Question stringlengths 2 3k | label int64 0 1 |
|---|---|
32. The reaction which proceeds in the forward direction is
(a) $\mathrm{Fe}_{2} \mathrm{O}_{3}+6 \mathrm{HCl} \rightarrow 2 \mathrm{FeCl}_{3}+3 \mathrm{H}_{2} \mathrm{O}$
(b) $\mathrm{NH}_{3}+\mathrm{H}_{2} \mathrm{O}+\mathrm{NaCl} \rightarrow \mathrm{NH}_{4} \mathrm{Cl}+\mathrm{NaOH}$
(c) $\mathrm{SnCl}_{4}+\mat... | 1 |
- Example 5.16 The sides of a triangle are $x^{2}+x+1,2 x+1$ and $x^{2}-1$, prove that the greatest angle is $120^{\circ}$. | 1 |
What is the best phone to buy below 15k? | 0 |
10. When a cube of wood floats in water, $60 \%$ of its volume is submerged. When the same cube floats in an unknown fluid $85 \%$ of its volume is submerged. Find the densities of wood and the unknown fluid. | 1 |
What is the name given to Japanese comics volumes? | 0 |
12.1 A geyser heats water flowing at the rate of 3.0 litres per minute from $27^{\circ} \mathrm{C}$ to $77^{\circ} \mathrm{C}$. If the geyser operates on a gas burner, what is the rate of consumption of the fuel if its heat of combustion is $4.0 \times 10^4 \mathrm{~J} / \mathrm{g}$ ? | 1 |
How would the relationship between Bruce Wayne and Rachel Dawes been had harvey died instead of Rachel? | 0 |
95 In Fig. 30-77, $R_1=8.0 \Omega, R_2=10 \Omega, L_1=0.30 \mathrm{H}, L_2=0.20 \mathrm{H}$, and the ideal battery has $\mathscr{C}=6.0 \mathrm{~V}$. (a) Just after switch S is closed, at what rate is the current in inductor 1 changing? (b) When the circuit is in the steady state, what is the current in inductor 1 ?
F... | 1 |
1. The speed of propagation of a wave in a medium is $300 \mathrm{~m} / \mathrm{s}$. The equation of motion of point at $x=0$ is given by $y=0.04 \sin 600 \pi t$ (metre). The displacement of a point $x=75 \mathrm{~cm}$ at $t=0.01 \mathrm{~s}$ is
(a) $0.02 \mathrm{~m}$
(b) $0.04 \mathrm{~m}$
(c) zero
(d) $0.028 \mat... | 1 |
23. A vector of magnitude $\sqrt{2}$ coplanar with the vectors $\vec{a}=\hat{i}+\hat{j}+2 \hat{k}$ and $\vec{b}=\hat{i}+2 \hat{j}+\hat{k}$, and perpendicular to the vector $\vec{c}=\hat{i}+\hat{j}+\hat{k}$, is
a. $-\hat{j}+\hat{k}$
b. $\hat{i}-\hat{k}$
c. $\hat{i}-\hat{j}$
d. $\hat{i}-\hat{j}$ | 1 |
Some bilaterians without a brain are what? | 0 |
24. At constant temperature, the equilibrium constant $\left(K_{p}\right)$ for the decomposition reaction $\mathrm{N}_{2} \mathrm{O}_{4} \rightleftharpoons 2 \mathrm{NO}_{2}$ is expressed by $K_{p}=$ $\left(4 x^{2} P\right) /\left(1-x^{2}\right)$, where $P=$ pressure, $x=$ extent of decomposition. Which one of the foll... | 1 |
25. Parabolas $y^{2}=4 a\left(x-c_{1}\right)$ and $x^{2}=4 a\left(y-c_{2}\right)$, where $c_{1}$ and $c_{2}$ are variable, are such that they touch each other. Locus of their point of contact is
a. $x y=2 a^{2}$
b. $x y=4 a^{2}$
c. $x y=a^{2}$
d. none of these | 1 |
How do you delete a question from Quora? | 0 |
7. Which of the following compounds does not dissolve in conc. $\mathrm{H}_{2} \mathrm{SO}_{4}$ even on warming?
[1983 - 1 Mark]
(a) ethylene
(b) benzene
(c) hexane
(d) aniline | 1 |
How much do Levis 501 shrink? | 0 |
47. Iron is rendered passive by treatment with concentrated
(a) $\mathrm{H}_{2} \mathrm{SO}_{4}$
(b) $\mathrm{H}_{3} \mathrm{PO}_{4}$
(c) $\mathrm{HCl}$
(d) $\mathrm{HNO}_{3}$ | 1 |
What results from possession by even benign spirits? | 0 |
How do I face ignorance by someone special? | 0 |
Which transportation vehicles were banned from entering New Delhi? | 0 |
What is the essence of the style of thinking embodied by people like Steven Levitt, Sam Harris and Daniel Kahneman? | 0 |
What is North Face's lifetime warranty policy? | 0 |
1. Consider a collision between an oxygen molecule and a hydrogen molecule in a mixture of oxygen and hydrogen kept at room temperature. Which of the following are possible?
(a) The kinetic energies of both the molecules increase.
(b) The kinetic energies of both the molecules decrease.
(c) kinetic energy of the oxyg... | 1 |
68. What is the IUPAC name of the following compound?
(A) 2-hydroxypropanoic acid
(B) 2-methyl-2-hydroxyethanoic acid
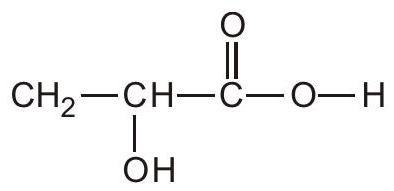
(C) Propanoic acid
(D) 2-carboxyethanol | 1 |
57. In which of the following reactions, an increase in volume of the container will favour the formation of products?
(a) $2 \mathrm{NO}_{2}(\mathrm{~g}) \rightleftharpoons 2 \mathrm{NO}(\mathrm{g})+\mathrm{O}_{2}(\mathrm{~g})$
(b) $4 \mathrm{NH}_{3}(\mathrm{~g})+5 \mathrm{O}_{2}(\mathrm{~g}) \rightleftharpoons 4 \mat... | 1 |
7. If $\ln (a+c), \ln (a-c)$, and $\ln (a-2 b+c)$ are in A.P., then
a. $a, b, c$ are in A.P.
b. $a^{2}, b^{2}, c^{2}$ are in A.P.
c. $a, b, c$ are in G.P.
d. $a, b, c$ are in H.P. | 1 |
4. The value of $\tan \alpha+\tan \beta+\tan \gamma+\tan \delta$ is
a. $1 / 3$
b. $8 / 3$.
c. $-8 / 3$
d. 0 | 1 |
2.12 Calculate the osmotic pressure in pascals exerted by a solution prepared by dissolving 1.0 g of polymer of molar mass 185,000 in 450 mL of water at $37^{\circ} \mathrm{C}$. | 1 |
What is a loam sand? | 0 |
7. A group 13 element ' $X$ ' reacts with chlorine gas to produce a compound $\mathrm{XCl}_{3} . \mathrm{XCl}_{3}$ is electron deficient and easily reacts with $\mathrm{NH}_{3}$ to form $\mathrm{Cl}_{3} \mathrm{X} \leftarrow \mathrm{NH}_{3}$ adduct, however, $\mathrm{XCl}_{3}$ does not dimerize. $\mathrm{X}$ is:
(a) $... | 1 |
40. The half-life period of a radioactive nuclide is 3 hours. In 9 hours its activity will be reduced by a factor of
(a) $1 / 9$
(b) $1 / 8$
(c) $1 / 27$
(d) $1 / 6$
[Hint: Cal. $N / N^{\circ}$ ] | 1 |
14.10 Enumerate the reactions of D-glucose which cannot be explained by its open chain structure. | 1 |
Storyboards are not considered what? | 0 |
What is the appeal of Nicki Minaj? | 0 |
30. Number of isomers of $\mathrm{C}_{5} \mathrm{H}_{10}$ are :
(A) 10
(B) 11
(C) 9
(D) 8 | 1 |
41. Three identical particles with velocities $v_{0} \hat{\mathbf{i}},-3 v_{0} \hat{\mathbf{j}}$ and $5 v_{0} \hat{\mathbf{k}}$ collide successively with each other in such a way that they form a single particle. The velocity vector of resultant particle is
(a) $\frac{v_{0}}{3}(\hat{\mathbf{i}}+\hat{\mathbf{j}}+\hat{\m... | 1 |
How can we take advantage and create real value out of data? | 0 |
Are there activities at NLS Bangalore for students to improve their public speaking skills? | 0 |
24. Starting with the same initial conditions, an ideal gas expands from volume $V_{1}$ to $V_{2}$ in three different ways, the work done by the gas is $W_{1}$ if the process is purely isothermal, $W_{2}$ if purely isobaric and $W_{3}$ if purely adiabatic, then
(a) $W_{2}>W_{1}>W_{3}$
(b) $W_{2}>W_{3}>W_{1}$
(c) $W_{... | 1 |
Ex. 44. Metallic mercury is obtained by roasting mercury (II) sulphide in a limited amount of air. Estimate the temperature range in which the standard reaction is product-favoured.
$$
\mathrm{HgS}(\mathrm{~s})+\mathrm{O}_{2}(\mathrm{~g}) \rightarrow \mathrm{Hg}(\mathrm{l})+\mathrm{SO}_{2}(\mathrm{~g})
$$
$\Delta H^{... | 1 |
Problem 7.7
The value of $K_{c}$ for the reaction
$2 \mathrm{~A} \rightleftharpoons \mathrm{B}+\mathrm{C}$ is $2 \times 10^{-3}$. At a given time, the composition of reaction mixture is $[\mathrm{A}]=[\mathrm{B}]=[\mathrm{C}]=3 \times 10^{-4} \mathrm{M}$. In which direction the reaction will proceed? | 1 |
42. For nitromethane molecule, write structure(s).
(i) showing significant resonance stabilisation.
(ii) indicating tautomerism.
 | 1 |
What is the meaning of this life? | 0 |
Is there an alternative supplements to viagra? | 0 |
Which martial art style is right for me? | 0 |
7. A person standing near the edge of the top of a building throws two balls $A$ and $B$. The ball $A$ is thrown vertically upward and $B$ is thrown vertically downward with the same speed. The ball $A$ hits the ground with a speed $v_{\mathrm{A}}$ and the ball $B$ hits the ground with a speed $v_{\mathrm{B}}$. We have... | 1 |
22. 0.1 mole of $\mathrm{CH}_{3} \mathrm{NH}_{2}\left(K_{b}=5 \times 10^{-4}\right)$ is mixed with 0.08 mole of $\mathrm{HCl}$ and diluted to one litre. What will be the $\mathrm{H}^{+}$concentration in the solution?
(a) $8 \times 10^{-2} \mathrm{M}$
(b) $8 \times 10^{-11} \mathrm{M}$
(c) $1.6 \times 10^{-11} \mathr... | 1 |
Eximple 1.21:: Prove that $\frac{2^{\log _{2} 1 / 4}-3^{\log _{27}\left(x^{2}+1\right)^{3}}-2 x}{7^{4 \log _{69} x}-x-1}>0 ; \forall x \in R$. | 1 |
What are some examples of organisms from the archaea kingdom? | 0 |
36. 4-Methylbenzenesulphonic acid reacts with sodium acetate to give
(a)
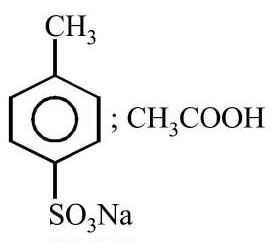
(b)
 bounded by the curves $y=|x|-1$ and $y=-|x|+1$ is
(a) 1
(b) 2
(c) $2 \sqrt{2}$
(d) 4 | 1 |
How is Ferrero Rocher made? | 0 |
What Rastrakuta king ruler for 64 years? | 0 |
5.102. A plane monochromatic light wave with intensity $I_{0}$ falls normally on the surfaces of the opaque screens shown in Fig. 5.20. Find the intensity of light $I$ at a point $P$
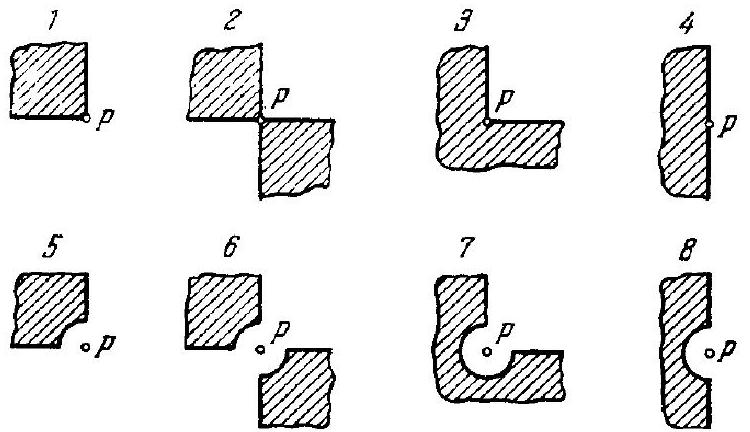
Fig. 5.... | 1 |
- Example 1.34 At what point the origin be shifted, if the coordinates of a point $(4,5)$ become $(-3,9)$ ? | 1 |
What are all the ways you can cook eggs? | 0 |
How do isotopes and atoms differ? | 0 |
68. Two vertices of a triangle are $(5,-1)$ and $(-2,3)$. If the orthocentre of the triangle is the origin, find the coordinates of the third vertex. | 1 |
What does "동" mean? Not the Google translate answer. What does it mean when someone puts it on their status? It’s Korean. Could it be related to love? | 0 |
How do I learn Kali Linux? | 0 |
Is the word "Quora" Spanish? | 0 |
Who can see my Facebook profile? | 0 |
What is copilotsearch.com? | 0 |
My friend said "Java is a girl's programming language and C++/C is for men" and I feel offended. How can I get over it? | 0 |
37. To what stable isotope would ${ }_{103}^{257} \mathrm{Lr}$ decay?
(a) ${ }_{82}^{208} \mathrm{~Pb}$
(b) ${ }_{83}^{209} \mathrm{Bi}$
(c) ${ }_{82}^{206} \mathrm{~Pb}$
(d) ${ }_{82}^{207} \mathrm{~Pb}$
[Hint: ${ }_{103}^{257} \mathrm{Lr}$ belongs to $(4 n+1)$ series] | 1 |
12. Which of the following salts is the most basic in aqueous solution?
(a) $\mathrm{Al}(\mathrm{CN})_{3}$
(b) $\mathrm{CH}_{3} \mathrm{COOK}$
(c) $\mathrm{FeCl}_{3}$
$$
\text { (d) } \mathrm{Pb}\left(\mathrm{CH}_{3} \mathrm{COO}\right)_{2}
$$ | 1 |
1. Consider the following table :
| Gas | $\mathrm{a} /\left(\mathrm{k} \mathrm{Pa} \mathrm{dm}^{6} \mathrm{~mol}^{-1}\right)$ | $\mathrm{b} /\left(\mathrm{dm}^{3} \mathrm{~mol}^{-1}\right)$ |
| ---: | :---: | :--- |
| A | 642.32 | 0.05196 |
| B | 155.21 | 0.04136 |
| C | 431.91 | 0.05196 |
| D | 155.21 | 0.4382 |
$\... | 1 |
What is the best way to tell myself no woman will love me because I don't look like a celebrity? | 0 |
Can I own a red panda? | 0 |
How many secondary hydrogens are present in the hydrocarbon below?
A.2
B.6
C.7
D.8
E.16 | 1 |
What issues to Savin address during this talk? | 0 |
How do bartenders actually become bartenders in Texas? | 0 |
What is an example of an extreme form of fear? | 0 |
Example 32.4 In interference, $\frac{I_{\max }}{I_{\min }}=\alpha$, find
$\left(a \frac{A_{\max }}{A_{\min }}\right.$
(b) $\frac{A_{1}}{A_{2}}$
(c) $\frac{I_{1}}{I_{2}}$ | 1 |
How do I get Reliance Jio Trial Sim in India before it's commercially launched? | 0 |
3. If $m$ is chosen in the quadratic equation $\left(m^{2}+1\right) x^{2}-3 x+\left(m^{2}+1\right)^{2}=0$ such that the sum of its roots is greatest, then the absolute difference of the cubes of its roots is
(a) $10 \sqrt{5}$
(c) $8 \sqrt{3}$
(b) $8 \sqrt{5}$
(d) $4 \sqrt{3}$
(2019 Main, 9 April II) | 1 |
1. Pick the correct options.
(a) All electrons have magnetic moment.
(b) All protons have magnetic moment.
(c) All nuclei have magnetic moment.
(d) All atoms have magnetic moment. | 1 |
Is it possible for international university students in Australia to get a job after graduation? Does it require permanent resident visa or something? | 0 |
In what year was Cluny Abbey founded? | 0 |
7. At $100^{\circ} \mathrm{C}$, copper $(\mathrm{Cu})$ has $\mathrm{FCC}$ unit cell structure with cell edge length of $x \AA$. What is the approximate density of $\mathrm{Cu}$ (in $\mathrm{g} \mathrm{cm}^{-3}$ ) at this temperature?
[Atomic Mass of $\mathrm{Cu}=63.55 \mathrm{u}$ ]
[Main Jan. 9, 2019 (II)]
(a) $\fra... | 1 |
What did Sagan argue was the only was to prove the existence of God? | 0 |
11. Equation $\frac{\pi^{e}}{x-e}+\frac{e^{\pi}}{x-\pi}+\frac{\pi^{\pi}+e^{e}}{x-\pi-e}=0$ has
(a) One real root in (e, $\pi$ ) and other in $(\pi-\mathrm{e}, \mathrm{e})$
(b) One real root in $(e, \pi)$ and other in $(\pi, \pi+e)$
(c) Two real roots in $(\pi-e, \pi+e)$
(d) Both the real roots are positive | 1 |
5. A particle is moving with a velocity $\mathbf{v}=k(y \hat{\mathbf{i}}+x \hat{\mathbf{j}})$, where $k$ is a constant.
The general equation for its path is
(a) $y=x^{2}+$ constant
(b) $y^{2}=x+$ constant
(c) $x y=$ constant
(d) $y^{2}=x^{2}+$ constant | 1 |
When did a victor emerge in World War I? | 0 |
What are your top 10 favorite reads? | 0 |
Do you seriously think time heals everything or we just learn to live with it? | 0 |
6. In a Wheatstone bridge (see figure), resistances $P$ and $Q$ are approximately equal. When $R=400 \Omega$, the bridge is balanced. On interchanging $P$ and $Q$, the value of $R$ for balance is $405 \Omega$. The value of $X$ is close to
(2019 Main, 11 Jan I)
 $\mathrm{Be}_{2}$
(b) $\mathrm{B}_{2}$
(c) $\mathrm{C}_{2}$
(d) $\mathrm{N}_{2}$ | 1 |
What is Forrest Gump based on? | 0 |
52. The thorium radioactive decay series produces one atom of ${ }^{208} \mathrm{~Pb}$ as the final disintegration product of an atom of ${ }^{232} \mathrm{Th} . t_{\frac{1}{2}}$ of ${ }^{232} \mathrm{Th}$ is $1.39 \times 10^{10}$ years. A certain rock is found to have a mass ratio of ${ }^{208} \mathrm{~Pb}$ and ${ }^... | 1 |
Does a person have to show their sexual oreintation in their personal acts? | 0 |
9. Water from a tap emerges vertically downwards with an initial speed of $1.0 \mathrm{~m} / \mathrm{s}$. The cross-sectional area of tap is $10^{-4} \mathrm{~m}^{2}$. Assume that the pressure is constant throughout the steam of water and that the flow is steady, the cross-sectional area of stream $0.15 \mathrm{~m}$ be... | 1 |
47. Three resistors having resistances of $1.60 \Omega, 2.40 \Omega$ and $4.80 \Omega$ are connected in parallel to a 28.0 V battery that has negligible internal resistance. Find
(a) the equivalent resistance of the combination.
(b) the current in each resistor.
(c) the total current through the battery.
(d) the vo... | 1 |
14. Two long parallel wires are at a distance $2 d$ apart. They carry steady equal currents flowing out of the plane of the paper as shown. The variation of the magnetic field $B$ along the line $X X^{\prime}$ is given by
 ${ }_{84}^{208} \mathrm{Po},{ }_{83}^{209} \mathrm{Bi}$
(b) ${ }_{19}^{39} \mathrm{~K},{ }_{19}^{40} \mathrm{~K}$
(c) ${ }_{31}^{71} \mathrm{Ga},{ }_{31}^{76} \mathr... | 1 |
16. The order of the oxidation state of the phosphorus atom in $\mathrm{H}_{3} \mathrm{PO}_{2}$, $\mathrm{H}_{3} \mathrm{PO}_{4}, \mathrm{H}_{3} \mathrm{PO}_{3}$ and $\mathrm{H}_{4} \mathrm{P}_{2} \mathrm{O}_{6}$ is
(a) $\mathrm{H}_{3} \mathrm{PO}_{3}>\mathrm{H}_{3} \mathrm{PO}_{2}>\mathrm{H}_{3} \mathrm{PO}_{4}>\math... | 1 |
What's your review of the newly released movie Baar Baar Dekho(2016)? | 0 |
How do I delete an answer for a question that I asked on Quora? | 0 |
26. The gravitational field at $B$ is
(a) $G M$
(b) $\frac{21 G M}{5}$
(c) $\frac{31}{16} G M$
(d) $\frac{31}{8} G M$ | 1 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.