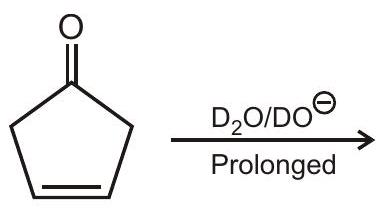
(A) 3
(B) 6
(C) 8
(D) 10 | 1 |
When was France occupied by Germany? | 0 |
3. Two impedances $Z_{1}$ and $Z_{2}$ when connected separately across a $230 \mathrm{~V}, 50 \mathrm{~Hz}$ supply consume $100 \mathrm{~W}$ and $60 \mathrm{~W}$ at power factor of 0.5 lagging and 0.6 leading respectively. If these impedances are now connected in series across the same supply, find
(a) total power abs... | 1 |
How many tourists did Greece welcome in 2009? | 0 |
14. The electron emitted in beta radiation originates from
(a) inner orbits of atom
(b) free electrons existing in nuclei
(c) decay of a neutron in a nucleus
(d) photon escaping from the nucleus | 1 |
2. Screw gauge is said to have a negative error
(a) when circular scale zero coincides with base line of main scale
(b) when circular scale zero is above the base line of main scale
(c) when circular scale zero is below the base line of main scale
(d) None of the above | 1 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.