Question stringlengths 2 3k | label int64 0 1 |
|---|---|
What do you think about ban on Rs. 500 and Rs. 1000 currency notes? | 0 |
What is Visual Studio Code? Why is it used? | 0 |
How can I stop being so insecure about myself? | 0 |
8. Which of the following is/are correct about the area bounded between the curves $y=\sqrt{4-x^{2}}$ and $y^{2}=3|x|$
(a) more than $2 \pi / 3$
(b) less than $\pi$
(c) more than $4 \pi / 3$
(d) less than $2 \pi / 3$ | 1 |
2. In the figure shown $P R Q$ is a curved vertical wall with rough inner surface with the floor is smooth. The radius of the curved wall is $r$ and its length is quarter of a circle. $A$ particle of mass $m$ is projected on the floor at $P$, grazing the rough wall as shown, with an initial velocity $v_{0}$. The veloci... | 1 |
8. A planet is moving round the sun in an elliptical orbit as shown. As the planet moves from $A$ to $B$
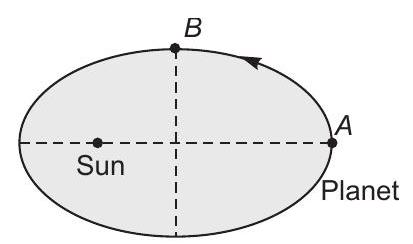
(a) its kinetic energy will decrease
(b) its potential energy will remain unchanged... | 1 |
Which are the best places for the holidays in India? | 0 |
26. If $\omega$ is an imaginary cube root of unity, then $\left(1+\omega-\omega^{2}\right)^{7}$ equals
a. $128 \omega$
b. $-128 \omega$
c. $128 \omega^{2}$
d. $-128 \omega^{2}$ | 1 |
Why would you get a nesting doll as a gift? | 0 |
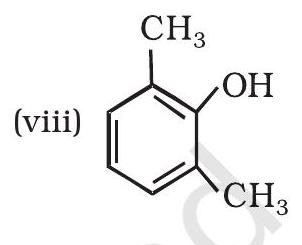 | 1 |
The constitution set boundaries for case law that originates from where? | 0 |
36. If the salts $\mathrm{M}_2 \mathrm{X}, \mathrm{QY}_2$ and $\mathrm{PZ}_3$ have the same solubilities $\left(<\frac{4}{27}\right)$, their $K_{\mathrm{sp}}$ values are related as
(a) $K_{\text {sp }}\left(\mathrm{M}_2 \mathrm{X}\right)=K_{\mathrm{sp}}\left(\mathrm{QY}_2\right)>K_{\mathrm{sp}}\left(\mathrm{PZ}_3\right... | 1 |
What are the worst reasons to pursue a computer science degree? | 0 |
2. The correct statement about probability density (except at infinite distance from nucleus) is :
(a) It can be zero for $1 s$ orbital
(b) It can be negative for $2 p$ orbital
(c) It can be zero for $3 p$ orbital
(d) It can never be zero for $2 s$ orbital | 1 |
57. A sample of gas has a molar volume of $10 \cdot 1 \mathrm{~L}$ at a pressure of 745 mmHg and a temperature of $-138^{\circ} \mathrm{C}$. Is the gas behaving ideally?
(No) | 1 |
Is your job allowed to tell you to come to work when you are sick? | 0 |
If prose uses sentences, what is the equivalent in poetry? | 0 |
How is the hostel 1B at Shiv Nadar University? | 0 |
What is generally the hottest month in Hyderabad? | 0 |
6. If $\omega(\neq 1)$ be a cube root of unity and $\left(1+\omega^{2}\right)^{n}=\left(1+\omega^{4}\right)^{n}$, then the least positive value of $n$ is
(2004, 1M)
(a) 2
(b) 3
(c) 5
(d) 6 | 1 |
18. For the compounds
$\mathrm{CH}_{3} \mathrm{Cl}, \mathrm{CH}_{3} \mathrm{Br}, \mathrm{CH}_{3} \mathrm{I}$ and $\mathrm{CH}_{3} \mathrm{~F}$,
the correct order of increasing $\mathrm{C}$-halogen bond length is:
(a) $\mathrm{CH}_{3} \mathrm{~F}<\mathrm{CH}_{3} \mathrm{Cl}<\mathrm{CH}_{3} \mathrm{Br}<\mathrm{CH}_{... | 1 |
Who finally was able to conquer the Sikh Empire? | 0 |
What are webinars best at? | 0 |
What's the gap between original Christian texts and subsequent editions of the same works? | 0 |
65 In Fig. 12-73, a uniform beam with a weight of 60 N and a length of 3.2 m is hinged at its lower end, and a horizontal force $\vec{F}$ of magnitude 50 N acts at its upper end. The beam is held vertical by a cable that makes angle $\theta=25^{\circ}$ with the ground and is attached to the beam at height $h=$ 2.0 m . ... | 1 |
3.2 Which important property did Mendeleev use to classify the elements in his periodic table and did he stick to that? | 1 |
What can I do if the person in the seat next to me on the plane is leaning into me and invading my space? | 0 |
I always get confused between a row and a column. What is any trick to remember the difference? | 0 |
After a history of raiding groups, what is not remarkable in the history of the subcontinent? | 0 |
10. Figure (12-Q1) represents two simple harmonic motions.
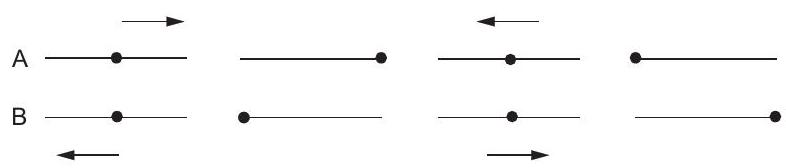
Figure 12-Q1 The parameter which has different values in the two
motions is
(a) amplitude (b) frequency
(c) phase (d) maximum ve... | 1 |
20. A plot of the number of neutrons $(\mathrm{N})$ against the number of protons (P) of stable nuclei exhibits upward deviation from linearity for atomic number, $Z>20$. For an unstable nucleus having N/P ratio less than 1 , the possible mode(s) of decay is (are)
(a) $\beta^{-}$-decay ( $\beta$ emission)
(b) orbit... | 1 |
4. An ideal monoatomic gas is carried around the cycle $A B C D A$ as shown in the figure. The efficiency of the gas cycle is
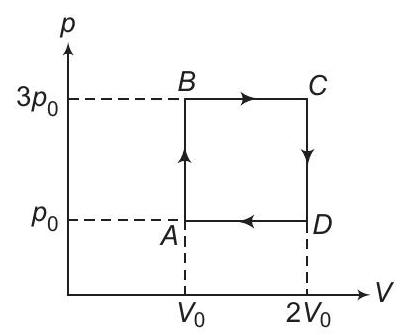
(a) $\frac{4}{21}$
(b) $\frac{2}{21}$
(c) $\frac{4}{31}$
(d) $\... | 1 |
44. Given the following $E^{0}$ values at $25^{\circ} \mathrm{C}$, calculate $K_{\text {sp }}$ for CdS .
$$
\begin{array}{ll}
\mathrm{Cd}^{2+}(\mathrm{aq})+2 \mathrm{e} \rightarrow \mathrm{Cd}(\mathrm{~s}) ; & E^{0}=-0.403 \mathrm{~V} \\
\mathrm{CdS}(\mathrm{~s})+2 \mathrm{e} \rightarrow \mathrm{Cd}(\mathrm{~s})+\math... | 1 |
86. If $\cos 2 B=\frac{\cos (A+C)}{\cos (A-C)}$, then $\tan A, \tan B, \tan C$ are in
a. A.P.
b GP.
c. H.P.
d none of these | 1 |
36. Water rises to a height of $10 \mathrm{~cm}$ in a certain capillary tube. An another identical tube when dipped in mercury the level of mercury is depressed by $3.42 \mathrm{~cm}$. Density of mercury is $13.6 \mathrm{~g} / \mathrm{cc}$. The angle of contact for water in contact with glass is $0^{\circ}$ and mercury... | 1 |
What is NOMA? | 0 |
73. $f(x)=\left\{\begin{array}{ll}x^{2}\left(\frac{e^{1 / x}-e^{-1 / x}}{e^{1 / x}+e^{-1 / x}}\right), & x \neq 0 \\ 0, & x=0\end{array}\right.$.Then
a. $f(x)$ is discontinuous at $x=0$
b. $f(x)$ is continuous but non-differentiable at $x=0$
c. $f(x)$ is differentiable at $x=0$
d $f^{\prime}(0)=2$ | 1 |
Which palace in Hyderabad did the work of Andrea Palladio influence? | 0 |
9. Statement 1: Let $A(\vec{a}), B(\vec{b})$ and $C(\vec{c})$ be threc points such that $\vec{a}=2 \hat{i}+\hat{k}, \vec{b}=3 \hat{i}-\hat{j}+3 \hat{k}$ and $\vec{c}=-\hat{i}+7 \hat{j}-5 \hat{k}$. Then $O A B C$ is a tetrahedron.
Statement 2: Let $A(\vec{a}), B(\vec{b})$ and $C(\vec{c})$ be three points such that vect... | 1 |
18. An ideal monoatomic gas undergoes a process in which its internal energy $U$ and density $\rho$ vary as $U \rho=$ constant. The ratio of change in internal energy and the work done by the gas is
(a) $\frac{3}{2}$
(b) $\frac{2}{3}$
(c) $\frac{1}{3}$
(d) $\frac{3}{5}$ | 1 |
7. A solution of $m$-chloroaniline, $m$-chlorophenol and $m$-chlorobenzoic acid in ethyl acetate was extracted initially with a saturated solution of $\mathrm{NaHCO}_{3}$ to give fraction $A$. The left over organic phase was extracted with dilute $\mathrm{NaOH}$ solution to give fraction $B$. The final organic layer wa... | 1 |
Once the slip laws are placed into the United States Statutes at Large, what are they called? | 0 |
In what city was a medical school located that was notably influenced by Islamic medicine? | 0 |
(ii) What are the atomic numbers of elements whose outermost electrons are represented by (a) $3 s^{1}$ (b) $2 p^{3}$ and (c) $3 p^{5}$ ? | 1 |
How does one find the volume of the solid region? | 0 |
How will replacing 1000 notes with 2000 notes going to stop corruption and black money? | 0 |
12. If $a, b, c$ are the sides of a triangle, then the minimum value of $\frac{a}{b+c-a}+\frac{b}{c+a-b}+\frac{c}{a+b-c}$ is equal to
a. 3
b. 6
c. 9
d. 12 | 1 |
What were the major effects of the cambodia earthquake, and how do these effects compare to the Cascadia earthquake in 1700? | 0 |
2. The Kjeldahl method of Nitrogen estimation fails for which of the following reaction products?
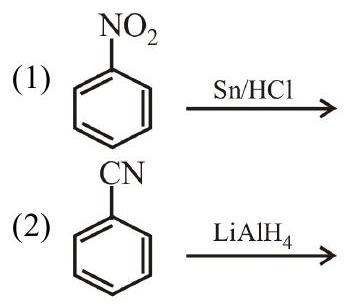
(3)
 a disc of radius $R$
(b) a ring of radius $R$
(c) a square lamina of side $2 R$
(d) four rods forming a square of side $2 R$ | 1 |
(i) $f: \mathbf{R} \rightarrow \mathbf{R}$ defined by $f(x)=3-4 x$ | 1 |
19. The mass and diameter of a planet are twice those of earth. What will be the period of oscillation of a pendulum on this planet. It is a second's pendulum on earth?
(a) $\sqrt{2} \mathrm{~s}$
(b) $2 \sqrt{2} \mathrm{~s}$
(c) $\frac{1}{\sqrt{2}} \mathrm{~s}$
(d) $\frac{1}{2 \sqrt{2}} \mathrm{~s}$ | 1 |
Why is my laptop slow? How can I fix it? | 0 |
12.30 Why is a solution of potassium hydroxide used to absorb carbon dioxide evolved during the estimation of carbon present in an organic compound? | 1 |
19. A vector is not changed if
(a) it is rotated through an arbitrary angle
(b) it is multiplied by an arbitrary scalar
(c) it is cross multiplied by a unit vector
(d) it is displaced parallel to itself | 1 |
Glial cells are also referred to as what? | 0 |
26. A constant voltage is applied to a series $R$ - $L$ circuit by closing the switch. The voltage across inductor $(L=2 \mathrm{H}$ ) is $20 \mathrm{~V}$ at $t=0$ and drops to $5 \mathrm{~V}$ at $20 \mathrm{~ms}$. The value of $R$ in $\Omega$ is
(a) $100 \ln 2 \Omega$
(b) $100(1-\ln 2) \Omega$
(c) $100 \ln 4 \Omega$
(... | 1 |
So what happens if you finish the Google foo.bar challenge? | 0 |
Why do Russians dislike Poles? | 0 |
10. Consider the charge configuration and a spherical Gaussian surface as shown in the figure. When calculating the flux of the electric field over the spherical surface, the electric field will be due to
 $3 \cdot 17$
(b) 0.317
(c) 317
(d) 31.7 | 1 |
Political corruption in legislature reduces what, overall? | 0 |
9. Solve $3^{\left(\log _{9} x\right)^{2}-\frac{9}{2} \log _{9} x+5}=3 \sqrt{3}$. | 1 |
22. Which of the following would you expect to be aromatic?

(A) I
(B) II
(C) III
(D) IV
(E) None of these | 1 |
What do Non-Americans think of Donald Trump? | 0 |
10. A circle of radius unity is centred at origin. Two particles start moving at the same time from the point $(1,0)$ and move around the circle in opposite direction. One of the particle moves counterclockwise with constant speed $v$ and the other moves clockwise with constant speed $3 v$. After leaving $(1,0)$, the t... | 1 |
11. Locus of the point $\sqrt{3 h}, \sqrt{3 k+2}$ if it lies on the line $x-y-1=0$ is a
a. straight line
b. circle
c. parabola
d. none of these | 1 |
2. $\mathrm{C}_{60}$, an allotrope of carbon cantains:
(a) 12 hexagons and 20 pentagons.
(b) 18 hexagons and 14 pentagons.
(c) 16 hexagons and 16 pentagons.
(d) 20 hexagons and 12 pentagons. | 1 |
How can a virus cause an infection? | 0 |
What happened to Mercedes-Benz's product strategy that resulted in their top models now costing over $200K? | 0 |
24. A thin lens, made of glass of refractive index $3 / 2$, produces a real and magnified image of an object in air. If the whole system, maintaining the same distance between the object and the lens, is immersed in water $(\mathrm{RI}=4 / 3)$, then the image formed will be
(a) real, magnified
(b) real, diminished
(c) ... | 1 |
42. Among the following, the intensive property is (properties are) Comprehension Passage Based Qquestions
(a) molar conductivity
(b) electromotive force
(c) resistance
(d) heat capacity | 1 |
What's the ACLU been up to? | 0 |
35. Acetic acid forms a dimer in the gas phase
The dimer is held together by two hydrogen bonds with a total strength of 66.5 kJ per mole of dimer. At $25^{\circ} \mathrm{C}$, the equilibrium constant for the dimerisation is $1.3 \times 10^{3}$ (pressure in atm). What is $\Delta S^{0}$ for the reaction? Assume that $... | 1 |
(i) 10 kg | 1 |
When did Larson finish his project? | 0 |
What percentage of Greece's economy is based on service? | 0 |
What two world championships were held in Hyderabad in 2009? | 0 |
12. Which of the following statements about anhydrous aluminium chloride is correct?
[1981 - 1 Mark]
(a) it exists as $\mathrm{AlCl}_{3}$ molecules
(b) it is not easily hydrolysed
(c) it sublimes at $100^{\circ} \mathrm{C}$ under vacuum
(d) it is a strong Lewis base | 1 |
How is your life after joining as an Income tax Inspector (be it excise, PO, examiner) through SSC CGL? | 0 |
Can you use an Apple Gift card on iTunes and the App Store? | 0 |
Self-Improvement: How can I motivate myself to work hard? | 0 |
(c) How much work must an external agent do to rotate this molecule by $180^{\circ}$ in this field, starting from its fully aligned position, for which $\theta=0$ ? | 1 |
What British company was heavily involved in the defeat of the Nawab's forces? | 0 |
28. A block $A$ of mass $45 \mathrm{~kg}$ is placed on another block $B$ of mass $123 \mathrm{~kg}$. Now block $B$ is displaced by external agent by $50 \mathrm{~cm}$ horizontally towards right. During the same time block $A$ just reaches to the left end of block $B$. Initial and final positions are shown in figures. T... | 1 |
39. If $\cos ^{-1} \sqrt{p}+\cos ^{-1} \sqrt{1-p}+\cos ^{-1} \sqrt{1-q}=\frac{3 \pi}{4}$, then the value of $q$ is
a. 1
b $\frac{1}{\sqrt{2}}$
c. $\frac{1}{3}$
d. $\frac{1}{2}$ | 1 |
Example 6.88 Find the values of $x$, for which $1 /(\sqrt{5+4 x})$ can be expanded as infinite series. | 1 |
3. At what separation should two equal charges, 1⋅0 C each,
be placed so that the force between them equals the
weight of a 50 kg person ? | 1 |
22. The addition of $\mathrm{HI}$ in the presence of peroxide catalyst does not follow anti-Markovnikov's rule because :
[Main Online April 9, 2013]
(a) $\mathrm{HI}$ is a strong reducing agent.
(b) H-I bond is too strong to be broken homolytically.
(c) I atom combines with $\mathrm{H}$ atom to give back $\mathrm{H... | 1 |
What was the origin of the second writing? | 0 |
11. Given
$$
\begin{aligned}
& \mathrm{E}_{\mathrm{Cl}_{2} / \mathrm{Cl}^{-}}^{\mathrm{o}}=1.36 \mathrm{~V}, \mathrm{E}_{\mathrm{Cr}^{3+} / \mathrm{Cr}}^{\mathrm{o}}=-0.74 \mathrm{~V}, \\
& \mathrm{E}_{\mathrm{Cr}_{2} / \mathrm{O}_{7}^{2-} / \mathrm{Cr}^{3+}}^{\mathrm{o}}=1.33 \mathrm{~V}, \mathrm{E}_{\mathrm{MnO}_{4}... | 1 |
How can you determine the chemical formula for C6H12? | 0 |
4. A point source emitting uniformly in all directions is placed above a table-top at a distance of $0.50 \mathrm{~m}$ from it. The luminous flux of the source is 1570 lumen. Find the illuminance at a small surface area of the table-top | 1 |
What's the best laptop I can buy for $700? | 0 |
For a addicted person its difficult to stop smoking? | 0 |
Can we use Jio 4G sim to 3G handsets? | 0 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.