Question stringlengths 2 3k | label int64 0 1 |
|---|---|
4. If the equation has four real roots, then
a. $|a| \geq 2 \sqrt{2}$
b $|a|<2 \sqrt{2}$
c. $a \geq-2 \sqrt{2}$
d none of these | 1 |
How do I overcome my extreme anxiety? | 0 |
4. For any odd integer $n \geq 1, n^{3}-(n-1)^{3}+\cdots+(-1)^{n-1} 1^{3}$ $=$ $\qquad$ . | 1 |
4. The number of diagonal matrix $A$ of order $n$ for which $A^{3}=A$ is
a. 1
b. 0
c. $2^{n}$
d. $3^{\prime \prime}$ | 1 |
3.163. The gap between the plates of a parallel-plate capacitor is filled up with an inhomogeneous poorly conducting medium whose conductivity varies linearly in the direction perpendicular to the plates from $\sigma_{1}=1.0 \mathrm{pS} / \mathrm{m}$ to $\sigma_{2}=2.0 \mathrm{pS} / \mathrm{m}$. Each plate has an area ... | 1 |
Where can we get a free guide on MMO games? | 0 |
7. Solve $\left|\frac{x^2}{x-1}\right| \leq 1$ using the graphical method. | 1 |
27. Assertion : Ozone is destroyed by CFCs in the upper stratosphere.
Reason : Ozone holes increase the amount of UV radiation reaching the earth.
(a) Assertion and reason are incorrect.
(b) Assertion and reason are both correct, and the reason is the correct explanation for the assertion.
(c) Assertion and reason... | 1 |
In what year did the occurrence of using different color schemes for the parties stop? | 0 |
3. The value of ${ }^{n} C_{0} \cdot{ }^{2 n} C_{r}-{ }^{n} C_{1} \cdot{ }^{2 n-2} C_{r}+{ }^{n} C_{2} \cdot{ }^{2 n-4} C_{r}+\ldots+(-1)^{n}{ }^{n} C_{n}{ }^{2 n-2 n} C_{n}$ is
(a) $2^{2 \mathrm{n}-\mathrm{r}+1 \mathrm{n}} C_{\mathrm{r}-\mathrm{n}}$, if $r>n$
(b) $2^{2 \mathrm{n}-\mathrm{r} \mathrm{n}} C_{\mathrm{r}-\... | 1 |
What is the Average salary for a Civil engineer in Australia.? | 0 |
51. Which of the following choice (s) is (are) correct for a gas?
(a) $p_{\text {ideal }}>p_{\text {real }}$
(b) $V_{\text {ideal }}>V_{\text {real }}$
(c) $p_{\text {ideal }}<p_{\text {real }}$
(d) $V_{\text {ideal }}<V_{\text {real }}$ | 1 |
Why is uranium the element used to create nuclear energy? | 0 |
14. The number of $\mathrm{P}-\mathrm{O}$ bonds in $\mathrm{P}_{4} \mathrm{O}_{6}$ is:
(a) 9
(b) 6
(c) 12
(d) 18 | 1 |
13. Two identical circular loops of metal wire are lying on a table without touching each other. Loop $A$ carries a current which increases with time. In response, the loop $B$
(a) remains stationary
(b) is attracted by the loop $A$
(c) is repelled by the loop $A$
(d) rotates about its $\mathrm{CM}$, with $\mathr... | 1 |
How is a court hierarchy established? | 0 |
2. If $\omega(\neq 1)$ is a cube root of unity, then value of the determinant
$$
\left|\begin{array}{ccc}
1 & 1+i+\omega^{2} & \omega^{2} \\
1-i & -1 & \omega^{2}-1 \\
-i & -i+\omega-1 & -1
\end{array}\right| \text { is }
$$
a. 0
b. 1
c. $i$
d. $\omega$
(IIT-JEE, 1995) | 1 |
(b) When concentrated sulphuric acid is added to an inorganic mixture containing chloride, we get colourless pungent smelling gas HCl , but if the mixture contains bromide then we get red vapour of bromine. Why ? | 1 |
What is it that you are doing makes you different from others? | 0 |
What is the opportunity cost of studying in a university? | 0 |
37. The electron in a hydrogen atom makes a transition $n_{1} \rightarrow n_{2}$, where $n_{1}$ and $n_{2}$ are the principal quantum numbers of two states. Assume the Bohr's model to be valid. The time period of the electron in the initial state is eight times that in the final state. The possible values of $n_{1}$ an... | 1 |
9. The decreasing order of basicity of the following amines is:
(A)
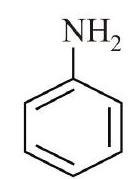
(B)
 What happens if the performer hangs stationary on the rope? (b) At what magnitude of acceleration does the performer just avoid breaking the rope? | 1 |
2. What is the IUPAC name for the organic compound with the condensed formula of $\mathrm{HCOCHBrCOCH}_{3}$ ?
(A) 3-bromo-4-formylpropan-2-one
(B) 4-formyl-3-bromopropan-2-one
(C) 2-bromo-3-oxobutanal
(D) 3-oxo-2-bromobutanal | 1 |
Example 22.10 Three rods made of the same material and having the same cross-section have been joined as shown in the figure. Each rod is of the same length. The left and right ends are kept at $0^{\circ} \mathrm{C}$ and $90^{\circ} \mathrm{C}$, respectively. The temperature of junction of the three rods will be
(JEE ... | 1 |
3. A positive point charge is released from rest at a distance $r_{0}$ from a positive line charge with uniform density. The speed $(v)$ of the point charge, as a function of instantaneous distance $r$ from line charge, is proportional to
 $\mathrm{HCl}$
(b) $\mathrm{HOCl}$
(c) $\mathrm{HBr}$
(d) $\mathrm{HI}$ | 1 |
Which one is your favourite song? | 0 |
Could you fire a bullet into orbit? | 0 |
If the Indian government has decided to demonetise 500 and 1000 rupee notes, why are they bringing back new 500 and 2000 Rs notes? | 0 |
1. The sum $\sum_{k=1}^{20} k \frac{1}{2^{k}}$ is equal to
(2019 Main, 8 April II)
(a) $2-\frac{11}{2^{19}}$
(b) $1-\frac{11}{2^{20}}$
(c) $2-\frac{3}{2^{17}}$
(d) $2-\frac{21}{2^{20}}$ | 1 |
How do sponges typically feed? | 0 |
13. Calculate the per cent concentration of a 9.28 N NaOH solution of density $1.31 \mathrm{~g} / \mathrm{mL}$.
(28.3\%) | 1 |
4. Find $x y z$ if $\quad \log _{2} x+\log _{4} y+\log _{4} z=2$
$$
\log _{9} x+\log _{3} y+\log _{9} z=2
$$
And $\quad \quad \quad \quad \quad \quad\log _{16} x+\log _{16} y+\log _{4} z=2$
(a) 24
(b) 32
(c) 12
(d) None of these | 1 |
How did the theory of evolution impact and/or change taxonomy? What did it help us learn? | 0 |
37. 3 g of activated charcoal was added to 50 mL of acetic acid solution $(0.06 \mathrm{~N})$ in a flask. After an hour it was filtered and the strength of the filtrate was found to be 0.042 N . The amount of acetic acid adsorbed (per gram of charcoal) is
(a) 18 mg
(b) 36 mg
(c) 42 mg
(d) 54 mg
[Hint: m.e. of $\mathrm... | 1 |
16. For a dilute solution containing $2.5 \mathrm{~g}$ of a non-volatile non-electrolyte solute in $100 \mathrm{~g}$ of water, the elevation in boiling point at $1 \mathrm{~atm}$ pressure is $2^{\circ} \mathrm{C}$. Assuming concentration of solute is much lower than the concentration of solvent, the vapour pressure ( $... | 1 |
16. Statement 1: $\int_{0}^{x}|\sin t| d t$, for $x \in[0,2 \pi]$ is a nondifferentiable function.
Statement 2: $|\sin t|$ is non-differentiable at $x=\pi$. | 1 |
(v) $\mathrm{CH}_{3} \mathrm{C}\left(p-\mathrm{ClC}_{6} \mathrm{H}_{4}\right)_{2} \mathrm{CH}(\mathrm{Br}) \mathrm{CH}_{3}$ | 1 |
5. The area bounded by the curve $y=x|x|, x$-axis and the ordinates $x=-1$ and $x=1$ is given by
(A) 0
(B) $\frac{1}{3}$
(C) $\frac{2}{3}$
(D) $\frac{4}{3}$
[Hint : $y=x^{2}$ if $x>0$ and $y=-x^{2}$ if $x<0$ ]. | 1 |
What is the benefit of exercising on a trampoline as an adult? | 0 |
Problem 2.11 Calculate the energy associated with the first orbit of $\mathrm{He}^{+}$. What is the radius of this orbit? | 1 |
What has been increasingly abandoned in the past two decades? | 0 |
2. In a room containing air, heat can go from one place to another
(a) by conduction only
(b) by convection only
(c) by radiation only
(d) by all the three modes. | 1 |
What does GRECO stand for? | 0 |
4. Naturally occurring boron consists of two isotopes whose atomic weights are 10.01 and 11.01 . The atomic weight of natural boron is 10.81 . Calculate the percentage of each isotope in natural boron.
(20\%; 80\%) | 1 |
49. 0.9546 g of a Rochelle salt, $\mathrm{NaKC}_{4} \mathrm{H}_{4} \mathrm{O}_{6} \cdot 4 \mathrm{H}_{2} \mathrm{O}$, on ignition, gave $\mathrm{NaKCO}_{3}$, which was treated with 41.72 mL of $0.1307 \mathrm{~N} \mathrm{H}_{2} \mathrm{SO}_{4}$. The unreacted $\mathrm{H}_{2} \mathrm{SO}_{4}$ was then neutralised by 1.9... | 1 |
17. Statement 1: The equations of the straight lines joining origin to the points of intersection of $x^{2}+y^{2}-4 x-2 y=4$ and $x^{2}+y^{2}-2 x-4 y-4=0$ is $x-y=0$.
Statement 2: $y+x=0$ is common chord of $x^{2}+y^{2}-4 x$ $-2 y=4$ and $x^{2}+y^{2}-2 x-4 y-4=0$. | 1 |
Why did pritz klein come up with the KSOG? | 0 |
If I want to start writing, where do I begin? How can I learn to structure my ideas and words? | 0 |
What is the name of one of the largest gardens in new Delhi? | 0 |
33. The distance between the line $\vec{r}=2 \hat{i}-2 \hat{j}+3 \hat{k}+\lambda(\hat{i}-\hat{j}+4 \hat{k})$ and the plane $\vec{r} \cdot(\hat{i}+5 \hat{j}+\hat{k})=5$ is
a. $\frac{10}{3 \sqrt{3}}$
b. $\frac{10}{9}$
c. $\frac{10}{3}$
d. $\frac{3}{10}$ | 1 |
65. Match each set of hybrid orbitals from LIST-I with complex(es) given in LIST-II
[Adv. 2018]
## List-I List-
II
(A) $d s p^{2}$
(B) $s p^{3} \quad$ (q) $\quad\left[\mathrm{Ti}\left(\mathrm{H}_{2} \mathrm{O}\right)_{3} \mathrm{Cl}_{3}\right]$
(C) $s p^{3} d^{2}$ (r) $\left[\mathrm{Cr}\left(\mathrm{NH}_{3}\right)_{... | 1 |
The minute hand of a clock overtakes the hour hand at intervals of 65 minutes of correct time. How much does the clock gain or lose in a day? | 0 |
What did these three categories correspond with? | 0 |
What countries have big public sectors but low corruption? | 0 |
The National Museum began in 1947 with a display of what type of art? | 0 |
My college open WiFi only connects to laptops, after connected it asks for a particular student ID and password provided by college, how to bypass it? | 0 |
What is control memory? | 0 |
What is `pressure drop? | 0 |
1. How many distinct terminal alkynes exist with a molecular formula of $\mathrm{C}_{5} \mathrm{H}_{8}$ ?
(A) 1
(B) 2
(C) 3
(D) 4
(E) 5 | 1 |
How did Zeus rule his family and what kind of relationship did he have with Ares, his son? | 0 |
1. If $f(x)=\sqrt{\frac{x-\sin x}{x+\cos ^{2} x}}$, then $\lim _{x \rightarrow \infty} f(x)$ is
$$
\begin{array}{ll}
\text { a. } 0 & \text { b. } \infty \\
\text { c. } 1 & \text { d. None of these }
\end{array}
$$ | 1 |
9. If $(x-2)^{6}+(x-4)^{6}=64$, then equation has
(a) real and irrational roots
(b) real and rational roots
(c) two real and two irrational roots
(d) None of the above | 1 |
What does an occupational therapy assistant do? | 0 |
86. If $f: R^{+} \rightarrow R, f(x)+3 x f\left(\frac{1}{x}\right)=2(x+1)$, then $f(99)$ is equal to
a. 40
b. 30
c. 50
d. 60 | 1 |
1. The rate constant $(k)$ of a reaction is measured at different temperatures (T), and the data are plotted in the given figure. The activation energy of the reaction in $\mathrm{kJ} \mathrm{mol}^{-1}$ is : ( $R$ is gas constant)
(a) $2 / R$
(b) $1 / R$
(c) $R$
(d) $2 R$ | 1 |
18. If $f:(0, \infty) \rightarrow R$ be given by
$$
f(x)=\int_{1 / x}^{x} e^{-t+\frac{1}{t}} \frac{d t}{t}
$$
Then,
(a) $f(x)$ is monotonically increasing on $[1, \infty)$
(b) $f(x)$ is monotonically decreasing on $[0,1)$
(c) $f(x)+f \frac{1}{x}=0, \forall x \in(0, \infty)$
(d) $f\left(2^{x}\right)$ is an odd f... | 1 |
How can I control on my anger? | 0 |
9. A copper ball of mass $100 \mathrm{~g}$ is at a temperature $T$. It is dropped in a copper calorimeter of mass $100 \mathrm{~g}$, filled with $170 \mathrm{~g}$ of water at room temperature. Subsequently, the temperature of the system is found to be $75^{\circ} \mathrm{C}$. T is (Given, room temperature $=30^{\circ} ... | 1 |
19. The bond between carbon atom (1) and carbon atom (2) in compound $\mathrm{N} \equiv \underset{1}{\mathrm{C}}-\underset{2}{\mathrm{CH}}=\mathrm{CH}_{2}$ involves the hybrids as
(a) $s p^{2}$ and $s p^{2}$
(b) $s p^{3}$ and $s p$
(c) $s p$ and $s p^{2}$
(d) $s p$ and $s p$ | 1 |
Ex. 19. 100 g of a sample of HCl solution of relative density 1.17 contains 33.4 g of HCl . What volume of this HCl solution will be required to neutralise exactly 5 litres of $\frac{\mathrm{N}}{10} \mathrm{NaOH}$ solution? | 1 |
Which is the best format to download and play from 1080p and 720p, Blu-ray, BDrip, or HDrip? | 0 |
7.21 Equilibrium constant, $K_{c}$ for the reaction
$$
\mathrm{N}_{2}(\mathrm{~g})+3 \mathrm{H}_{2}(\mathrm{~g}) \rightleftharpoons 2 \mathrm{NH}_{3}(\mathrm{~g}) \text { at } 500 \mathrm{~K} \text { is } 0.061
$$
At a particular time, the analysis shows that composition of the reaction mixture is $3.0 \mathrm{~mol} ... | 1 |
6. Which one of the following bases is not present in DNA?
(a) Quinoline
(b) Adenine
(c) Cytosine
(d) Thymine | 1 |
Who undermined The League Of Nations? | 0 |
1. A satellite orbiting close to the surface of earth does not fall down because the gravitational pull of earth
(a) is balanced by the gravitational pull of moon
(b) is balanced by the gravitational pull of sun
(c) provides the necessary acceleration for its motion along the circular path
(d) makes it weightless | 1 |
Ex. 15. The standard heats of formation at 298 K for $\mathrm{CCl}_{4}(\mathrm{~g}), \mathrm{H}_{2} \mathrm{O}(\mathrm{g}), \mathrm{CO}_{2}(\mathrm{~g})$ and $\mathrm{HCl}(\mathrm{g})$ are $-25 \cdot 5,-57 \cdot 8,-94 \cdot 1$ and $-22 \cdot 1 \mathrm{kcal}$ per mole respectively. Calculate $\Delta H^{0}$ at 298 K for ... | 1 |
When did the clerics and tradesmen start spreading architectural information? | 0 |
What is the best way to learn AngularJS? | 0 |
Who is better, Trump or Clinton? | 0 |
Where can I get very reasonable for any promotional lanyards in Melbourne? | 0 |
Why must there be compititors? | 0 |
What happens when you lack a chromosome? | 0 |
Why does Gmail ask for my phone number whenever I try to create a new account? | 0 |
Example 11 Consider two points P and Q with position vectors $\overrightarrow{\mathrm{OP}}=3 \vec{a}-2 \vec{b}$ and $\overrightarrow{\mathrm{OQ}}=\vec{a}+\vec{b}$. Find the position vector of a point R which divides the line joining P and Q in the ratio $2: 1$, (i) internally, and (ii) externally. | 1 |
13. If $y=\frac{\sin ^{-1} x}{\sqrt{1-x^{2}}}$, then $\left(1-x^{2}\right) \frac{d y}{d x}$ is equal to
a. $x+y$
b. $1+x y$
c. 1-xy
d. $x y-2$ | 1 |
Is it wrong that I find it ignorant of Americans to say that America is the best country in the world? I'm American living in Sydney, Australia | 0 |
3. The electric field in the gaps between the plates and the dielectric slab will be
(a) $\frac{\varepsilon_{0} A V}{d}$
(b) $\frac{V}{d}$
(c) $\frac{K V}{d}$
(d) $\frac{V}{d-t}$ | 1 |
How can I learn Japanese as a beginner? | 0 |
1. The major product of the following reaction is :
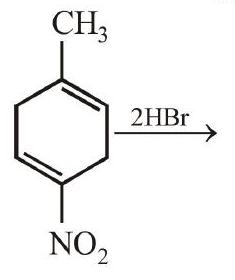
[Main Sep. 06, $2020(\mathrm{I})]$
(a)
? How efficient would it be compared to a tree of binary join operators? | 0 |
14. Let $P=(-1,0), Q=(0,0)$ and $R=(3,3 \sqrt{3})$ be three points. Then the equation of the bisector of the angle $\angle P Q R$ is
a. $\sqrt{3} / 2 x+y=0$
b. $x+m \sqrt{3} y=0$
c. $\sqrt{3} x+y=0$
d. $x+(\sqrt{3} / 2) y=0$ | 1 |
2. Find the angle between the vectors $\hat{i}-2 \hat{j}+3 \hat{k}$ and $3 \hat{i}-2 \hat{j}+\hat{k}$ | 1 |
What should you do if you want to lose a lot of weight? | 0 |
7. Water is filled in a cylindrical container to a height of $3 \mathrm{~m}$. The ratio of the cross-sectional area of the orifice and the beaker is 0.1 . The square of the speed of the liquid coming out from the orifice is $\left(g=10 \mathrm{~m} / \mathrm{s}^{2}\right)$
 $0.44 \times 10^{3} \mathrm{~kg}$
(b) $44.0 \mathrm{~kg}$
(c) $44 \mathrm{~kg}$
(d) $44.00 \mathrm{~kg}$ | 1 |
What do you wear with light blue jeans? | 0 |
69. Fusion of $\mathrm{MnO}_{2}$ with $\mathrm{KOH}$ in presence of $\mathrm{O}_{2}$ produces a salt $W$. Alkaline solution of $W$ upon electrolytic oxidation yields another salt $X$. The manganese containing ions present in $\mathrm{W}$ and $X$, respectively are $Y$ and $Z$. Correct statement(s) is (are)
(a) In both... | 1 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.