Question stringlengths 2 3k | label int64 0 1 |
|---|---|
60. The group having isoelectronic species is
(a) $\mathrm{O}^{2-}, \mathrm{F}^{-}, \mathrm{Na}^{+}, \mathrm{Mg}^{2+}$
(b) $\mathrm{O}^{-}, \mathrm{F}^{-}, \mathrm{Na}, \mathrm{Mg}^{2+}$
(c) $\mathrm{O}^{2-}, \mathrm{F}^{-}, \mathrm{Na}, \mathrm{Mg}^{2+}$
(d) $\mathrm{O}^{-}, \mathrm{F}^{-}, \mathrm{Na}^{+}, \mathrm{Mg... | 1 |
45. Give reasons for the following :
(i) Bond dissociation energy of $\mathrm{F}_{2}$ is less than that of $\mathrm{Cl}_{2}$.
(ii) Fluorine cannot be prepared from fluorides by chemical oxidation.
(iii) Anhydrous $\mathrm{HCl}$ is a bad conductor of electricity but aqueous $\mathrm{HCl}$ is a good conductor;
(... | 1 |
19. $\int_{0}^{\infty} \frac{d x}{\left[x+\sqrt{x^{2}+1}\right]^{3}}$ is equal to
a. $\frac{3}{8}$
h. $\frac{1}{8}$
c. $-\frac{3}{8}$
d. None of these | 1 |
Which is the nearest railway station to reach IIT Bombay? | 0 |
{ Example } 15.1 \text { Is }+\mathrm{CH}_2-\mathrm{CH}\left(\mathrm{C}_6 \mathrm{H}_5\right) \mathrm{t}_{\mathrm{n}} \text { a homopolymer or a copolymer? } | 1 |
31. Diborane is a potential rocket fuel which undergoes combustion according to the reaction.
$\mathrm{B}_{2} \mathrm{H}_{6}(\mathrm{~g})+3 \mathrm{O}_{2}(\mathrm{~g}) \rightarrow \mathrm{B}_{2} \mathrm{O}_{3}(\mathrm{~s})+3 \mathrm{H}_{2} \mathrm{O}(\mathrm{g})$
From the following data, calculate the enthalpy chan... | 1 |
7. System shown in figure is in equilibrium. Find the magnitude of net change in the string tension between two masses just after, when one of the springs is cut. Mass of both the blocks is same and equal to $m$ and spring constant of both the springs is $k$
(a) $\frac{m g}{2}$
(b) $\frac{m g}{4}$
(c) $\frac{m g}{3}$
(... | 1 |
What is that we as Indians are collectively doing right? | 0 |
11. If $\left(m_{i}, \frac{1}{m_{i}}\right), m_{i}>0, \mathrm{i}=1,2,3,4$ are four distinct points on a circle, then show that $m_{1} m_{2} m_{3} m_{4}=1$.
(IIT-JEE, 1989) | 1 |
How can I delete all my answers that are on Quora? | 0 |
How do I become an expert in UNIX and shell scripting? | 0 |
14. Hydrolysis of one mole of peroxodisulphuric acid produces
(a) two moles of sulphuric acid
(b) two moles of peroxomonosulphuric acid
(c) one mole of sulphuric acid and one mole of peroxomonosulphuric acid
(d) one mole of sulphuric acid, one mole of peroxomonosulphuric acid and one mole of hydrogen peroxide. | 1 |
27. The correct structure of ethylenediaminetetraacetic acid (EDTA) is
(a)
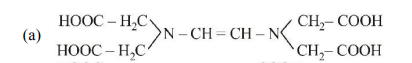
(b)
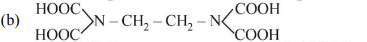
(c)
 | 1 |
What is the physical criteria for the army? | 0 |
How I can speak English fluently? | 0 |
I have forgotten my Facebook password and the number and email that was used in this Facebook account are also inactive and can't be used for recovery. Is there any other effective option available? | 0 |
What have somes studies show when researching how someone uses "gaydar"? | 0 |
Example 8.28 Let $A, B, C, D$ be (not necessarily square)
real matrices such that$A^{T}=B C D ; B^{T}=C D A ; C^{T}=D A B$ and $D^{T}=A B C$
for the matrix $S=A B C D$, prove that $S^{3}=S$. | 1 |
20. The differential coefficient of $f\left(\log _{e} x\right)$ with respect to $x$, where $f(x)=\log _{e} x$, is
a. $\frac{x}{\log _{e} x}$
b. $\frac{1}{x} \log _{e} x$
c. $\frac{1}{x \log _{e} x}$
d. None of these | 1 |
Example 35.1
A wire placed along north-south direction carries a current of $10 \mathrm{~A}$ from south to north. Find the magnetic field due to a $1 \mathrm{~cm}$ piece of wire at a point $200 \mathrm{~cm}$ northeast from the piece. | 1 |
How do I score very good marks in class 11? | 0 |
8. The eccentricity of an ellipse whose centre is at the origin is $1 / 2$. If one of its directrices is $x=-4$, then the equation of the normal to it at $1, \frac{3}{2}$ is
(a) $2 y-x=2$
(b) $4 x-2 y=1$
(c) $4 x+2 y=7$
(d) $x+2 y=4$ | 1 |
In what year was the Tamworth manifesto introduced? | 0 |
Why should one learn Tamil? | 0 |
Why is red onion actually purple? | 0 |
44. If the density of air is $\rho_{a}$, and that of the liquid $\rho_{l}$, then for a given piston speed the rate (volume per unit time) at which the liquid is sprayed will be proportional to
(a) $\sqrt{\frac{\rho_{a}}{\rho_{l}}}$
(b) $\sqrt{\rho_{a} \rho_{l}}$
(c) $\sqrt{\frac{\rho_{l}}{\rho_{a}}}$
(d) $\rho_{l}$ | 1 |
(b) Lithium is the only alkali metal to form a nitride directly. | 1 |
14. A container of large uniform cross-sectional area $A$ resting on a horizontal surface, holds two immiscible, non-viscous and incompressible liquids of densities $d$ and $2 d$, each of height $H / 2$ as shown in figure. The lower density liquid is open to the atmosphere having pressure $p_{0}$.
$(1995,5+5$ M)
$ will precipitate :
(a) $\mathrm{Hg}_{2} \mathrm{Cl}_{2}$ only
(b) $\mathrm{PbCl}_{2}$ only
(c) $\mathrm{PbCl}_{2}$ and $\mathrm{Hg}_{2} \mathrm{Cl}_{2}$
(... | 1 |
What is the difference between stainless steel grade SS304 and SUS304? | 0 |
How many elementary particles are in the universe? | 0 |
30. The hyperconjugative stabilities of tert-butyl cation and2-butene, respectively, are due to
(a) $\sigma \rightarrow \mathrm{p}$ (empty) and $\sigma \rightarrow \pi^{*}$ electron delocalisations
(b) $\sigma \rightarrow \sigma^{*}$ and $\sigma \rightarrow \pi$ electron delocalisations
(c) $\sigma \rightarrow \ma... | 1 |
What do the candles at Advent mean? | 0 |
What is the difference between yoghurt and curd? | 0 |
What is the transfer price in e-commerce? | 0 |
Will you feel something if you accidentally without knowing ingested a few specks of weed? | 0 |
Which framework will you choose? | 0 |
9. The structures of beryllium chloride in the solid state and vapour phase, respectively, are: [Main April 9, $2019($ III)]
(a) chain and chain
(b) dimeric and dimeric
(c) chain and dimeric
(d) dimeric and chain | 1 |
3. Charge stored by the $1.5 \mu \mathrm{F}$ capacitor in the steady state circuit shown is
(a) $5 \mu \mathrm{C}$
(b) $7.5 \mu \mathrm{C}$
(c) $2.5 \mu \mathrm{C}$
(d) Zero
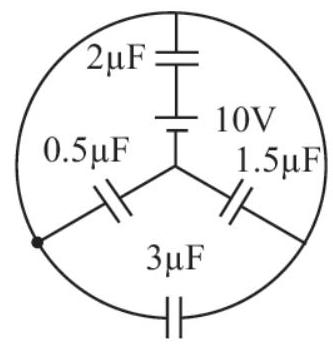 | 1 |
9. The isotopes of hydrogen are:
(a) Tritium and protium only
(b) Protium and deuterium only
(c) Protium, deuterium and tritium
(d) Deuterium and tritium only | 1 |
My phone's invoice has my address in place of my name. Will it cause any problem if I take it to the service center.? | 0 |
Who are the people with the most followers on Quora? | 0 |
8. Among the following vitamins the one whose deficiency causes rickets (bone deficiency) is
(a) Vitamin A
(b) Vitamin B
(c) Vitamin D
(d) Vitamin C | 1 |
Which grocery stores in the US sell marzipan? | 0 |
How many crop seasons are there in Rajasthan? | 0 |
Example 2.31 Evaluate $\lim _{x \rightarrow \frac{\pi}{2}} \frac{1+\cos 2 x}{(\pi-2 x)^{2}}$. | 1 |
46. An acidic solution of $\mathrm{Cu}^{2+}$ salt containing $0.4 \mathrm{~g}$ of $\mathrm{Cu}^{2+}$ is electrolysed until all the copper is deposited. The electrolysis is continued for seven more minutes with the volume of solution kept at $100 \mathrm{~mL}$. and the current at $1.2 \mathrm{amp}$. Calculate the volume... | 1 |
54. Refer to Q. 53. The incorrect statement among the following for this reaction is
(a) decrease in total pressure will result in the formation of more moles of gaseous $X$.
(b) At the start of the reaction, dissociation of gaseous $X_{2}$ takes place spontaneouly.
(c) $\beta_{\text {eqb. }}=0.7$
(d) $K_{C}<1$ | 1 |
15. Resistance of $0.2 \mathrm{M}$ solution of an electrolyte is $50 \Omega$. The specific conductance of the solution is $1.4 \mathrm{~S} \mathrm{~m}^{-1}$. The resistance of $0.5 \mathrm{M}$ solution of the same electrolyte is $280 \Omega$. The molar conductivity of $0.5 \mathrm{M}$ solution of the electrolyte in $\m... | 1 |
What is phase in waves? | 0 |
5. Let $g(x)=f(x) \sin x$, where $f(x)$ is a twice differentiable $L_{1}$ function on $(-\infty, \infty)$ such that $f^{\prime}(-\pi)=1$. The value of $\left|g^{\prime \prime}(-\pi)\right|$ equals | 1 |
25. The ratio of their common difference is
a. 12
b. 24
c. 26
d. 9 | 1 |
What is SAP hybris? | 0 |
Who founded what came to be known Hyderabad? | 0 |
The historical aspect of cartooning can be applied to mass reproduction or what? | 0 |
Which branch of government promulgates state regulations? | 0 |
Is there more to the wave-particle duality than semantics? | 0 |
7. The segment $A B$ of wire carrying current $I_{1}$ is placed perpendicular to a long straight wire carrying current $I_{2}$ as shown in figure. The magnitude of force experienced by the straight wire $A B$ is
(a) $\frac{\mu_{0} I_{1} I_{2}}{2 \pi} \ln 3$
(b) $\frac{\mu_{0} I_{1} I_{2}}{2 \pi} \ln 2$
(c) $\frac{2 \mu... | 1 |
10. Find the component of vector $\overrightarrow{\mathbf{A}}=(2 \hat{\mathbf{i}}+3 \hat{\mathbf{j}})$ along the direction $(\hat{\mathbf{i}}-\hat{\mathbf{j}})$
(a) $-\frac{1}{2}(\hat{i}-\hat{j})$
(b) $-\frac{1}{2}(\hat{i}+\hat{j})$
(c) $\frac{1}{2}(\hat{i}-\hat{j})$
(d) $\frac{1}{2}(\hat{i}+\hat{j})$ | 1 |
What belief was considered sacred by ancient civilizations? | 0 |
14. The standard enthalpy of formation of $\mathrm{NH}_{3}$ is $-46.0 \mathrm{~kJ} / \mathrm{mol}$. If the enthalpy of formation of $\mathrm{H}_{2}$ from its atoms is $-436 \mathrm{~kJ} / \mathrm{mol}$ and that of $\mathrm{N}_{2}$ is $-712 \mathrm{~kJ} / \mathrm{mol}$, the average bond enthalpy of $\mathrm{N}-\mathrm{H... | 1 |
32. In a class of 140 students numbered 1 to 140 , all even numbered students opted Mathematics course, those whose number is divisible by 3 opted Physics course and those whose number is divisible by 5 opted Chemistry course. Then, the number of students who did not opt for any of the three courses is
(a) 42
(b) 1... | 1 |
37. A capillary glass tube records a rise of $20 \mathrm{~cm}$ when dipped in water. When the area of cross-section of the tube is reduced to half of the former value, water will rise to a height of
(a) $10 \sqrt{2} \mathrm{~cm}$
(b) $10 \mathrm{~cm}$
(c) $20 \mathrm{~cm}$
(d) $20 \sqrt{2} \mathrm{~cm}$ | 1 |
85. The value of $\cos 2(\theta+\phi)+4 \cos (\theta+\phi) \sin \theta \sin \phi+2 \sin ^{2} \phi$ is
a. independent of $\theta$ only .
$\mathbf{h}$ independent of $\phi$ only
c. independent of both $\theta$ and $\phi$
d dependent on $\theta$ and $\phi$ | 1 |
What type of language is Maltese? | 0 |
3. Consider the nuclear fission reaction $W \rightarrow X+Y$. What is the $Q$-value (energy released) of the reaction?
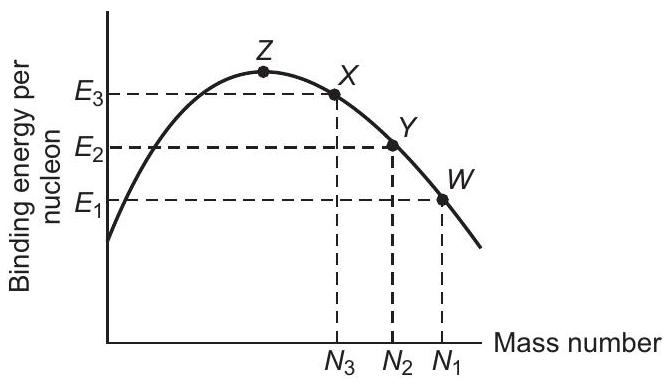
(a) $E_{1} N_{1}-\left(E_{2} N_{2}+E_{3} N_{3}\right)$
(b) $\left(E_{2... | 1 |
What are the steps of starting a manufacturing company? | 0 |
29. Consider the ratio $r=\frac{(1-a)}{(1+a)}$ to be determined by measuring a dimensionless quantity $a$. If the error in the measurement of a is $\Delta a(\Delta a / a \ll 1$, then what is the error $\Delta r$ in determining $r$ ?
(a) $\frac{\Delta a}{(1+a)^{2}}$
(b) $\frac{2 \Delta a}{(1+a)^{2}}$
(c ) $\frac{2 \D... | 1 |
What is the best all-mountain bike? | 0 |
33. Let $f(x)=x^{3}+3 x+2$ and $g(x)$ is the inverse of it. Then the area bounded by $g(x)$, the $x$-axis and the ordinate at $x=-2$ and $x=6$ is
a. $1 / 4$ sq. units
c. $5 / 4$ sq. units
d. $7 / 3$ sq. units
h. $4 / 3$ sq. units | 1 |
1. The value of ratio $M / m$ is
(a) $2: 3$
(b) $3: 2$
(c) $4: 3$
(d) $3: 4$ | 1 |
3. A proton and an $\alpha$-particle (with their masses in the ratio of $1: 4$ and charges in the ratio of $1: 2$ ) are accelerated from rest through a potential difference $V$. If a uniform magnetic field $B$ is set up perpendicular to their velocities, the ratio of the radii $r_{p}: r_{\alpha}$ of the circular paths ... | 1 |
What group repeatedly tried and failed to conquer the Ahom? | 0 |
What is the best Photoshop app for an Android tablet? | 0 |
Ex. 27. A solution of a 0.4 -g sample of $\mathrm{H}_{2} \mathrm{O}_{2}$ reacted with 0.632 g of $\mathrm{KMnO}_{4}$ in the presence of sulphuric acid. Calculate the percentage purity of the sample of $\mathrm{H}_{2} \mathrm{O}_{2}$. | 1 |
10. Consider a system of three charges $q / 3, q / 3$ and $-2 q / 3$ placed at points $A, B$ and $C$ respectively, as shown in the figure. Take $O$ to be the centre of the circle of radius $R$ and angle $C A B=60^{\circ}$
 $p$
(b) $2 p$
(c) $p / 2$
(d) $p^{2}$ | 1 |
37. A circle passes through the points $A(1,0), B(5,0)$ and touches the $y$-axis at $C(0, h)$. If $\angle A C B$ is maximum then
a. $h=3 \sqrt{5}$
b. $h=2 \sqrt{5}$
c. $h=\sqrt{5}$
d. $h=2 \sqrt{10}$ | 1 |
Ex. 59. Bond energies of $\mathrm{F}_{2}$ and $\mathrm{Cl}_{2}$ are respectively 36.6 and 58.0 kcal per mole. If the heat liberated in the reaction $\mathrm{F}_{2}+\mathrm{Cl}_{2} \rightarrow 2 \mathrm{FCl}$ is 26.6 kcal , calculate the bond energy of $\mathrm{F}-\mathrm{Cl}$ bond. | 1 |
What is the property tax rate in Granville, Ohio? How is it compared to the one of Georgia? | 0 |
What was the actual intention of the British to invade India? | 0 |
What does chansons de geste mean in English? | 0 |
(iv) $x=2$ and $x=3$ are the roots of the equation $3 x^{2}-x-10=0$. | 1 |
How do you plant flower bulbs? | 0 |
14. In a body-centred cubic arrangement the ion A occupies the centre while the ions of B occupy the corners of a cube. The formula of the crystal is
(a) AB
(b) $\mathrm{A}_{2} \mathrm{~B}$
(c) $\mathrm{AB}_{2}$
(d) $\mathrm{AB}_{3}$ | 1 |
What is the salary of a grade b officer? | 0 |
Why do I not like making friends? | 0 |
What is the best way to teach yourself piano? | 0 |
7. The variation of induced emf $(e)$ with time $(t)$ in a coil if a
short bar magnet is moved along its axis with a constant velocity is best represented as
 and Item - II (reagent) for the preparation of benzaldehyde is :

(a) (I) - (Q), (II) - (R) and (III) - (P)
(b) (I) - (P), (II) - (Q) and (III) - (R)
(c) $(... | 1 |
Singapore & Vietnam shows which influence predominantly? | 0 |
What are the major themes of Mr. Robot season 2? | 0 |
Subsets and Splits
No community queries yet
The top public SQL queries from the community will appear here once available.